Clear Sky Science · en
NALIRIFOX versus gemcitabine plus nab-paclitaxel in Chinese patients with advanced pancreatic adenocarcinoma: a randomized, open-label phase II trial
Why this study matters
Pancreatic cancer is one of the deadliest cancers, in part because it is usually discovered only after it has quietly spread. For many patients, surgery is no longer possible and chemotherapy becomes the main hope for slowing the disease. This study tested whether a newer drug combination called NALIRIFOX could keep the cancer in check longer, and with acceptable side effects, compared with a widely used standard treatment in Chinese patients.
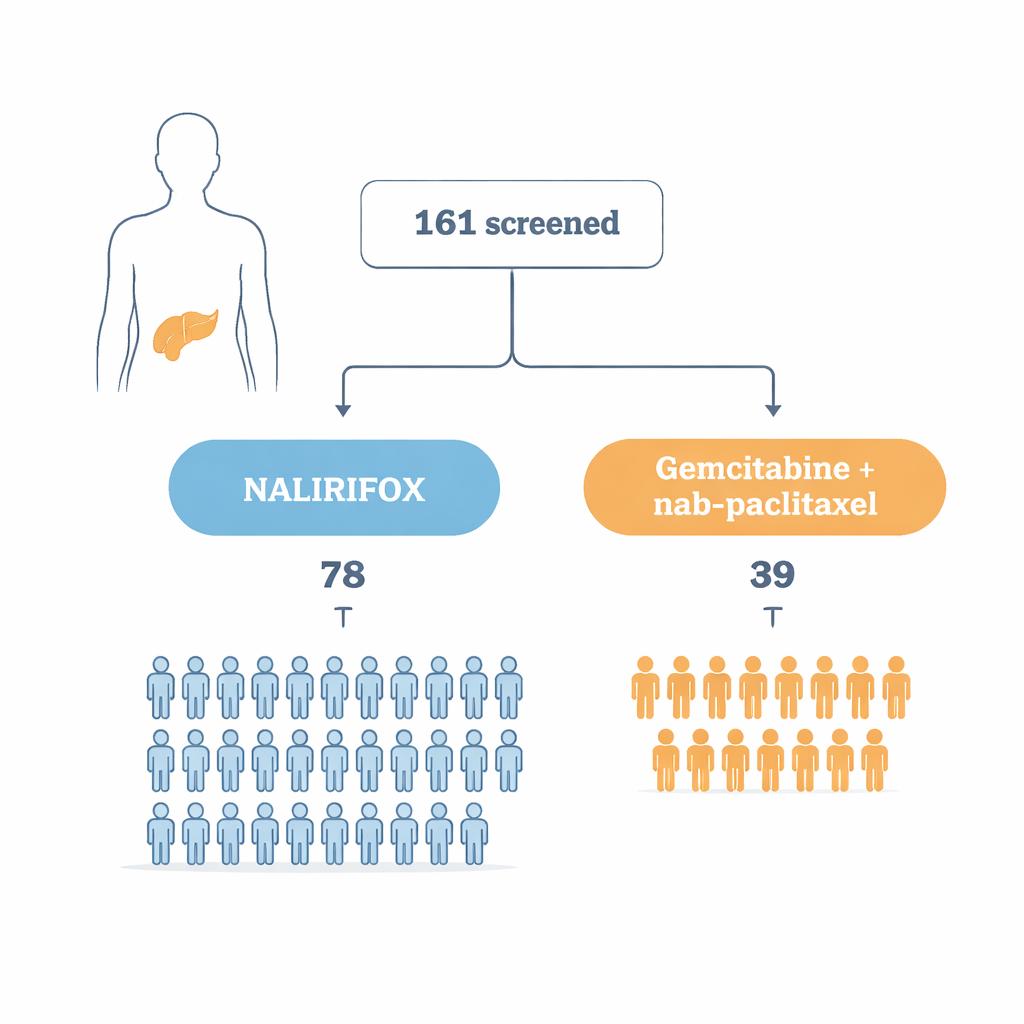
Two treatment paths, one difficult disease
The researchers focused on adults in China with advanced pancreatic adenocarcinoma, the most common form of pancreatic cancer, whose tumors could not be removed surgically. All patients were strong enough for combination chemotherapy. They were randomly assigned, in a 2-to-1 ratio, to receive either NALIRIFOX or the standard regimen of gemcitabine plus nab-paclitaxel. Random assignment and similar starting characteristics between groups helped ensure that any differences in outcomes were likely due to the treatments rather than to who happened to enroll.
Measuring extra time before the cancer worsens
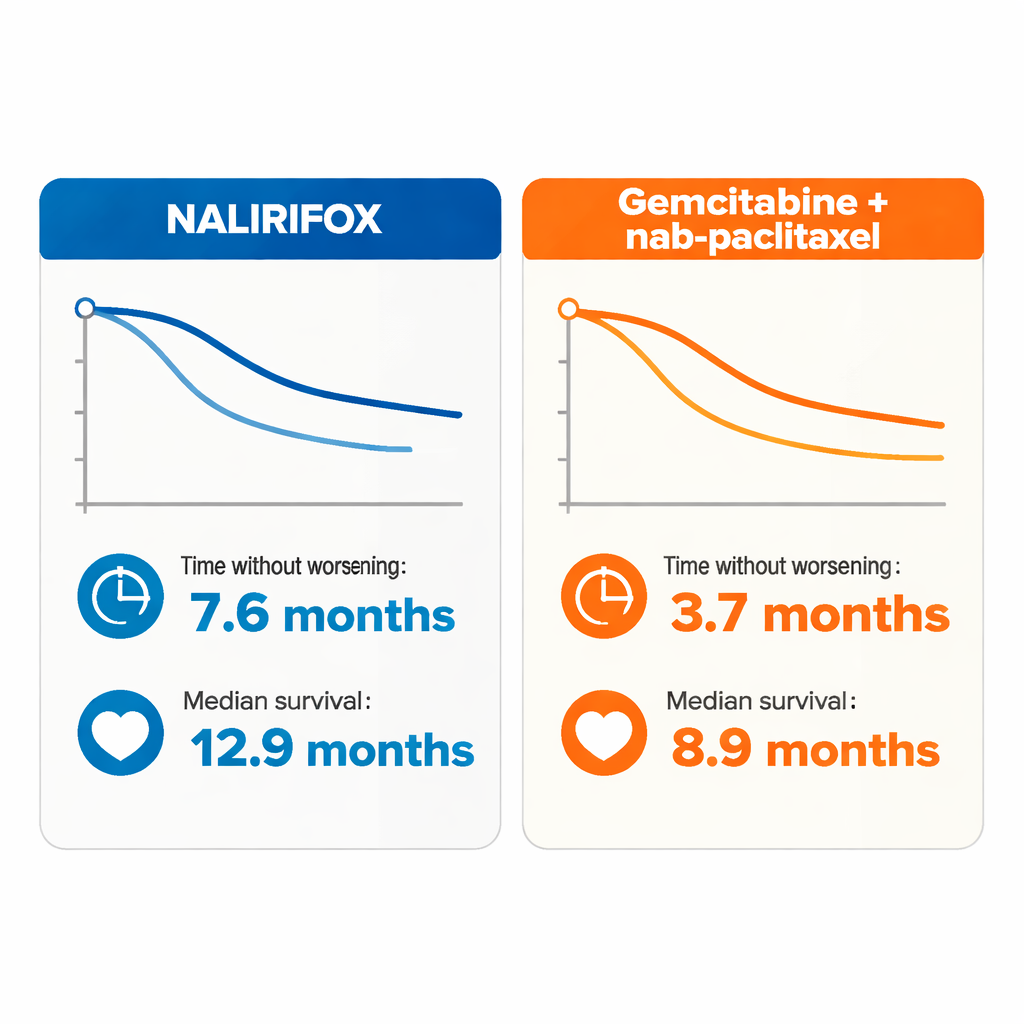
The main yardstick was “progression-free survival,” which simply means how long patients lived before scans showed their cancer growing or they died. With NALIRIFOX, the middle value (median) was 7.6 months, compared with 3.7 months for gemcitabine plus nab-paclitaxel. In other words, patients on NALIRIFOX, on average, enjoyed about four additional months before their disease clearly worsened. This advantage was seen across many subgroups, including people with liver metastases and those with more extensive spread, groups that usually fare poorly.
Living longer and shrinking tumors
The study also looked at overall survival, or how long patients lived from the start of treatment. Those receiving NALIRIFOX had a median survival of 12.9 months, compared with 8.9 months for the standard regimen. While this survival improvement did not reach strict statistical significance in this phase II trial, the trend favored NALIRIFOX in most patient subgroups. Tumor shrinkage told a similar story: about one in three patients on NALIRIFOX saw their tumors notably shrink, versus about one in five on gemcitabine plus nab-paclitaxel. When tumors did respond, the benefit lasted longer with NALIRIFOX—about 8.5 months versus 3.8 months.
Balancing benefit with side effects
Strong chemotherapy almost always brings side effects, and both regimens were intensive. Nearly all patients had some treatment-related problems such as low blood counts, nausea, or fatigue. Serious side effects (the most severe grades) occurred in about seven out of ten NALIRIFOX patients and more than eight out of ten patients on gemcitabine plus nab-paclitaxel. Low white blood cell counts, which can raise infection risk, were actually more frequent and more severe in the standard-treatment group. NALIRIFOX caused more diarrhea, especially delayed diarrhea starting a day or more after infusion, but episodes were usually manageable. Genetic testing suggested that certain inherited differences in drug-processing genes may predict who is more likely to develop severe drops in white blood cells when receiving NALIRIFOX.
What this means for patients
Despite being stopped early and including fewer patients than originally planned, this trial shows that NALIRIFOX can give people with advanced pancreatic cancer in China more time before their disease worsens, and possibly extra months of life, compared with a widely used standard regimen. The side effects were significant but generally controllable and not clearly worse overall than those of the existing option. Together with larger international studies, these results support NALIRIFOX as a strong first-line choice for many patients and help tailor treatment guidelines to better reflect the needs and responses of Chinese populations.
Citation: Gao, C., Zhang, Y., Qu, X. et al. NALIRIFOX versus gemcitabine plus nab-paclitaxel in Chinese patients with advanced pancreatic adenocarcinoma: a randomized, open-label phase II trial. Nat Commun 17, 1715 (2026). https://doi.org/10.1038/s41467-026-68409-0
Keywords: pancreatic cancer, chemotherapy, NALIRIFOX, clinical trial, Chinese patients