Clear Sky Science · en
Tissue-specific and sex-biased glycoproteomic landscape of Schistosoma mansoni
Why Sugary Coats on Parasites Matter
Schistosomiasis is a debilitating disease that affects hundreds of millions of people, and it is currently fought with just one main drug that does not stop people from getting reinfected. The culprit is the blood-dwelling worm Schistosoma mansoni, which hides from our immune system partly by decorating its proteins with complex chains of sugars. This study maps those sugar coatings in unprecedented detail, revealing how they differ across worm tissues and between males and females, and pointing to new ways to design vaccines and treatments.
Exploring the Parasite’s Sugar Armor
Proteins on and inside cells are often covered with branching sugar chains, a process called glycosylation. In parasites, these sugar decorations can make the difference between being destroyed by the immune system or slipping past unnoticed. Until now, most research on S. mansoni sugars focused on broad mixtures rather than specific proteins and exact sugar sites. In this work, researchers used advanced mass spectrometry tools to directly read intact sugar–protein combinations from adult male and female worms. They cataloged thousands of individual sugar sites on hundreds of proteins, building the first large-scale "glycoprotein atlas" for this parasite. 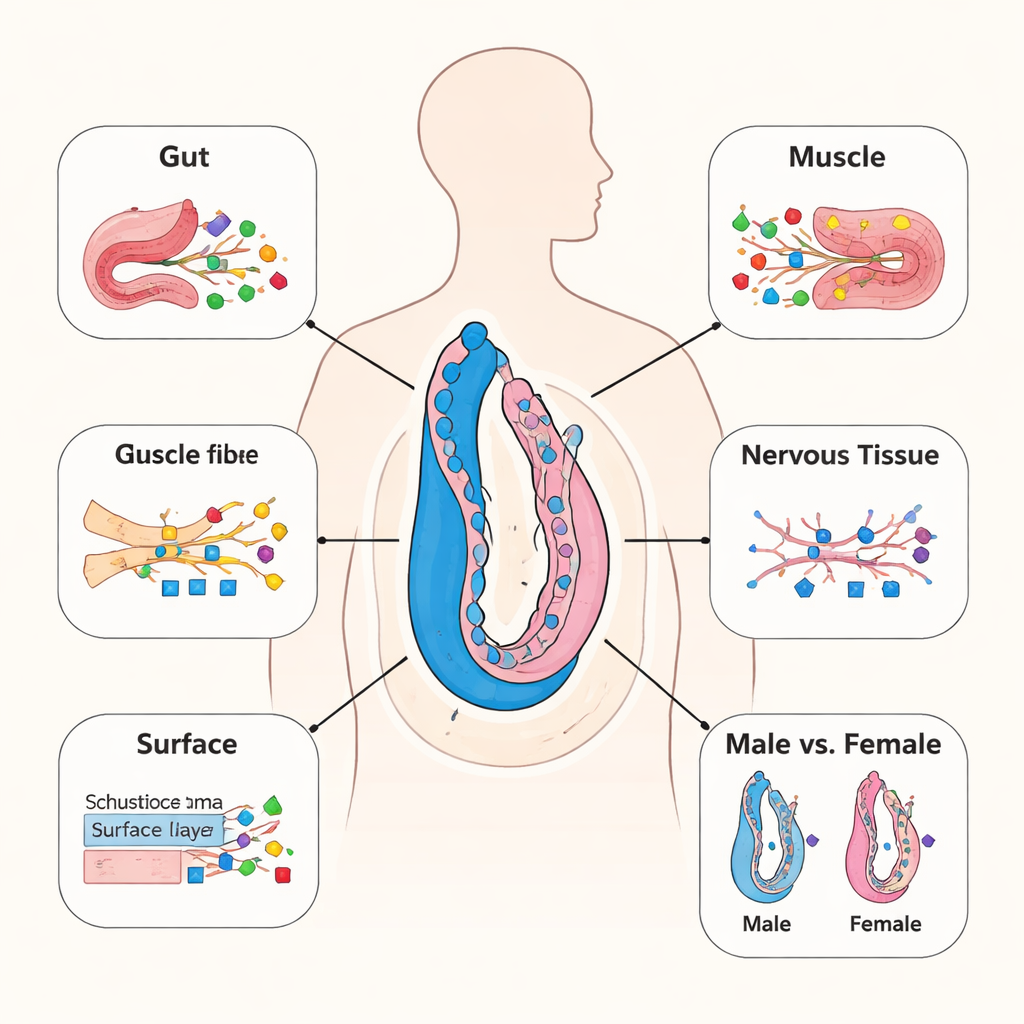
Different Tissues, Different Sugar Patterns
Not all worm tissues wear the same sugary coat. By combining their protein data with single-cell gene maps, the team connected glycosylated proteins to particular worm organs, such as the gut, muscles, outer surface (tegument), and reproductive glands. They found that the gut and inner body tissue (parenchyma) carry especially complex and diverse sugar chains, often with many sugar units and multiple modified sites on a single protein. In contrast, muscles and nerve cells tend to use smaller, simpler sugar patterns. Some sugar types, including those containing fucose, xylose, or a special sugar called hexuronic acid, were enriched in particular tissues like the egg-forming glands or the worm’s surface, hinting that these structures may shape how the parasite feeds, moves, and interacts with the host’s immune system.
How Male and Female Worms Differ
Schistosoma mansoni has separate sexes, and male and female worms play very different roles in infection and egg laying. The study shows that their sugar decorations differ as well. Many glycoproteins and specific sugar sites are more abundant in males, especially in muscles, neurons, and surface tissues, which may support movement and pairing. Females, on the other hand, show stronger glycosylation in the gut and in glands that produce eggs, consistent with their role in digestion and reproduction. Although the overall types and sizes of sugar chains are similar between the sexes, the detailed composition—how many of each sugar building block and how many fucose units—shifts in a sex-biased way. This suggests that the same protein can be fine-tuned in males and females simply by altering its sugar coat. 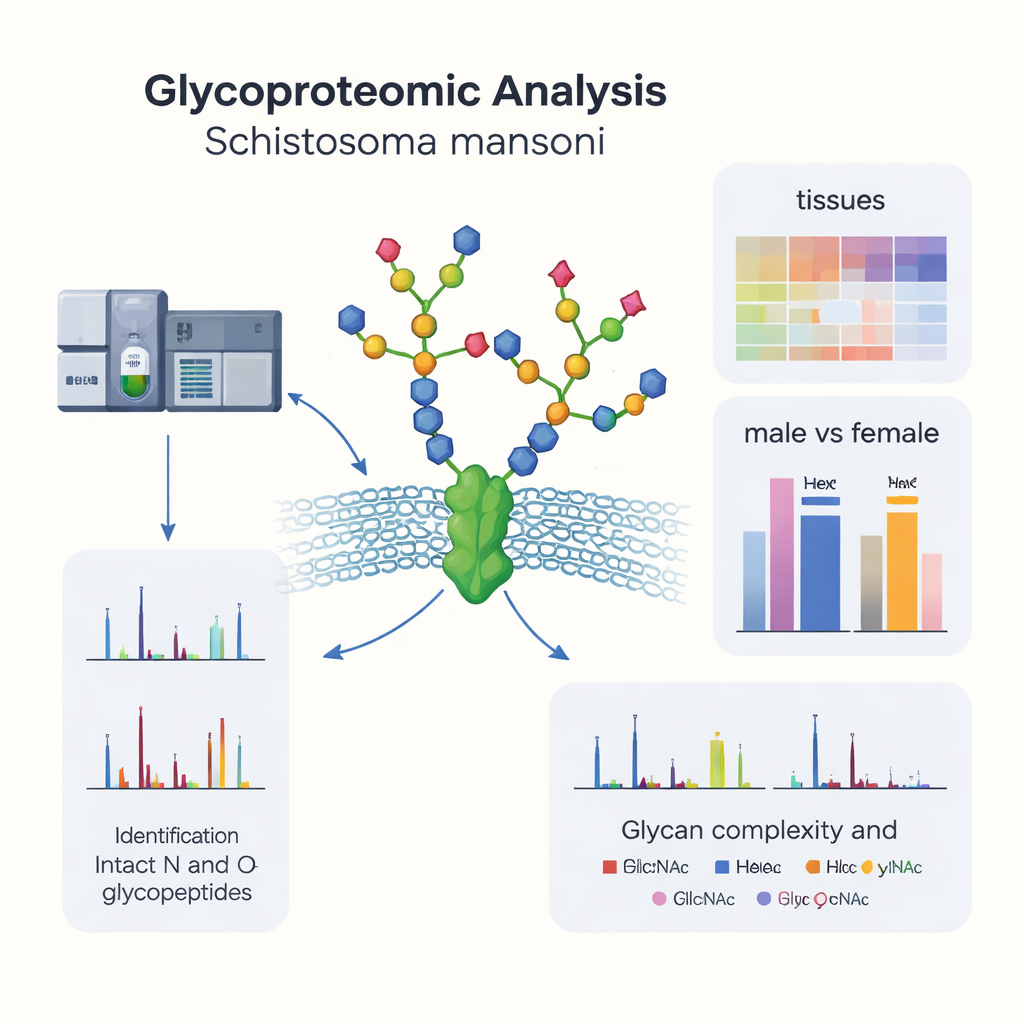
Unusual Sugars and Essential Enzymes
Beyond cataloging known sugar patterns, the researchers also uncovered unusual sugar compositions and confirmed the presence of hexuronic acid-based structures that had been suspected but not well defined in adult worms. They showed that most of the parasite’s sugar chains differ from those of common laboratory animals, yet are somewhat similar to those in mice, perhaps reflecting an evolutionary strategy to blend into mammalian hosts while retaining parasite-specific features. To test how important these modifications are, the team used RNA interference to switch off four key enzymes that build N-linked and O-linked sugars. Knocking down these enzymes damaged the worms’ outer surface, intestines, and overall health, in some cases killing them. This confirms that proper glycosylation is vital for the parasite’s survival.
New Clues for Vaccine Design
Because the host immune system mostly “sees” the parasite’s outer and gut-facing proteins, the authors focused on glycoproteins at this host–parasite interface, including several well-known vaccine candidates such as Sm25, Sm29, and Cathepsin B. They showed that these proteins carry distinct and sometimes highly complex sugar patterns, including multi-fucosylated and xylose-containing chains that are known to provoke strong immune reactions in animals. The study also pinpoints the exact sites where sugars attach and shows which sugar forms are more common in males or females. For vaccine developers, this map is crucial: it suggests that mimicking the natural, sugar-decorated versions of these proteins—rather than using bare or differently glycosylated recombinant forms—may greatly improve protection. Overall, this work turns the parasite’s sugary disguise into a detailed blueprint for designing smarter vaccines and new ways to weaken or kill the worms.
Citation: Chen, X., You, Y., Liu, W. et al. Tissue-specific and sex-biased glycoproteomic landscape of Schistosoma mansoni. Nat Commun 17, 1696 (2026). https://doi.org/10.1038/s41467-026-68400-9
Keywords: schistosomiasis, Schistosoma mansoni, glycosylation, parasite vaccines, glycoproteomics