Clear Sky Science · en
A nanosystem targeting tissue inhibitor of metalloproteinase-1 for continuous spatiotemporal idiopathic pulmonary fibrosis therapy
Why stubborn lung scarring matters
Idiopathic pulmonary fibrosis (IPF) is a relentless lung disease in which normal, sponge‑like lung tissue is slowly replaced by stiff scar tissue. People with IPF become short of breath doing everyday activities, and most survive only a few years after diagnosis. Today’s drugs can slow the disease for some, but they rarely undo existing scars and often cause side effects. This study explores a smart, inhaled “nano‑therapy” designed not just to slow IPF, but to actively clear scar tissue and help lungs repair themselves.
The problem: sticky scars and chemical stress
In IPF, the spaces where oxygen normally moves into the blood fill up with excess proteins such as collagen, turning flexible air sacs into rigid patches. The authors focused on a protein called TIMP‑1, which acts like a brake on natural enzymes that normally chew up extra collagen. By examining lung samples from patients and from mice with induced lung scarring, they found TIMP‑1 levels were several times higher than in healthy lungs and rose in step with collagen buildup. At the same time, damaged lungs showed surging levels of reactive oxygen species (ROS)—chemical “sparks” that injure cells—and a loss of key markers of healthy air‑sac lining cells. Together, these findings pointed to a vicious cycle: too much TIMP‑1 slows scar breakdown, while oxidative stress further damages lung structure.
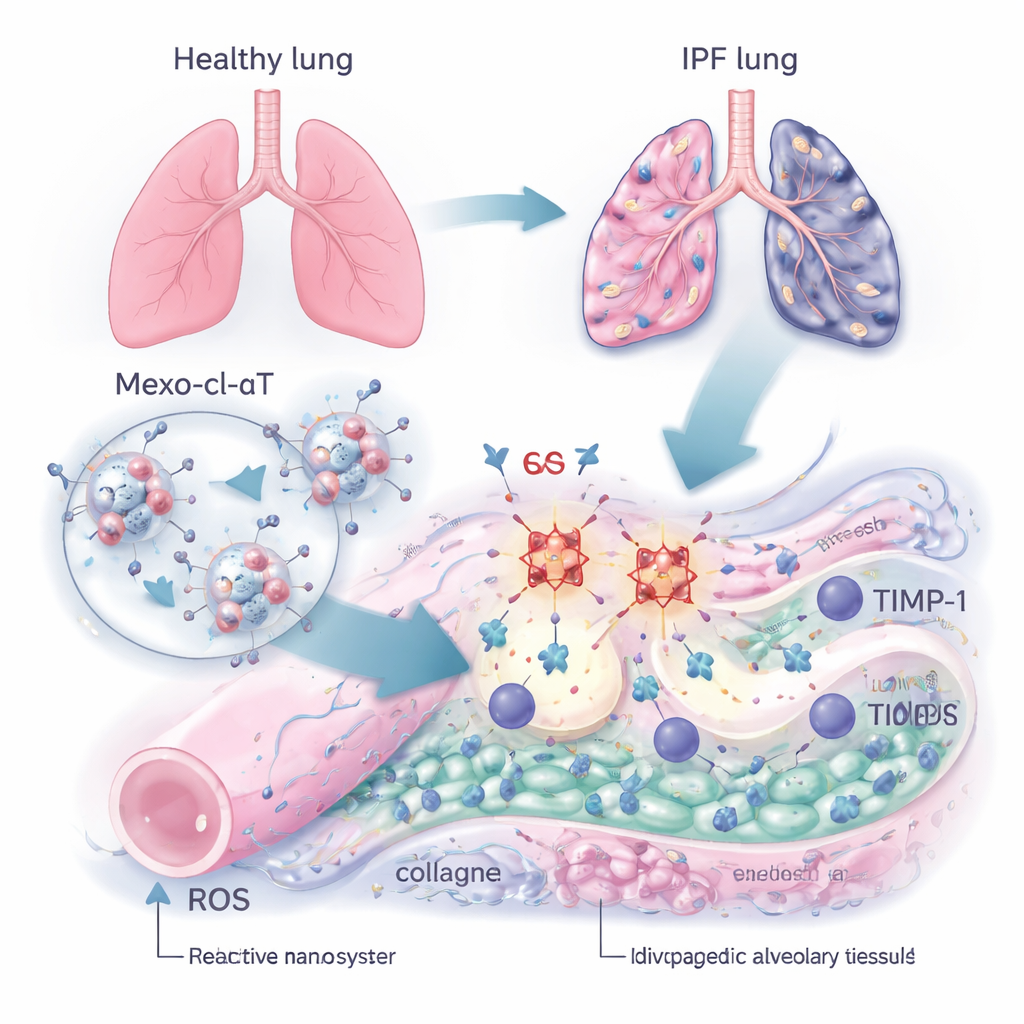
A smart delivery vehicle: tiny repair packages
To break this cycle, the team built a nanoscale therapy called Mexo‑cl‑aT. They started with exosomes—natural, bubble‑like particles released by stem cells that are already known to home in on injured tissues and support healing. Onto the surface of these exosomes they attached antibodies that specifically latch onto TIMP‑1. The connection between the exosome and the antibody was made using a special chemical linker that can be cut by ROS. In other words, the harsh chemical environment of a scarred lung is used as a signal: when the exosome arrives in a damaged region rich in ROS, the linker breaks, soaking up some of the ROS and releasing the antibody right where TIMP‑1 is concentrated, while the exosome itself continues to promote tissue repair.
How the nanosystem behaves and works
In cell studies, the researchers showed that Mexo‑cl‑aT stays stable in fluid but rapidly lets go of its antibodies when exposed to levels of hydrogen peroxide similar to those in diseased lungs. At the same time, it sharply lowers the amount of peroxide present, confirming its ROS‑scavenging role. In scar‑like cell models, the released antibodies reduced TIMP‑1 levels and freed up enzymes that break down collagen, while the exosome component decreased cell death, boosted cell growth, and sped wound closure in both lung and blood‑vessel cells. Compared with versions where the linker could not be cut, the ROS‑responsive design removed more collagen and reduced chemical stress more effectively, highlighting the importance of controlled release.
Testing in a severe lung‑scarring model
The team then tested the therapy in mice with advanced lung fibrosis triggered by the drug bleomycin, a model chosen to mimic late‑stage IPF. A single dose of inhaled Mexo‑cl‑aT lingered in the lungs for days and held onto more antibody than a simple mixture of its parts. Treated lungs looked healthier to the naked eye and under the microscope: air spaces reopened, scar thickness dropped, and total collagen content nearly returned to normal. Markers of activated scar‑forming cells fell, while proteins linked to healthy air‑sac and blood‑vessel lining cells rose. Chemical measurements showed that the treatment sharply cut TIMP‑1 levels, restored the balance of collagen‑clearing enzymes, and removed about three‑quarters of excess ROS. Importantly, measures of inflammation decreased, and blood tests and organ exams did not reveal obvious toxicity.
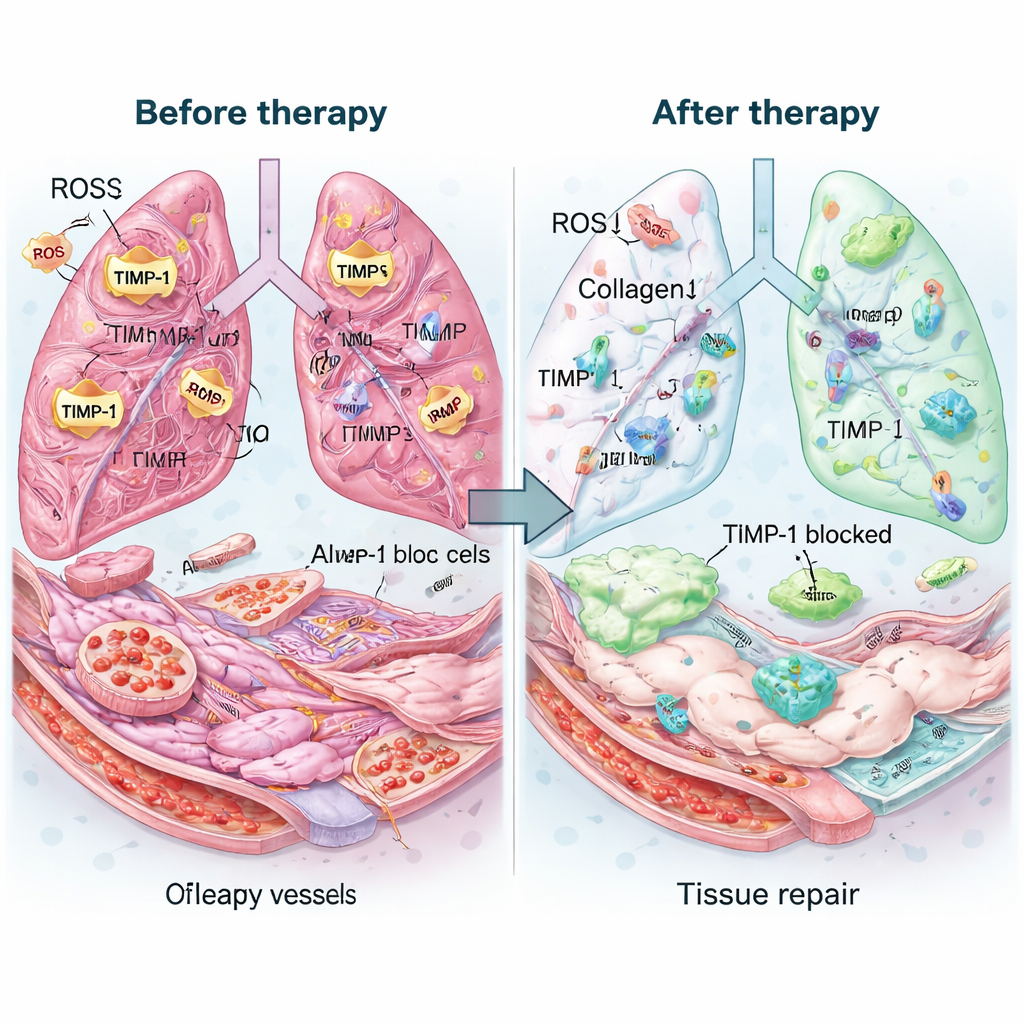
What this could mean for patients
For people living with IPF, this work does not yet offer an immediate cure, but it introduces a promising strategy. Instead of only slowing new damage, the Mexo‑cl‑aT nanosystem aims to actively undo existing scars, calm harmful chemical stress, and support rebuilding of fragile lung tissue—all in a targeted, inhaled form that concentrates treatment where it is needed most. While challenges such as large‑scale exosome production and testing in humans remain, the study suggests that precision nano‑therapies tuned to the diseased environment could one day transform how stubborn lung scarring, and perhaps other fibrotic diseases, are treated.
Citation: Li, C., Lu, G., Chen, H. et al. A nanosystem targeting tissue inhibitor of metalloproteinase-1 for continuous spatiotemporal idiopathic pulmonary fibrosis therapy. Nat Commun 17, 1694 (2026). https://doi.org/10.1038/s41467-026-68398-0
Keywords: idiopathic pulmonary fibrosis, lung scarring, nanomedicine, exosome therapy, TIMP-1