Clear Sky Science · en
Injured epithelial cell states impact kidney allograft survival after T-cell-mediated rejection
Why this kidney research matters to patients
Kidney transplants can be life‑saving, but many fail years after surgery even when doctors seem to control the immune attack known as T‑cell–mediated rejection. This study asks a simple but crucial question: if the immune reaction looks “treated” under the microscope, why do some transplanted kidneys still wear out early? By zooming in on individual cells in transplanted kidneys from mice and humans, the researchers uncover a hidden layer of damage inside the kidney’s filtering tubes that helps explain which grafts survive and which do not.
Looking beyond classic rejection signs
Doctors usually diagnose T‑cell–mediated rejection by seeing white blood cells invading the kidney and attacking the tiny tubes that process urine. Powerful anti‑rejection drugs can often drive these immune cells away and make the biopsy look better. Yet patients with this form of rejection still face a high risk of graft failure. Earlier large‑scale gene studies hinted that signals of tissue injury inside kidney cells predict outcome better than measures of how many immune cells are present. The authors set out to define these injury signals at single‑cell resolution and to test how closely they are tied to long‑term transplant survival.
Dissecting rejection one cell at a time
The team first used a controlled mouse model, transplanting kidneys either between genetically identical mice or between mismatched strains to trigger rejection. They then applied single‑nucleus RNA sequencing, which reads the activity of thousands of genes in individual cells, and spatial transcriptomics, which shows where those cells sit within the tissue slice. These tools revealed that, during rejection, the strongest molecular changes occur not in immune cells but in the kidney’s own epithelial cells that line the proximal tubules and a segment called the thick ascending limb. In these areas, cells switched on injury and stress genes, inflammatory signals and markers of a more primitive, less specialized state, while turning down transport functions needed for healthy filtration.
Hidden pockets of severely injured tubule cells
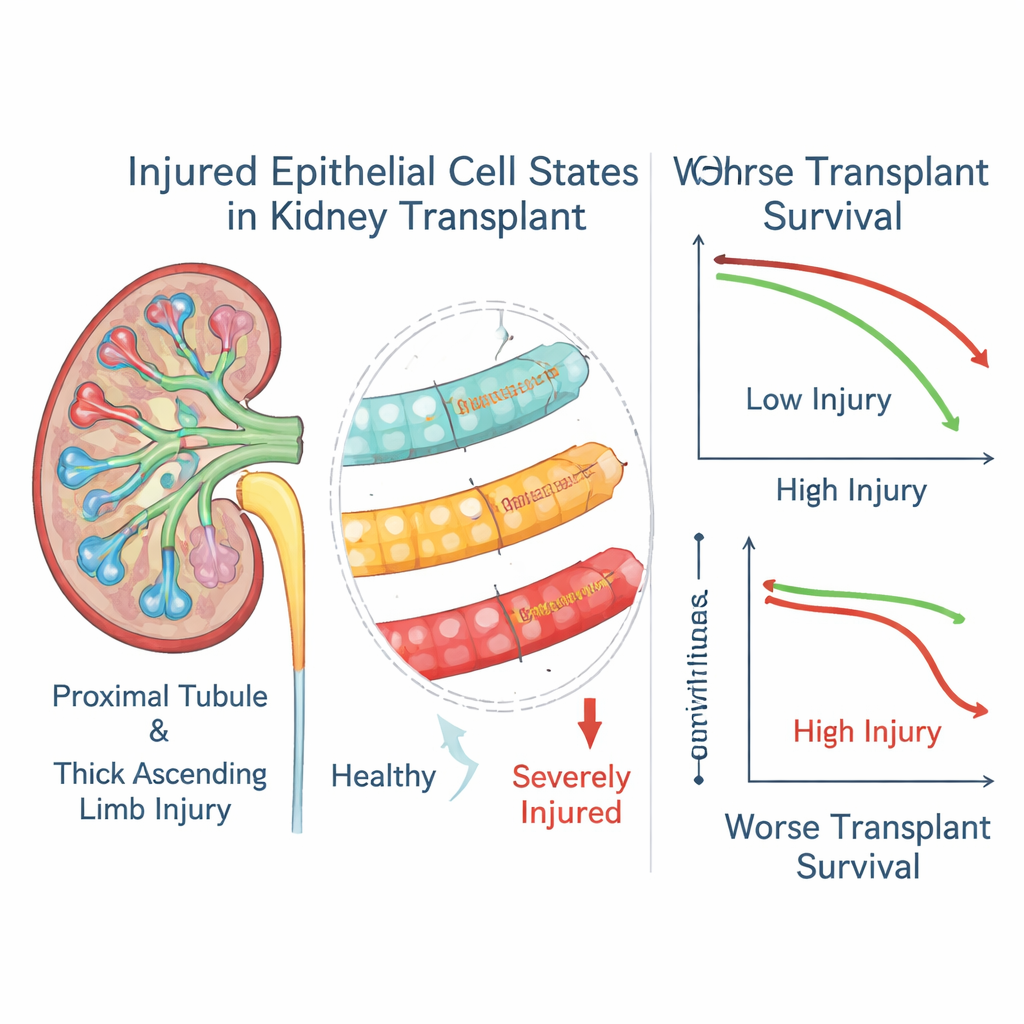
By clustering cells with similar gene activity, the researchers discovered several distinct “injury states” among tubular cells in the mouse kidneys. Some reflected early stress responses, while others represented severely injured, highly altered cells that had lost normal identity and produced many inflammatory and scarring‑related molecules. Spatial mapping showed that these cell states did not spread evenly: certain severe injury states formed patchy pockets across the kidney cortex and outer medulla and sat in complex neighborhoods with T cells, macrophages and fibroblasts. In contrast, some injured cells in deeper regions were surprisingly isolated from immune cells, hinting at different ways damage can arise and persist.
Matching mouse injury states to human transplants
The authors then asked whether similar cell states exist in people. They analyzed biopsies from human kidney transplants with active T‑cell–mediated rejection and from stable grafts. Single‑cell data again revealed injured proximal tubule and thick ascending limb cells whose gene patterns closely matched the most severely injured mouse cell states. Using these human signatures, the team “deconvoluted” thousands of older bulk biopsy gene‑expression profiles and found that severely injured tubule states were more common in T‑cell–mediated rejection and mixed rejection than in antibody‑only rejection or no rejection. Importantly, they built compact gene sets representing each injured state and scored them in a large cohort with detailed follow‑up.
Injured tubule cells as predictors of graft fate
When the authors linked these gene scores to three‑year outcomes in over a thousand transplant recipients, a clear pattern emerged. High levels of markers for the most severely injured epithelial states in both proximal tubules and thick ascending limbs were strongly associated with a higher chance of graft loss, even among patients whose biopsies were otherwise classified as treated rejection. A milder tubule injury state showed the opposite trend and was linked to better survival, suggesting it may represent a more successful repair program. In a subset of patients with repeated biopsies, the “severe injury” scores often stayed high long after the rejection episode had apparently resolved, implying that these altered cells can linger and continue to shape disease.
What this means for transplant care
For non‑specialists, the key message is that not all damage in a rejected kidney comes directly from immune cells, and not all injury disappears when the immune attack is quieted. This study shows that stubborn pockets of badly injured tubule cells act as an early warning system for future graft failure. In the long run, simple tests based on these injury gene signatures—possibly even using cells shed into the urine—could help doctors identify high‑risk patients, tailor treatments and monitor whether kidneys truly recover. The work also points to these injured epithelial cells themselves as potential targets for new therapies aimed at protecting transplanted kidneys beyond standard immune suppression.
Citation: Pfefferkorn, A.M., Jahn, L., Gauthier, P.T. et al. Injured epithelial cell states impact kidney allograft survival after T-cell-mediated rejection. Nat Commun 17, 1060 (2026). https://doi.org/10.1038/s41467-026-68397-1
Keywords: kidney transplant rejection, tubule cell injury, single-cell sequencing, transplant survival, epithelial cell states