Clear Sky Science · en
CD39 polymorphism enables lung thrombosis in sickle cell disease
Why this matters for people with sickle cell disease
Sickle cell disease affects millions worldwide and often damages the lungs, sometimes leading to sudden, life-threatening blood clots. Yet only some patients develop these dangerous lung clots, even though they all share the same underlying blood problem. This study uncovers a built-in protective system in many people with sickle cell disease—and explains why a genetic variant can switch that protection off for an at-risk subset of patients.
A tug-of-war inside sickled blood
In sickle cell disease, red blood cells break apart more easily than normal. When they burst, they release a small molecule called ADP into the bloodstream. ADP acts like an emergency flare for platelets, the cells that form clots. Too much ADP in the lungs can trigger clumps of platelets that plug tiny lung arteries, causing painful and sometimes fatal episodes of respiratory failure. The puzzle has been why this does not happen in every patient, even though all of them experience ongoing red blood cell damage.
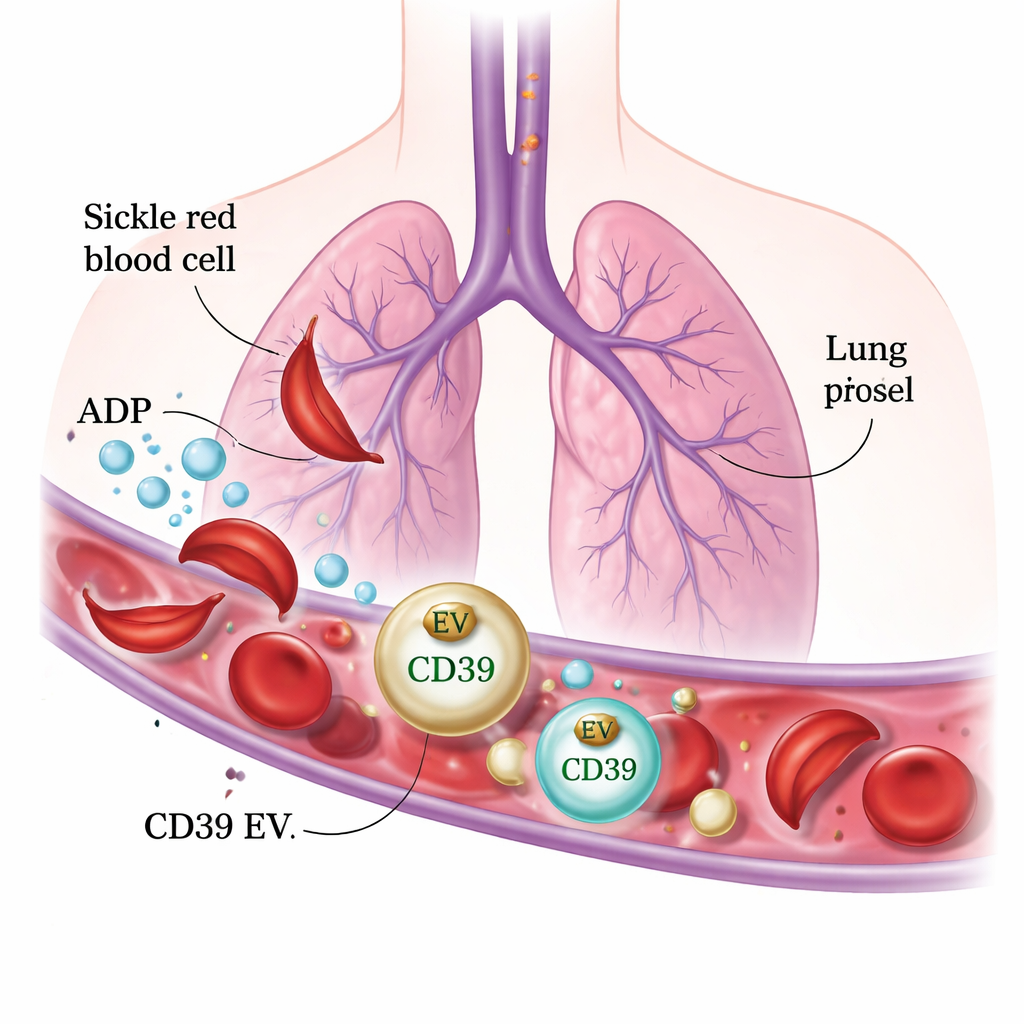
Tiny protective bubbles from the lung wall
Working with specially bred mice that carry human sickle hemoglobin, the researchers used live high-resolution imaging to watch what happens in the lungs when ADP is injected into the bloodstream. In normal mice, ADP quickly caused platelet-rich clots to form and temporarily block small lung vessels. In contrast, sickle cell mice were unexpectedly protected: their lung vessels stayed open and their platelets did not clump in response to ADP. When the team examined the blood, they found many more tiny membrane “bubbles,” called extracellular vesicles, in sickle mice than in controls. These vesicles carried large amounts of a surface enzyme called CD39, which breaks down ADP into a less active form. In lab tests, vesicles from sickle mice powerfully degraded ADP and prevented platelets from aggregating.
Switching protection off reveals CD39’s role
To prove that CD39 was responsible for this protection, the scientists blocked CD39’s activity in sickle mice using a chemical inhibitor. Once CD39 was shut down, ADP suddenly behaved as expected: it triggered strong platelet clumping and lung vessel blockages, along with a drop in circulating platelet counts, signs of active clot formation. Similarly, when CD39 was inhibited in blood samples from sickle mice, platelet responses to ADP were restored to normal strength. These experiments showed that CD39 carried on vesicles in the bloodstream acts as a powerful brake on clotting signals in sickle cell disease, specifically in the lungs.
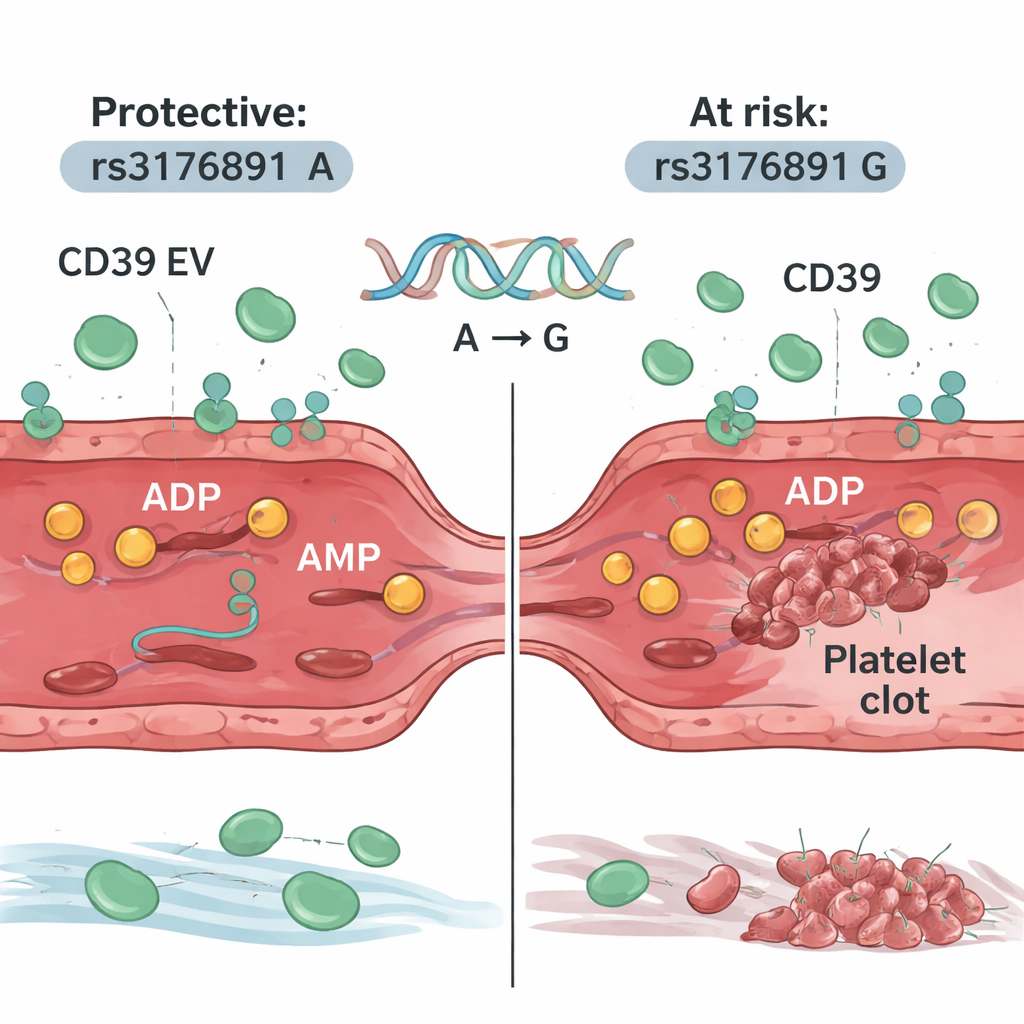
A genetic variant that weakens the shield
The researchers then turned to people living with sickle cell disease. They found that many patients also have abundant CD39-carrying vesicles in their blood, and their platelets respond poorly to ADP, consistent with a natural protective effect. However, genetic analysis of more than 400 patients revealed that those with a particular version of the CD39 gene—called rs3176891 G—were more likely to have a history of lung blood clots. In this genetic subgroup, patients’ blood contained fewer CD39-positive vesicles, lower CD39 activity, and platelets that reacted more strongly to ADP. Importantly, the same genetic variant did not increase clot risk in a large group of people of African ancestry without sickle cell disease, suggesting that the danger appears mainly when ongoing red blood cell destruction is present.
What this means for patients and future care
For people with sickle cell disease, these findings highlight that the body is not simply overrun by clotting signals—it also deploys its own countermeasures. In many patients, lung blood vessel cells release CD39-rich vesicles that soak up excess ADP and help keep platelets from clogging fragile lung arteries. But those who inherit the rs3176891 G version of the CD39 gene may have a weaker shield, leaving them more vulnerable to lung thrombosis. In the future, testing for this genetic variant and measuring CD39-bearing vesicles could help identify higher-risk patients and tailor treatments that target platelet signaling more precisely, turning this new understanding of a natural defense system into better protection against one of sickle cell disease’s most serious complications.
Citation: Brzoska, T., Kaminski, T.W., Katoch, O. et al. CD39 polymorphism enables lung thrombosis in sickle cell disease. Nat Commun 17, 1693 (2026). https://doi.org/10.1038/s41467-026-68396-2
Keywords: sickle cell disease, lung blood clots, platelets, extracellular vesicles, CD39 polymorphism