Clear Sky Science · en
Modeling tissue-specific Drosophila metabolism identifies high sugar diet-induced metabolic dysregulation in muscle at reaction and pathway levels
Why this fruit fly study matters to human health
Too much sugar in our diets can strain the body’s ability to manage fuel, eventually contributing to type 2 diabetes. This study uses fruit flies to uncover, in remarkable detail, how different tissues handle metabolism and how a high-sugar diet disrupts muscle function. Because flies share many metabolic genes and organ systems with humans, these insights help explain what may go wrong in our own muscles under long-term sugar overload.
Mapping metabolism tissue by tissue
Our bodies, like those of fruit flies, are made of tissues that play very different metabolic roles: muscle burns fuel, fat stores it, the gut processes food, and so on. Instead of measuring every enzyme directly, the researchers built large-scale “maps” of metabolism—called genome-scale metabolic models—for 32 distinct fly tissues. They combined an existing, curated network of chemical reactions with single-cell gene expression data, which indicate which metabolic genes are turned on in each tissue. This allowed them to compare how many reactions, metabolites, and genes are active in each tissue-specific network and to see which pathways are emphasized in muscle, fat body, gut, and various nerve cell types.
Distinct fuel jobs for different organs
The comparison revealed clear metabolic “personalities” across tissues. Fat body and oenocytes—fly counterparts of human fat and liver—contained the richest set of reactions, especially those that burn fats via beta-oxidation. Muscle, in contrast, did not have the largest network, but it did have the highest fraction of reactions that move substances in and out of cells, suggesting that muscle is a major hub for exchanging fuels and building blocks with the rest of the body. The scientists also checked whether the predicted pathways matched real metabolite patterns measured in different body regions. Using targeted metabolomics, they analyzed hundreds of small molecules from fly heads, thoraces, guts, and abdomens and then asked whether pathways enriched in the data overlapped with those predicted by the models. In muscle and fat body, the match was strong, giving confidence that the tissue-specific models captured real biology better than gene expression alone.
What a high-sugar diet does to muscle fuel flow
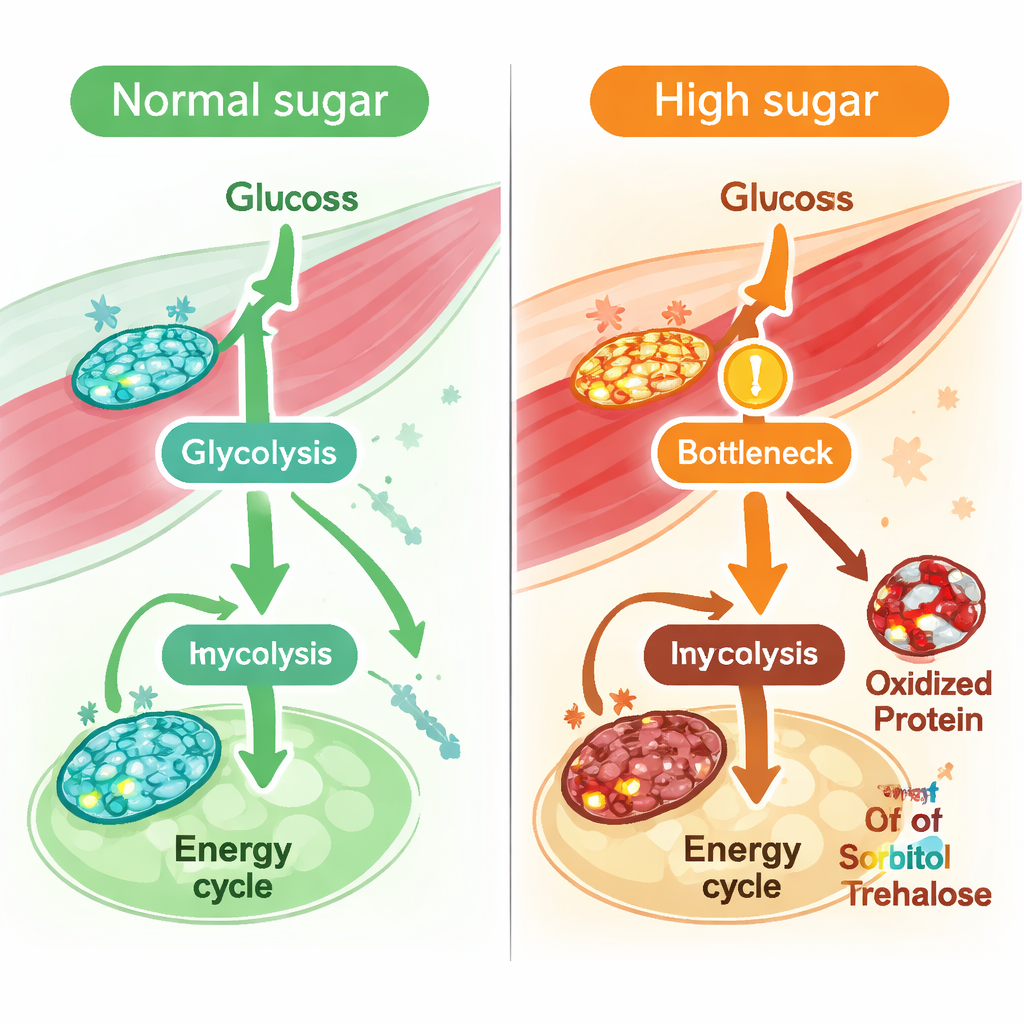
With a validated muscle model in hand, the team simulated what happens when flies are fed a long-term high-sugar diet, a well-established model of type 2 diabetes. They constrained the model using known features of diabetic muscle, such as reduced glucose uptake and sluggish activity in the central energy cycle in mitochondria. Computational “flux” analyses—calculations of how fast reactions can run—showed broad reductions in reactions that depend on the redox pair NAD and NADH, molecules that shuttle electrons and are essential for energy production. In particular, reactions in the central sugar-burning pathway, glycolysis, slowed down, including those catalyzed by the enzyme GAPDH. The model also predicted a roughly one-quarter drop in the muscle’s capacity to produce NADH overall, hinting at stressed redox balance. Direct measurements in fly thoracic muscle confirmed that the ratio of NAD to NADH indeed fell under a high-sugar diet.
Following labeled sugar and oxidized proteins
To see whether these predicted bottlenecks truly occurred in living animals, the researchers fed flies a high-sugar diet containing uniformly labeled carbon-13 glucose and then tracked where the labeled carbon went. They found that early glycolytic intermediates upstream of GAPDH accumulated, while downstream products and their labeled fractions dropped, signaling a real slowdown at and below this step. Similarly, contributions of glucose carbon to the central energy cycle were reduced. At the same time, redox proteomics—a method that detects oxidative changes on specific amino acids in proteins—showed increased oxidation of many glycolytic enzymes, including multiple sites on GAPDH. Across glycolysis, enzymes that carried more oxidative modifications tended to show larger drops in predicted flux, even though their overall protein levels stayed mostly unchanged. This suggests that chemical damage from oxidative stress, rather than loss of enzyme abundance, is a key driver of impaired sugar processing in muscle.
Hidden trouble in fructose and sucrose handling
Looking beyond single reactions, the team averaged fluxes across whole pathways to see which ones were most perturbed by high sugar. Glycolysis, the energy cycle, and oxidative phosphorylation all showed declines, but one of the strongest predicted decreases was in fructose metabolism. Metabolite profiling of muscle supported this idea: sorbitol and trehalose/sucrose levels rose, and a key enzyme that converts trehalose into glucose, Trehalase, showed both reduced predicted activity and increased oxidation at a sensitive methionine residue. Together, these findings point to a broader disturbance in how muscle handles dietary sugars—especially fructose-like and sucrose-derived fuels—under chronic sugar overload.
What this means for understanding diabetes
In plain terms, this work shows that too much sugar does not just overload the bloodstream; it also quietly reshapes how muscle cells route and burn fuel. By building detailed, tissue-specific metabolic maps for the fruit fly and cross-checking them with metabolomics, isotope tracing, and redox proteomics, the researchers reveal that high sugar drives redox stress, oxidizes key glycolytic enzymes such as GAPDH, slows sugar breakdown, and disrupts fructose-related pathways. These insights, obtained in a highly tractable model organism, offer a powerful framework for pinpointing which reactions and pathways might be most important to protect or restore in human muscle to prevent or treat type 2 diabetes.
Citation: Moon, S.J., Hu, Y., Dzieciatkowska, M. et al. Modeling tissue-specific Drosophila metabolism identifies high sugar diet-induced metabolic dysregulation in muscle at reaction and pathway levels. Nat Commun 17, 1692 (2026). https://doi.org/10.1038/s41467-026-68395-3
Keywords: Drosophila metabolism, tissue-specific metabolic models, high-sugar diet, muscle glycolysis, redox regulation