Clear Sky Science · en
Zinc-based metal halide electrolytes for all-solid-state zinc-metal batteries
Solid Batteries for a Safer, Greener Future
As our homes, cars, and entire power grids lean more on renewable energy, we need batteries that are not only powerful and cheap, but also safe and long‑lasting. Today’s workhorse lithium‑ion batteries come with cost and safety concerns, while common zinc batteries often rely on water‑based liquids that limit performance. This study explores a new class of solid materials that could let zinc‑metal batteries store energy safely and efficiently, potentially reshaping how we power everything from portable gadgets to large‑scale storage.
Why Zinc Batteries Need an Upgrade
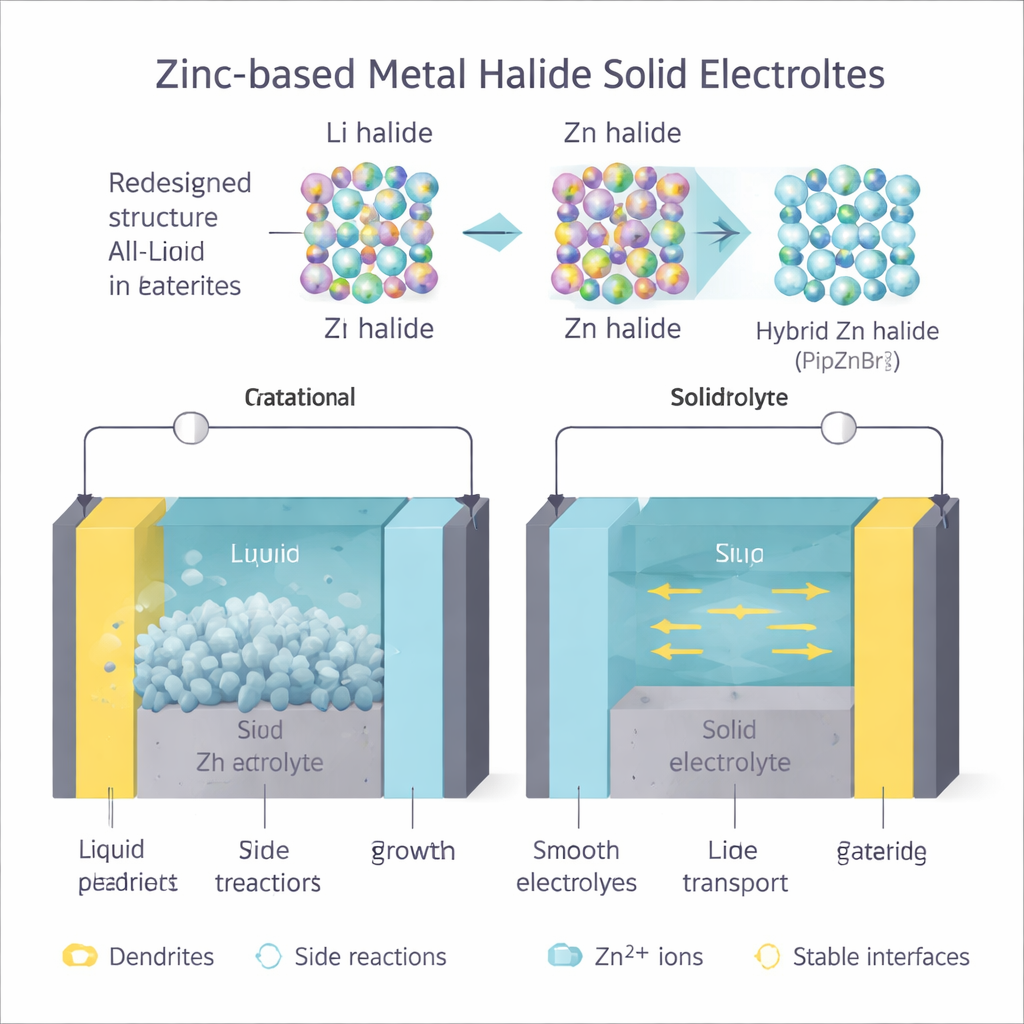
Zinc‑metal batteries are attractive because zinc is abundant, inexpensive, and far safer than lithium in many settings. However, most zinc batteries today use water‑based (aqueous) liquids as electrolytes—the medium that shuttles charged particles between the two electrodes. These liquids bring several problems: they tend to decompose at high voltages, can dissolve parts of the positive electrode, and encourage unwanted reactions at the zinc surface, including gas formation and needle‑like “dendrites” that can short‑circuit the battery. Solid electrolytes, in principle, can avoid these issues by acting like an ion‑conducting ceramic or plastic, while keeping electrons and troublesome side reactions at bay. Yet designing solids that let relatively heavy, doubly charged zinc ions move quickly has proven challenging.
From Lithium Clues to Zinc Solutions
The researchers began by asking why many metal halide crystals that work so well as solid electrolytes for lithium fail for zinc. On the surface, lithium and zinc ions can occupy very similar spots in a crystal, and both form neat tetrahedral or octahedral cages with surrounding halogen atoms such as chlorine or bromine. But a closer look at their electron orbitals shows a key difference: lithium forms mostly ionic, easily broken bonds, while zinc forms stronger, more covalent bonds with halogens. Computer calculations confirmed that in typical zinc halide crystals, the energy barrier for a zinc ion to hop from one site to another is much higher than for lithium, making zinc transport sluggish. The team concluded that simply copying lithium‑based designs would not work; the zinc environment itself had to be re‑engineered.
Designing a Softer Path for Zinc Ions
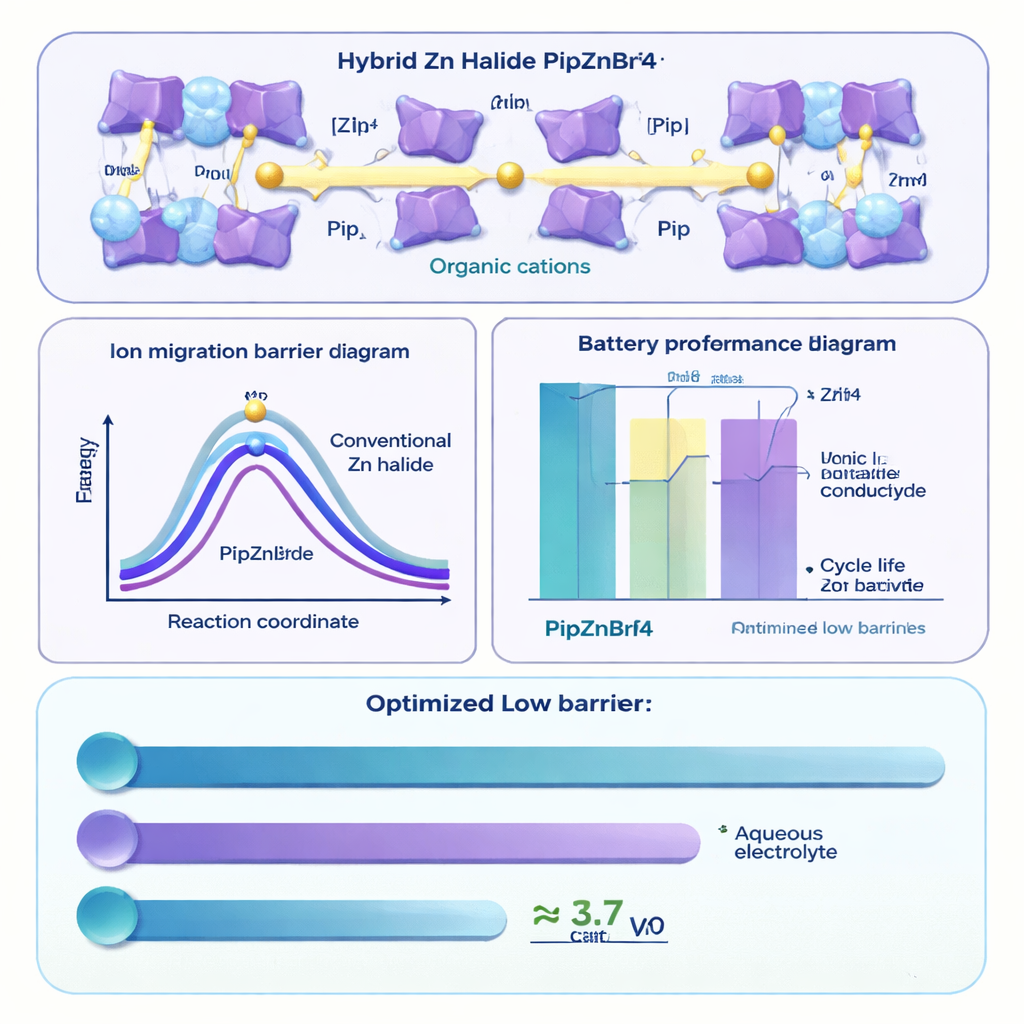
To open up easier pathways, the team proposed replacing some of the rigid, spherical inorganic cations in zinc halide structures with larger, softer organic molecules. In their design, an organic “pillar” (derived from the molecule piperazine) carries positive charge and helps hold zinc‑halide units in place, but leaves more open space and flexibility in the crystal. This led to two hybrid materials, called PipZnBr4 and PipZnCl4, where zinc and halide ions are surrounded by organic groups in a more loosely packed arrangement. Advanced quantum‑mechanical calculations showed that both materials are excellent electrical insulators (they block electrons) but allow zinc ions to move along channels with relatively low energy barriers—comparable to those in good lithium solid electrolytes. Among the two, PipZnBr4 emerged as the most promising candidate, combining stable bonding with favorable zinc‑ion motion.
Putting the New Solid Electrolyte to the Test
The researchers then synthesized PipZnBr4 using a straightforward solution process and pressed the resulting powder into solid pellets. Measurements showed that at room temperature the material conducts ions about a thousand times better than many early solid electrolytes, and it maintains this performance over a practical temperature range. It also remains stable over a wide span of voltages, meaning it can support higher‑energy battery designs without breaking down. When paired with a zinc metal anode, PipZnBr4 forms a tight, uniform interface that keeps resistance low. Imaging methods, including electron microscopes and 3D X‑ray scans, revealed that zinc deposits grow as smooth, dense spheres rather than sharp dendrites. Over repeated charge–discharge cycles, the solid electrolyte helps generate a robust protective layer on the zinc that further guides even plating and stripping of zinc metal.
Long‑Lasting Performance in a Full Battery
To see how this translates into real‑world behavior, the team built full solid‑state zinc‑metal batteries using PipZnBr4 as the electrolyte and iodine as the positive electrode material. These cells delivered high capacity and retained 234.5 milliamp‑hours per gram of iodine even after 200 cycles at moderate current, with only 0.056% capacity loss per cycle. Additional tests with symmetric zinc cells and zinc‑titanium cells showed highly reversible zinc plating and stripping with low energy losses and minimal side reactions. The authors also carefully ruled out the possibility that bromide or chloride ions, rather than zinc ions, dominate the charge transport, confirming that zinc is indeed doing the main work inside the solid.
What This Means for Everyday Technology
For non‑experts, the main message is that this work introduces a smart way to redesign the “traffic lanes” that ions use inside a battery. By weaving zinc and halide ions into a flexible, organic‑inorganic crystal, the researchers created a solid material that safely moves zinc ions while blocking electrons and harmful reactions. This solid electrolyte supports smooth, dendrite‑free zinc growth and enables stable, long‑lasting all‑solid‑state zinc‑metal batteries. While more steps are needed before such materials appear in commercial products, the study lays a clear foundation for safer, more sustainable batteries that could complement or, in some uses, even replace today’s lithium‑ion technology.
Citation: Hu, S., Chang, C., Lin, YP. et al. Zinc-based metal halide electrolytes for all-solid-state zinc-metal batteries. Nat Commun 17, 1691 (2026). https://doi.org/10.1038/s41467-026-68394-4
Keywords: solid-state zinc batteries, zinc metal halide electrolytes, PipZnBr4, dendrite-free zinc anodes, energy storage materials