Clear Sky Science · en
Regulating zinc nucleation and growth with low-surface-tension electrolytes for practical aqueous zinc metal batteries
Why this new battery idea matters
As we add more electric cars to the roads and more renewable power to the grid, we need batteries that are not only powerful but also safe, affordable, and made from abundant elements. This research explores a promising alternative to today’s lithium‑ion batteries: rechargeable zinc metal batteries that use water‑based liquids. The authors show that by carefully tuning how “stretchy” or “tight” the surface of the battery liquid is—its surface tension—they can dramatically extend the life and safety of zinc batteries, even under demanding, real‑world conditions.
From spiky zinc to smooth surfaces
In conventional zinc batteries, the metal anode tends to grow needle‑like structures called dendrites when the battery is charged. These spiky growths form because zinc ions and electric fields crowd around tiny bumps on the surface, causing them to grow faster than flatter regions. Over time, dendrites can pierce the separator inside the battery, leading to short circuits, gas formation, and a rapid loss of usable zinc. This instability has held back zinc batteries from large‑scale use, despite zinc being cheap, abundant, and safer than lithium in many respects.

Using the liquid’s “feel” to guide metal growth
The team focused on a property of the electrolyte—the liquid that carries ions between electrodes—that is usually overlooked: surface tension, the same effect that lets water form beads on a surface. Using classical physics of how new solid particles form and grow, they showed mathematically that the surface tension of the liquid strongly controls how easily zinc first appears as tiny “seed” spots and how those seeds grow. High surface tension raises the energy cost to form new zinc seeds and favors fewer, larger particles that quickly develop into protrusions. Lowering surface tension does the opposite: it makes it easier for many small seeds to form and encourages a fine, tightly packed zinc layer rather than big spikes.
A simple tweak to the liquid recipe
To put this idea into practice, the researchers started from a standard water‑based zinc electrolyte and added small amounts of low‑polarity organic liquids—especially a molecule called triethyl phosphate (TEP). These additives weaken the strong water‑water hydrogen bonds near the zinc surface, which lowers the surface tension without drastically changing how well the liquid conducts ions. With only 5 percent TEP by volume, the surface tension dropped to about half that of the original liquid, while most of the conductivity was preserved. Simulations and X‑ray measurements confirmed that TEP mostly sits near the interface and disrupts the water network there, rather than binding directly to zinc ions, so it can keep working over long times without being consumed.
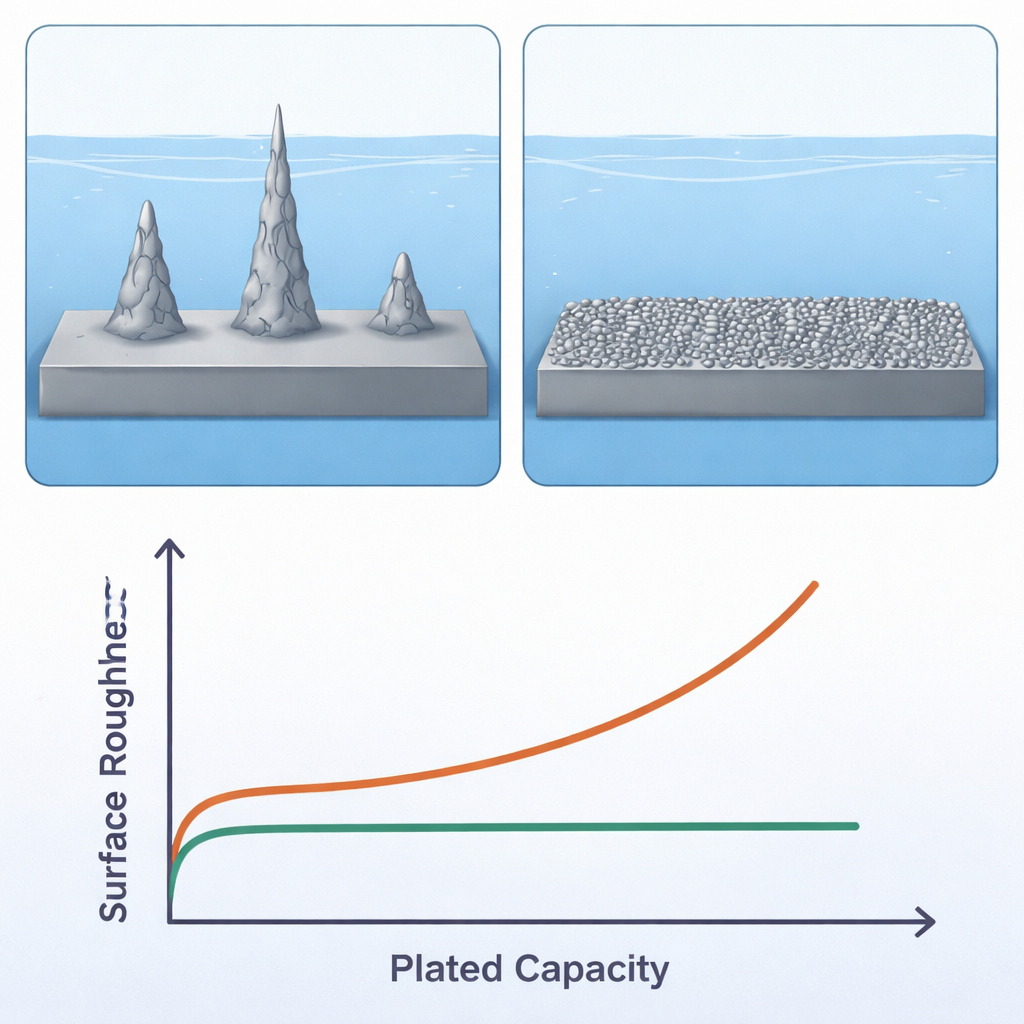
Smoother zinc, fewer side reactions, longer life
Microscope images of zinc grown in the modified liquids reveal a striking transformation. In the traditional, higher‑tension electrolyte, zinc deposits as sparse, rough islands that evolve into tall, porous dendrites and give the surface a very uneven profile. In the low‑tension electrolyte with TEP, zinc forms many small, dense nuclei that grow into a smooth, compact layer, even when large amounts of zinc are plated at high current. This fine‑grained coating also favors a particular crystal face of zinc that is more resistant to corrosion and gas evolution. Chemical probes show that the protective surface film on zinc becomes richer in stable zinc carbonate and poorer in corrosive hydroxides, while direct gas measurements reveal a sharp drop in hydrogen generation, a sign that damaging side reactions are strongly suppressed.
Toward practical, large zinc batteries
Because the zinc surface stays smooth and protected, cells using the low‑tension electrolyte can be driven very hard without failing. Laboratory cells reach an average efficiency of about 99.7 percent over nearly a year of continuous cycling and survive thousands of charge‑discharge cycles at currents and capacities relevant to commercial systems. Even under harsh conditions that normally destroy zinc anodes quickly, the modified cells last tens to hundreds of times longer than those with the conventional liquid. Full batteries paired with a vanadium‑based positive electrode deliver high capacity at fast charge rates, operate with thin zinc foils and limited electrolyte, and scale up to a 1.27‑ampere‑hour pouch cell while maintaining high efficiency.
What this means for future batteries
For non‑specialists, the core message is that the “feel” of the battery liquid at its surface—how tightly its molecules pull together—can be a powerful lever for controlling how metal grows and ages inside a battery. By modestly lowering surface tension, the authors turn messy, spiky zinc growth into a smooth, durable coating, cutting wasteful reactions and greatly extending battery life. Because the approach relies on small amounts of relatively simple additives and keeps the system water‑based, it offers a potentially low‑cost and safe path toward practical zinc batteries for grid storage, backup power, and perhaps some electric vehicles. The same design principle may also inspire better metal‑based batteries beyond zinc, including future lithium and sodium systems.
Citation: Wang, H., Li, G., Fu, J. et al. Regulating zinc nucleation and growth with low-surface-tension electrolytes for practical aqueous zinc metal batteries. Nat Commun 17, 1690 (2026). https://doi.org/10.1038/s41467-026-68393-5
Keywords: zinc metal batteries, surface tension, electrolyte design, dendrite suppression, aqueous energy storage