Clear Sky Science · en
The simplest iminophosphane HPNH and its photoisomerization to aminophosphinidene H2NP
Why tiny phosphorus molecules matter in space and on Earth
Phosphorus is essential for life, yet we still know surprisingly little about how its simplest molecules behave in space and under extreme conditions. This paper explores two of the smallest phosphorus–nitrogen species, called HPNH and H2NP, which are thought to be part of the chemical network that turns simple interstellar molecules into the building blocks of life. By finally making and characterizing these elusive compounds in the laboratory, the authors reveal how light can shuffle atoms within them and how these reactions could play out in the cold darkness of space.
From starlight to strange molecules
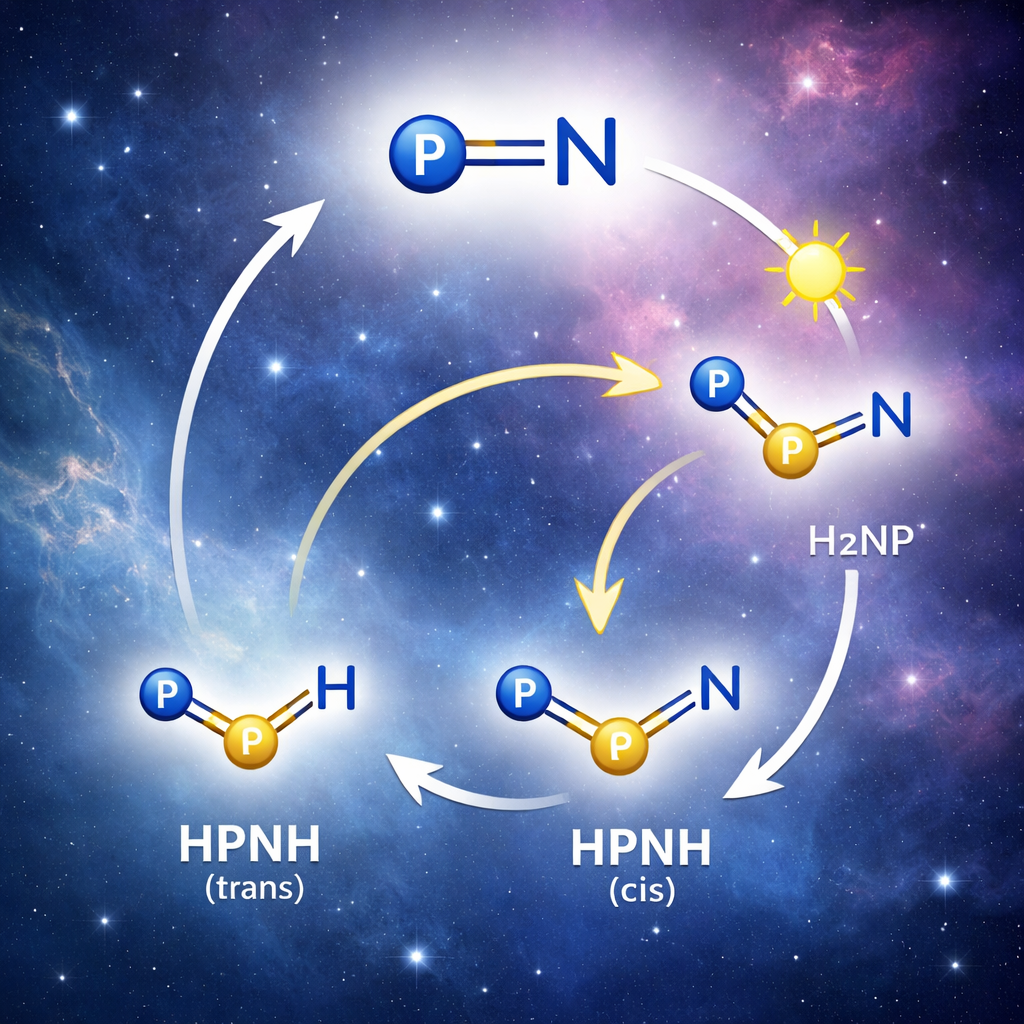
A key starting point for this work is a very small molecule, phosphorus mononitride (PN), which was the first phosphorus-containing compound detected in interstellar clouds. PN is highly reactive and can link up into chains and rings, but it can also be transformed into even simpler hydrogen-containing relatives. Chemists have long suspected that three closely related species—HPNH, H2NP, and H2PN—sit on the same energy landscape and might form when PN picks up hydrogen atoms in space. These species contain short, multiple bonds between phosphorus and nitrogen and are tiny prototypes for a much wider world of phosphorus chemistry in stars, planets, and laboratories.
Making a long‑sought molecule at 950 degrees
Despite decades of theoretical predictions, no one had convincingly made the parent molecule HPNH in the lab. The authors achieved this by heating a larger phosphorus–nitrogen compound, di‑tert‑butylphosphanamine, to about 950 K in a high-vacuum “flash pyrolysis” setup. The heat snaps off bulky carbon groups, leaving behind bare HPNH in the gas phase. They then immediately trapped the products in an extremely cold nitrogen matrix at just 10 K, where the molecules are immobilized and can be probed by infrared and ultraviolet–visible spectroscopy without falling apart or reacting further.
Bending, stretching, and flipping under light
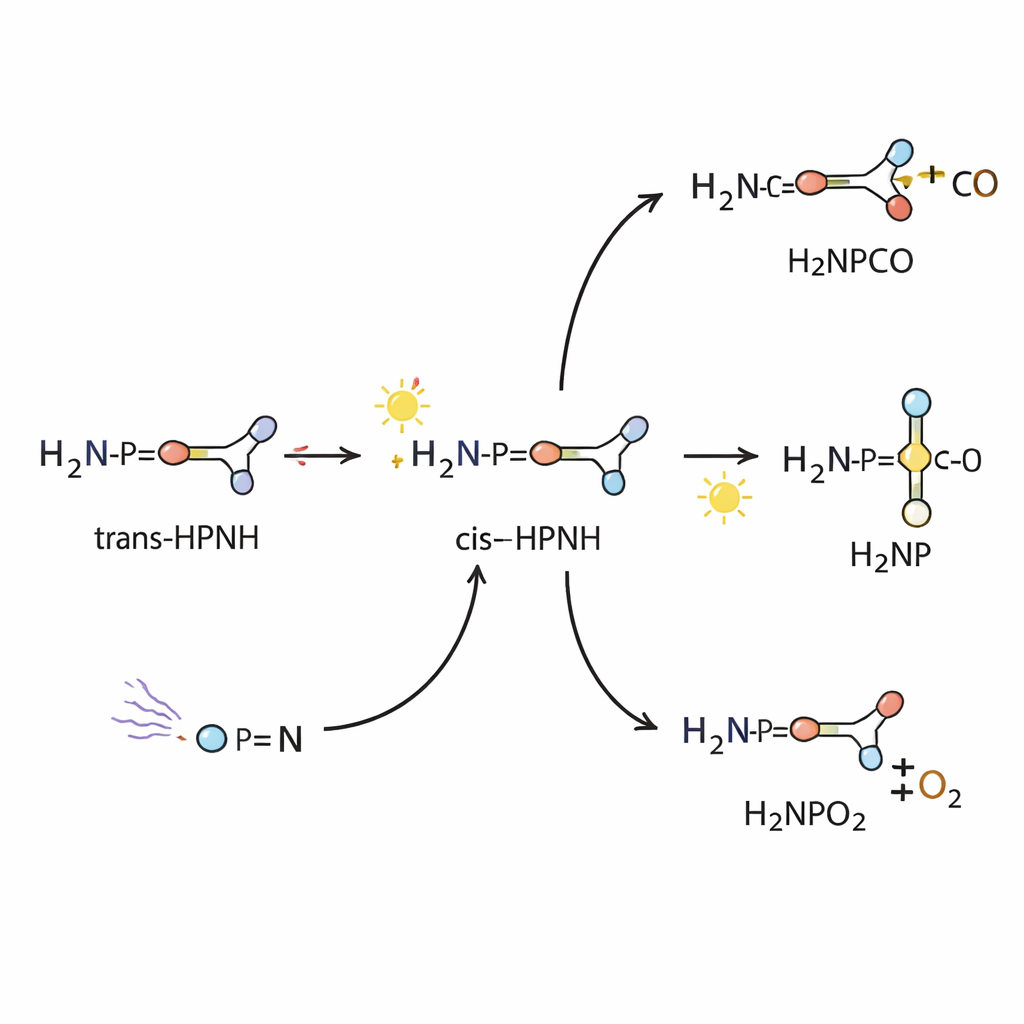
Once HPNH was in this icy cage, the authors used carefully chosen wavelengths of light to watch how it moves and changes. HPNH can adopt two shapes, called trans and cis, that differ by how the two hydrogen atoms sit around the phosphorus–nitrogen unit. Light of around 410 nm converts the trans form to the cis form, and light at 365 nm reverses the process. These shape changes leave distinct fingerprints in the infrared spectrum, which the team matched with high-level quantum-chemical calculations. This not only confirms the presence of both forms but also nails down how their bonds vibrate and how strongly atoms are connected.
Light-driven reshuffling into a new reactive species
More energetic light triggers a deeper transformation: a hydrogen atom within HPNH migrates from phosphorus to nitrogen, turning HPNH into a different isomer, H2NP. This subtle rearrangement changes which atom carries which hydrogen but leaves the overall formula the same. The new species shows its own distinct set of infrared bands and ultraviolet absorptions. By comparing these with detailed theoretical spectra, the authors conclude that H2NP exists in a “singlet” ground state, meaning its electrons are paired rather than unpaired. In this state, H2NP behaves like a very reactive phosphorus center with a strong double bond to nitrogen, ready to attack other small molecules.
Testing reactivity with simple gases
To see how reactive H2NP really is, the researchers let it interact with two common small molecules: carbon monoxide (CO) and oxygen (O2). When HPNH is photolyzed in solid CO at 10 K, H2NP forms and is immediately trapped by CO to give a new compound, H2NPCO. In an oxygen-doped matrix, light again generates H2NP, which reacts with O2 to form H2NPO2, a phosphorus analogue of nitrous acid derivatives. These reactions show that once H2NP appears, it can readily build more complex phosphorus–carbon and phosphorus–oxygen structures, even at temperatures only a few degrees above absolute zero.
What this means for space chemistry and synthesis
By finally generating and characterizing HPNH and its photoisomer H2NP, this study fills in missing pieces of the puzzle surrounding phosphorus–nitrogen chemistry. For astrochemistry, it provides concrete data on how light can shuffle hydrogen atoms around PN-based species and how the resulting reactive intermediates might combine with CO and O2 in cold molecular clouds, helping to form more complex molecules. For synthetic chemistry on Earth, H2NP emerges as a fundamentally new, highly reactive building block that could be harnessed to construct novel phosphorus-containing materials. In both realms, these tiny molecules act as crucial waypoints on the journey from simple atoms to the rich chemistry that underpins life.
Citation: Jiang, J., Guo, Y., Huang, L. et al. The simplest iminophosphane HPNH and its photoisomerization to aminophosphinidene H2NP. Nat Commun 17, 1687 (2026). https://doi.org/10.1038/s41467-026-68391-7
Keywords: astrochemistry, phosphorus nitrogen chemistry, photochemistry, interstellar molecules, reactive intermediates