Clear Sky Science · en
Solvent-mediated partial ionicity enhances mechanical nanosizing effect of Mg-based hydrogen storage alloys
Why shrinking metals could transform clean energy
Hydrogen is often hailed as a clean fuel of the future, but storing it safely and efficiently remains a stubborn challenge. This research shows how a common light metal, magnesium, can be reshaped into ultra-small particles that soak up and release hydrogen remarkably fast—and how a simple liquid and a thin plastic-like coating make this possible in a way that could be scaled up for real-world energy storage.
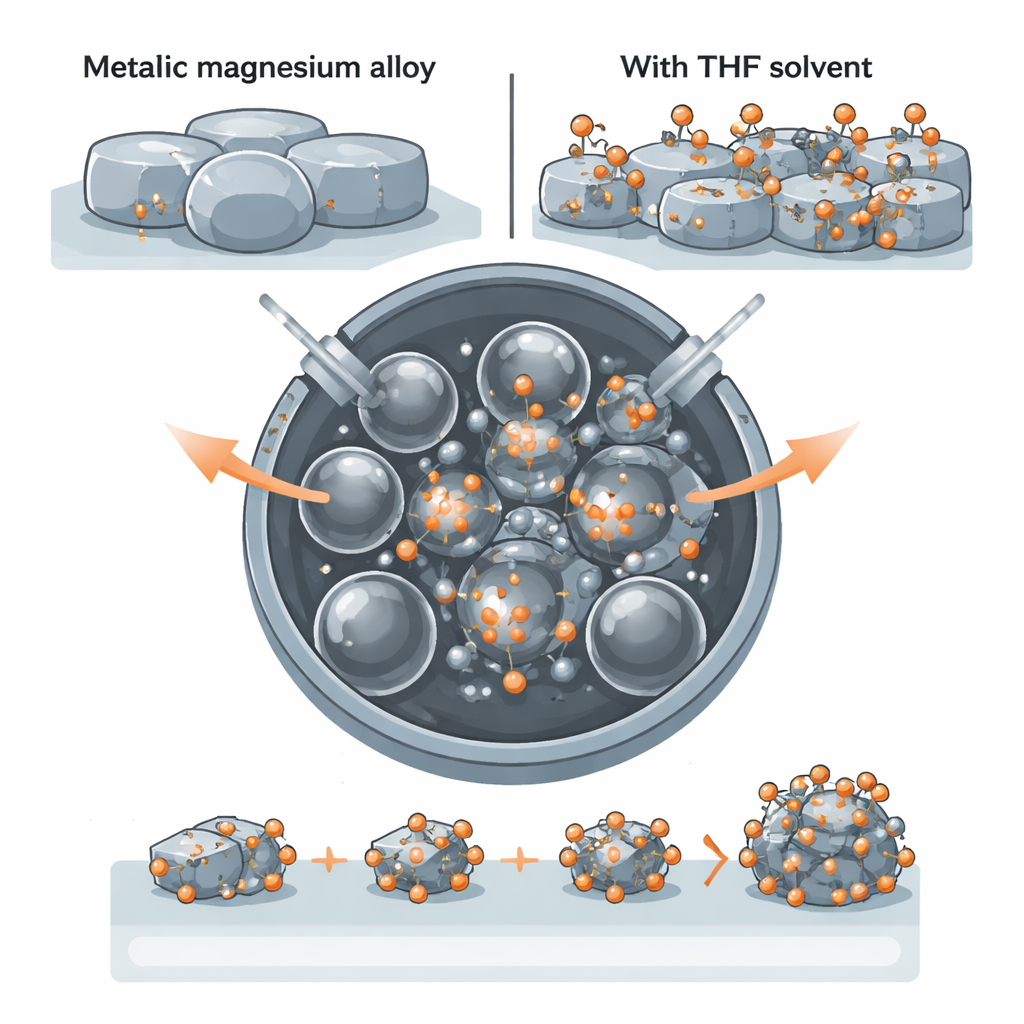
Turning a soft metal into tiny hard particles
Magnesium can theoretically store a great deal of hydrogen by weight, but in bulk form it reacts slowly and only at high temperatures. One promising solution is to chop magnesium-based alloys into nanoparticles, which shorten the paths that hydrogen atoms must travel and create many more reactive surface sites. However, magnesium is mechanically soft and ductile: instead of shattering under impact, it tends to smear and weld together. This makes conventional ball milling—a low-cost way to grind materials—surprisingly ineffective for producing magnesium nanoparticles.
A smart solvent that changes the metal’s surface
The authors tackled this problem using a magnesium–nickel–yttrium alloy and a common organic solvent called THF (tetrahydrofuran). When they performed ball milling without solvent, the alloy stubbornly stayed as large particles about 45 micrometers across. Adding just 1 milliliter of THF transformed the outcome: the average particle size dropped to about 0.5 micrometers—an 88-fold reduction—and the size distribution became much more uniform. Microscopy and surface analysis confirmed that the alloy remained largely unoxidized and that the nickel and yttrium components were well distributed, preparing the material to act as both a hydrogen sponge (magnesium) and a built‑in catalyst (nickel and yttrium hydrides).
How partial charges make a toughened shell
To understand why THF is so effective, the team combined experiments with computer simulations. Calculations showed that THF molecules tend to sit atop magnesium atoms at the surface, pulling a small amount of electronic charge away from those atoms and pushing it toward their neighbors. This creates tiny positive–negative pairs—so‑called dipoles—between adjacent magnesium atoms, a state the authors call partial ionicity. This subtle charge rearrangement stiffens the surface: hardness tests revealed that THF-treated magnesium became about 22% harder than untreated metal. In practical terms, the alloy behaves less like a soft metal and a bit more like a brittle ionic solid, so the violent impacts inside the ball mill now generate cracks and fractures instead of plastic smearing, greatly boosting the nanosizing effect.
Protecting nanoparticles without suffocating them
Nanoparticles bring new problems as well as benefits. Their increased surface area makes them far more prone to corrosion by moisture, which can quickly form magnesium hydroxide and degrade performance. To counter this, the researchers coated the nanosized alloy with very small amounts of PMMA, a transparent polymer widely used in everyday plastics. Even a 0.1% PMMA layer sharply reduced unwanted hydrogen generation from reaction with water and suppressed the formation of corrosion products in air, while still allowing hydrogen to move in and out of the particles. Thicker coatings improved protection further but began to slow hydrogen release, showing that a careful balance between shielding and accessibility is essential.
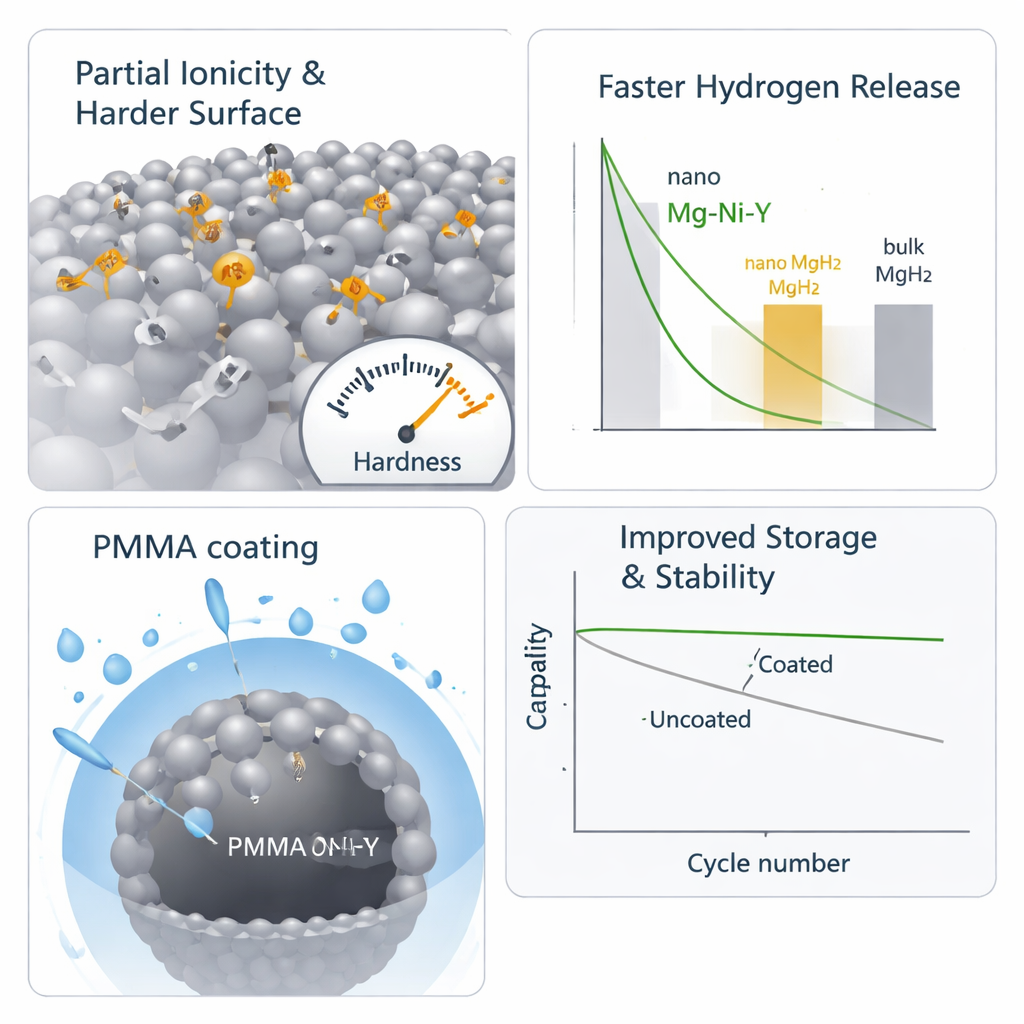
Fast hydrogen cycling and long-term durability
When tested for hydrogen storage, the THF-milled nanoparticles showed strikingly rapid behavior. They released more than 95% of their theoretical hydrogen capacity in just three minutes at 300 °C and maintained strong performance even at 240 °C, far outperforming typical magnesium hydride materials. The energy barrier for releasing hydrogen was less than half that of conventional bulk magnesium hydride, reflecting both the nanoscale structure and the catalytic roles of nickel and yttrium hydrides. With the optimized 0.1% PMMA coating, these nanoparticles could be cycled at least 500 times with almost no loss of storage capacity or speed, substantially better than many previously reported magnesium-based systems.
What this means for future hydrogen storage
In simple terms, this study shows that carefully chosen solvents can temporarily "rewire" the surface of a soft metal, making it easier to grind into tiny, highly active particles, and that a thin protective skin can keep those particles working reliably over many cycles. By providing a relatively cheap, scalable route to robust magnesium-based hydrogen storage materials, the work points toward practical solid-state hydrogen tanks that operate faster, at lower temperatures, and with greater durability—important steps on the path to a hydrogen-powered energy system.
Citation: Sun, T., Tang, Z., Liu, J. et al. Solvent-mediated partial ionicity enhances mechanical nanosizing effect of Mg-based hydrogen storage alloys. Nat Commun 17, 1688 (2026). https://doi.org/10.1038/s41467-026-68390-8
Keywords: hydrogen storage, magnesium alloys, nanoparticles, solvent-assisted ball milling, energy materials