Clear Sky Science · en
OCT4 enhances the firing efficiency of late DNA replication origins in mouse embryonic stem cells
Why this matters for our cells
Every time a cell divides, it must copy its entire DNA accurately and on schedule. If this copying process goes wrong, it can lead to developmental problems or disease, including cancer. This study looks at how a key stem-cell protein, OCT4, helps organize when and where DNA copying starts in mouse embryonic stem cells—offering a window into how early embryos keep their genomes stable while dividing at breakneck speed.
Mapping when DNA gets copied
The authors focus on “replication timing”—the order in which different parts of the genome are duplicated during the DNA-copying phase of the cell cycle, called S phase. The genome is divided into large neighborhoods, or timing domains, that are copied early, in the middle, or late in S phase. Using high-throughput sequencing methods that label newly made DNA, the team charted the replication timing landscape of mouse embryonic stem cells and compared it with that of more mature cell types, such as fibroblasts and mesenchymal stem cells. They identified thousands of “initiation zones,” stretches of DNA where replication actually begins, and classified them as early, mid, or late based on which timing domain they fall into.
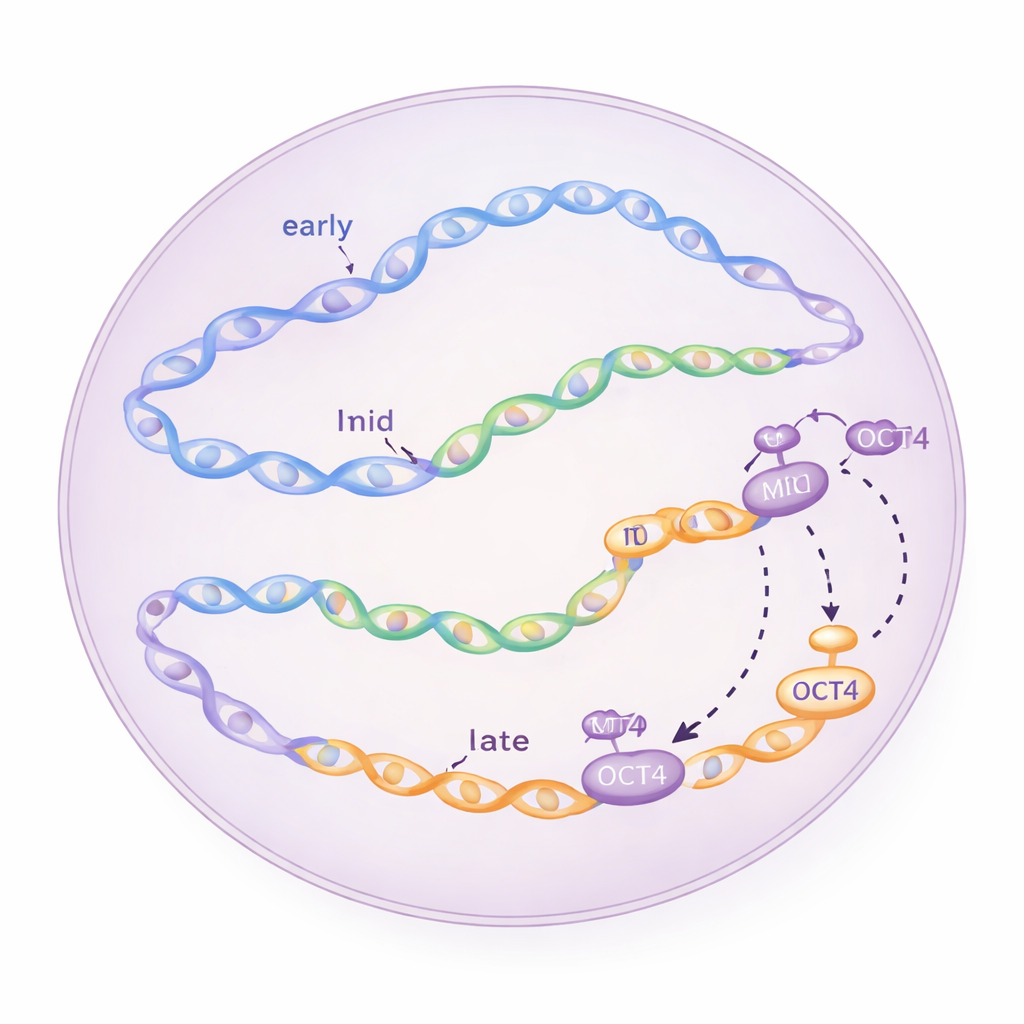
Late starters that jump the line
Conventional wisdom says that regions scheduled for late replication mostly wait until the end of S phase to start copying. Surprisingly, in embryonic stem cells the researchers saw some initiation zones located in late domains begin firing very soon after S phase starts—within just one to two hours. By synchronizing cells at different cell-cycle stages and repeatedly taking snapshots of newly synthesized DNA, they confirmed that these “late” zones really do light up early and that their activity depends on standard cell-cycle regulators like the CDC7 and CDK1 kinases, as well as the ATR checkpoint pathway, which normally helps prevent overloading the replication machinery.
Open DNA neighborhoods and the role of OCT4
To understand what makes these unusual early-firing late regions special, the team examined their local environment. They overlaid replication maps with data on RNA production and chromatin marks—chemical tags and structural features that indicate whether DNA is in an open, accessible state or packed away. Early initiation zones tended to sit near active genes and open chromatin, while late zones were more often in repressed, tightly packed regions. Yet the particular late zones that fired early in embryonic stem cells showed signatures of openness: they coincided with accessible chromatin, enhancer-like elements, and binding sites for pluripotency factors such as OCT4, SOX2, NANOG, and KLF4. This suggested that the same proteins that keep stem cells flexible in their fate might also be priming specific late regions of the genome for earlier replication.
Turning OCT4 off shifts the schedule
The authors then put this idea to the test using a special stem-cell line in which OCT4 levels can be rapidly reduced by adding the drug doxycycline. When OCT4 was depleted for just part of a single cell cycle, the overall progression into S phase stayed largely the same, but the firing of many mid and late initiation zones was weakened or delayed. Genome-wide analyses showed that, in regions where OCT4 normally binds, both chromatin accessibility and replication initiation signals dropped in parallel when OCT4 was turned off. Statistical modeling confirmed that this effect was strongest in late-replicating zones: the more an initiation zone depended on OCT4 for open chromatin, the more its replication efficiency suffered in OCT4’s absence.
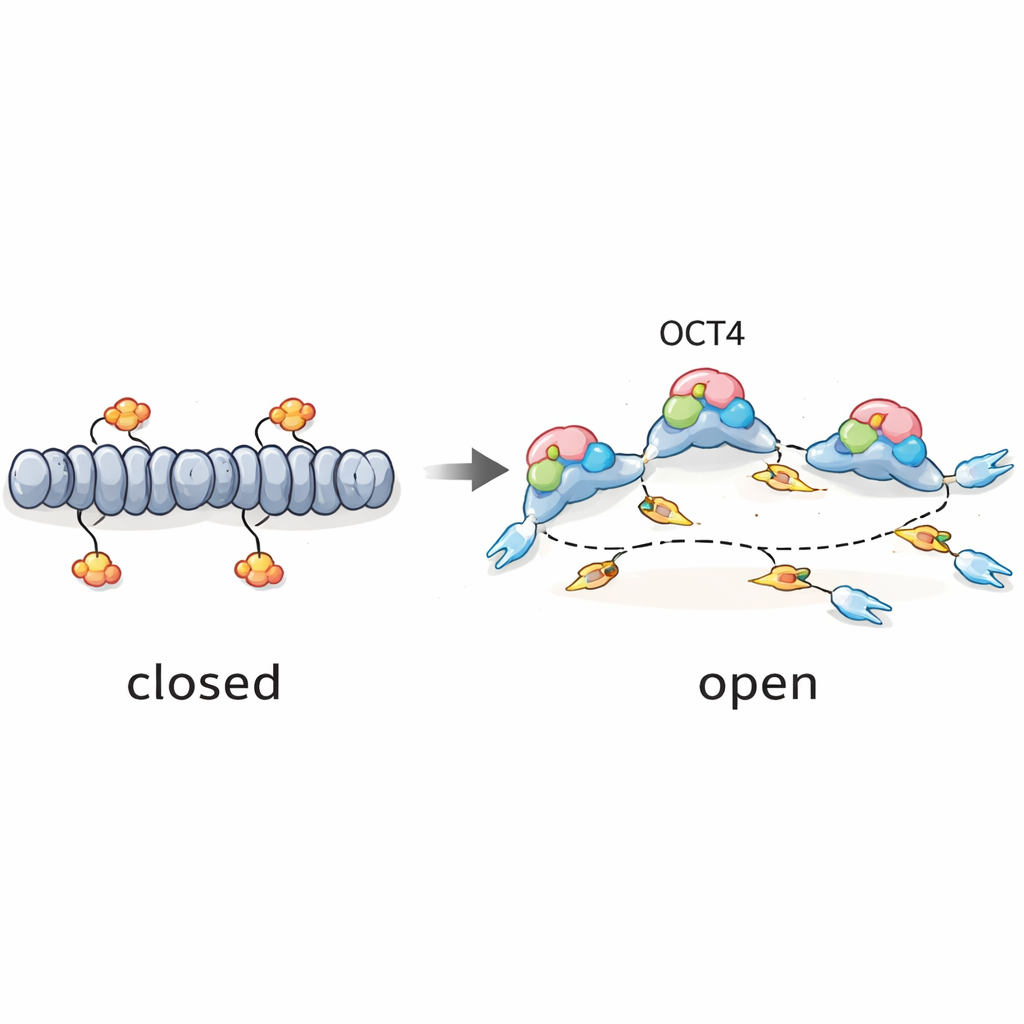
A pioneer factor that also times DNA copying
Taken together, the results support a simple idea: OCT4 acts as a “pioneer” factor not just for gene activity, but also for DNA replication. By prying open specific late regions of the genome in embryonic stem cells, OCT4 creates efficient starting points for DNA copying that can fire earlier and more reliably than they would in differentiated cells. At the same time, global regulators such as ATR, CDC7, and CDK1 tune how many of these sites are allowed to fire, preventing the replication system from being overwhelmed. For a lay reader, the key message is that the same proteins that keep stem cells versatile also help choreograph when different parts of their DNA are copied, adding another layer to our understanding of how rapidly dividing embryonic cells maintain order in their genomes.
Citation: Rodriguez-Carballo, E., Dionellis, V.S., Ntallis, S.G. et al. OCT4 enhances the firing efficiency of late DNA replication origins in mouse embryonic stem cells. Nat Commun 17, 1686 (2026). https://doi.org/10.1038/s41467-026-68389-1
Keywords: DNA replication timing, embryonic stem cells, OCT4, chromatin accessibility, origin firing