Clear Sky Science · en
OTX2 inhibits human pluripotent stem cell reprogramming toward 8-cell-like and morula-like states
Unlocking a Hidden Early Power in Human Cells
Every human begins as a single cell that quickly divides into a tiny ball of early cells, each able to form every tissue in the body and the support structures like placenta. Scientists are eager to recreate this rare, early “do‑anything” state in the lab because it could transform regenerative medicine and deepen our understanding of infertility and early pregnancy loss. This study reveals a key genetic brake, a gene called OTX2, that keeps human stem cells from rolling back to this powerful 8‑cell‑like state—and shows what happens when that brake is released. 
The Short Window When Cells Can Do Almost Anything
In human embryos, a dramatic handover happens at the 8‑cell stage. Control shifts from molecules stored in the egg to the embryo’s own genome, a process called zygotic genome activation. At this moment, cells are closest to full “totipotency”: each one can, in principle, form not only the fetus but also the supporting tissues such as the placenta. Because human embryos are scarce and ethically sensitive to study, researchers have been building lab models called 8‑cell‑like cells, or 8CLCs, by reprogramming ordinary human pluripotent stem cells. These stem cells are already versatile, but they normally can only make body tissues, not both body and extraembryonic tissues. The central question of this paper is: what stops pluripotent stem cells from re‑entering this earlier, more powerful state?
OTX2 Emerges as a Genetic Gatekeeper
The team began by combing through existing datasets from human embryos to spot transcription factors—genes that control other genes—that change activity around the 8‑cell stage. One candidate stood out: OTX2, long known for its roles in brain development and in pushing stem cells toward a more mature, “primed” state. They found that OTX2 is abundant in the earliest stages, including the zygote and 2‑ and 4‑cell embryos, but its levels drop sharply after the 8‑cell stage. In lab‑grown stem cell models that are gradually driven toward an 8‑cell‑like identity, OTX2 shows the opposite pattern of a key 8‑cell marker called TPRX1: as TPRX1 rises, OTX2 falls. This inverse relationship hinted that OTX2 might be acting as a barrier to reprogramming back to the 8‑cell‑like state.
Removing the Brake Recreates Early Embryo Features
To test this idea, the researchers engineered human pluripotent stem cells with a fluorescent reporter that lights up when TPRX1, and thus 8‑cell‑like identity, is turned on. Using a stepwise chemical protocol, they converted “primed” stem cells through an intermediate “naive” state into 8CLCs. Under standard conditions, only a modest fraction of cells became TPRX1‑positive. When they deleted the OTX2 gene using CRISPR, the proportion of 8CLCs nearly doubled to about 28 percent. These OTX2‑deficient cells switched on many 8‑cell‑stage genes and specific mobile DNA elements typically active only at this time in embryos. Their overall gene activity patterns and DNA accessibility closely matched those of natural 8‑cell and morula (slightly later) embryos. Crucially, when introduced into early mouse embryos, these cells contributed not only to fetal tissue but also to extraembryonic structures like the placenta, a hallmark of totipotent‑like potential. 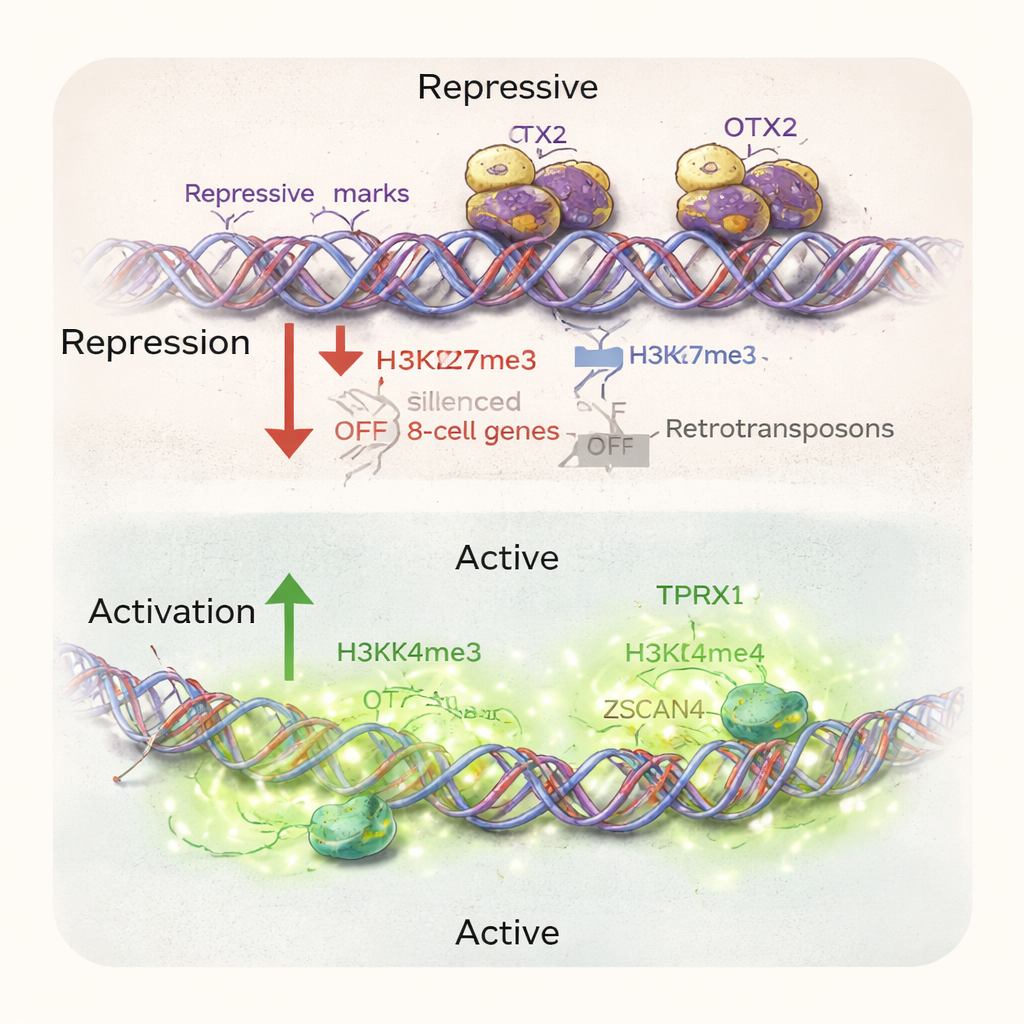
How OTX2 Silences the Earliest Embryo Program
Digging deeper, the authors mapped where OTX2 binds on DNA in these reprogrammed cells. They found that OTX2 sits directly on stretches of DNA near many 8‑cell‑specific genes and their associated repetitive elements. Where OTX2 is present, the surrounding chromatin carries molecular “off” marks and is relatively closed; where OTX2 is absent, these regions open up and gain “on” marks linked to active genes. Overexpressing OTX2 in the cells had the opposite effect of deletion: it suppressed 8‑cell‑like genes and reduced the number of 8CLCs. Interestingly, this action was largely independent of another famous early‑embryo gene, DUX4. While both influence many of the same targets, OTX2 does not simply switch DUX4 itself on or off; instead, it acts as an upstream repressor that helps keep the 8‑cell program locked away.
From Early Embryos to Future Therapies
Taken together, the study shows that OTX2 acts as a molecular gatekeeper that stops human pluripotent stem cells from reverting to an 8‑cell‑like, totipotent‑like condition. Removing OTX2 opens this gate, allowing a sizable fraction of cells to more faithfully mimic the gene activity, epigenetic landscape, and developmental range of real 8‑cell and morula embryos. For a layperson, this means scientists have identified a key switch that helps decide whether a cell remains in a flexible but limited state, or regains the broader potential of the very earliest stages of human life. In the long term, learning how to safely control this switch could lead to new ways of generating cells for research, improving cloning‑related technologies, and perhaps one day treating diseases by rebuilding damaged tissues from the ground up.
Citation: Kong, X., Jiang, N., Chen, S. et al. OTX2 inhibits human pluripotent stem cell reprogramming toward 8-cell-like and morula-like states. Nat Commun 17, 1685 (2026). https://doi.org/10.1038/s41467-026-68388-2
Keywords: totipotent stem cells, zygotic genome activation, OTX2, early human embryo, cell reprogramming