Clear Sky Science · en
Mechanical rejuvenation of senescent stem cells and aged bone via chromatin remodeling
Why keeping bones young matters
As we age, our bones gradually lose strength, becoming thinner and more fragile. This raises the risk of fractures, loss of independence, and chronic pain. Most current treatments focus on drugs that slow bone breakdown or add minerals, but they do not directly repair the aging of the stem cells that build bone. This study explores a different angle: using carefully controlled physical forces—rather than medicines—to “rejuvenate” aging bone-forming stem cells and improve bone health and overall fitness in older mice.
When bone’s builders grow old
Deep inside our bones live bone marrow mesenchymal stem cells, the master builders that can turn into bone-forming cells. With age, these stem cells divide less, form less bone, and show classic signs of cellular aging, such as DNA damage and inflammatory signals. The researchers compared cells from younger and older human donors and found that aged stem cells were not just biochemically different—they were mechanically weaker. They generated less internal pulling force, had floppier internal scaffolds, and showed poorer transmission of mechanical signals into the cell nucleus. In bone tissue from old mice, a similar decline appeared: the spongy bone network was thinned, key mechanical signaling proteins were reduced, and aging markers were elevated.
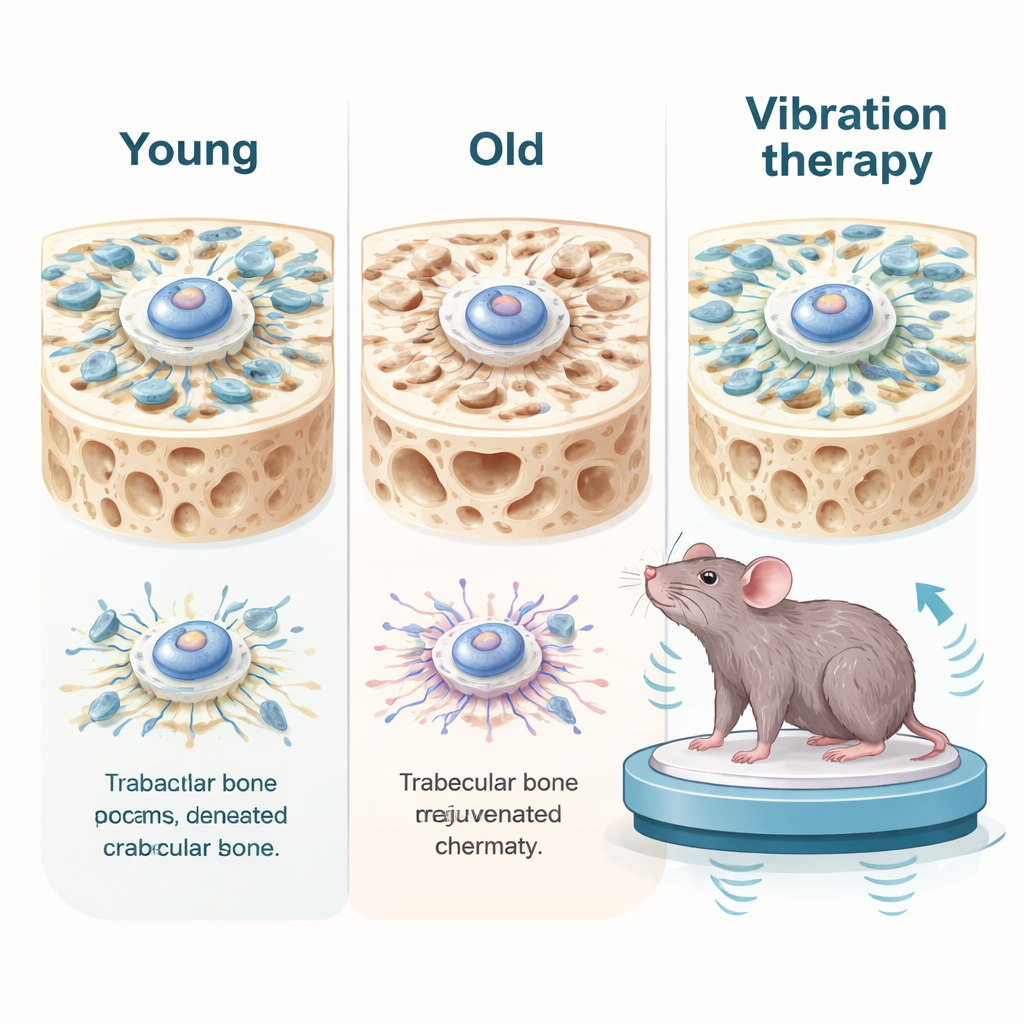
Gently pulling cells back to youth
The team then asked whether restoring mechanical force could reverse these aging features. In dishes, they applied mild stretching to senescent stem cells or used a drug that boosts the activity of molecular motors. These treatments strengthened cellular pulling forces and improved the organization of the internal scaffold. Remarkably, aged cells began to look and act younger: markers of senescence dropped, DNA damage lessened, and the cells multiplied more readily. The opposite was also true—when they softened the environment under young cells or chemically weakened their contractile machinery, the cells slid into an aging-like state, slowing down and turning on aging markers even in a bone-mimicking 3D gel.
How force talks to genes
To understand how physical forces could change cell behavior so deeply, the researchers turned to chromatin—the packaged form of DNA inside the nucleus. In old stem cells, chromatin was more tightly condensed and less accessible, making it harder for helpful genes to turn on. Restoring mechanical force caused the chromatin to relax locally, increasing chemical marks associated with open, active DNA. Using a genome-wide assay of chromatin accessibility, the team found that gentle mechanical stimulation opened up regions near genes involved in aging control. One gene, called FOXO1, stood out as especially sensitive to force. When force was increased, FOXO1’s DNA region became more open, its activity rose, and cells became less senescent. Blocking FOXO1 erased the benefits of mechanical stimulation, revealing it as a key switch that links physical strain to genetic programs for resilience and repair.
Finding the sweet spot of mechanical therapy
The researchers then tested mechanical stimulation in living mice. Older animals were placed on a low-intensity vibration platform for short daily sessions over a month, while others had their hind legs unloaded by tail suspension, mimicking extreme inactivity. In aged mice, gentle vibration boosted mechanical signaling in bone, reduced aging and inflammatory markers, and partially rebuilt the spongy bone network. It also improved grip strength, exercise endurance, memory-related tasks, and lowered inflammation in the blood, liver, and kidneys. However, when vibration was applied continuously without rest days, the benefits disappeared and even reversed: bone structure worsened, DNA damage increased, and inflammation rose. In cell studies, excessively strong or frequent stretching caused DNA breaks and pushed cells back into senescence, showing that mechanical force can heal or harm depending on dose.
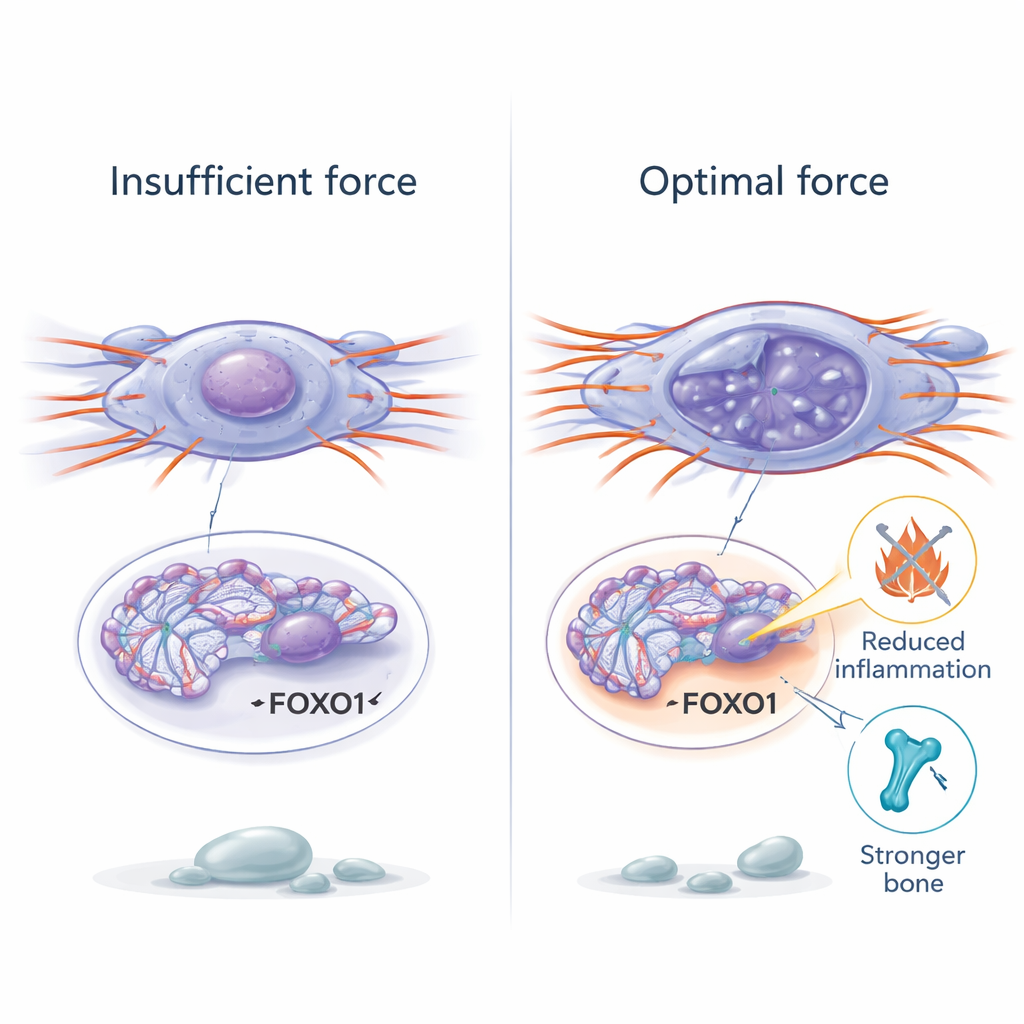
From exercise mimics to future treatments
To a layperson, the central message is that our cells do not just respond to hormones and chemicals—they also “listen” closely to physical forces. In this work, the authors show that moderate, well-tuned mechanical stimulation can reawaken aging bone stem cells, open up protective gene programs such as FOXO1, rebuild weakened bone, and calm chronic inflammation in older mice. At the same time, too little or too much force accelerates cellular wear and tear. These findings hint that carefully designed mechanical therapies—perhaps specific exercise regimens or safe vibration-based devices—could one day complement drugs in keeping bones, and possibly other tissues, healthier for longer.
Citation: Liu, X., Ye, Y., Li, Z. et al. Mechanical rejuvenation of senescent stem cells and aged bone via chromatin remodeling. Nat Commun 17, 1684 (2026). https://doi.org/10.1038/s41467-026-68387-3
Keywords: bone aging, mechanical stimulation, stem cell rejuvenation, chromatin remodeling, FOXO1