Clear Sky Science · en
Efficient and stable catalytic hydrolysis of perfluorocarbon enabled by SO2-mediated proton supply
Why this stubborn greenhouse gas matters
Some industrial gases are so stable that once released, they linger in the atmosphere for tens of thousands of years. Tetrafluoromethane (CF4), a type of perfluorocarbon used and emitted in aluminum smelting and chip fabrication, is one of the worst offenders: it traps heat about 7,400 times more effectively than carbon dioxide. This study explores a new way to break down CF4 efficiently and reliably, turning an almost indestructible pollutant into safer products under conditions that are realistic for factories.
A tough molecule that refuses to break
CF4 belongs to the broader family of PFAS, chemicals notorious for their persistence in the environment. What makes CF4 especially challenging is its extremely strong carbon–fluorine bonds and its very long atmospheric lifetime, estimated at more than 50,000 years. Traditional methods to destroy CF4 require very high temperatures and often lead to rapid wear and loss of activity in the catalysts that drive the reaction. Yet new climate policies, such as the European Union’s Carbon Border Adjustment Mechanism, are putting increasing pressure on heavy industries to clean up these emissions without greatly increasing their energy use.
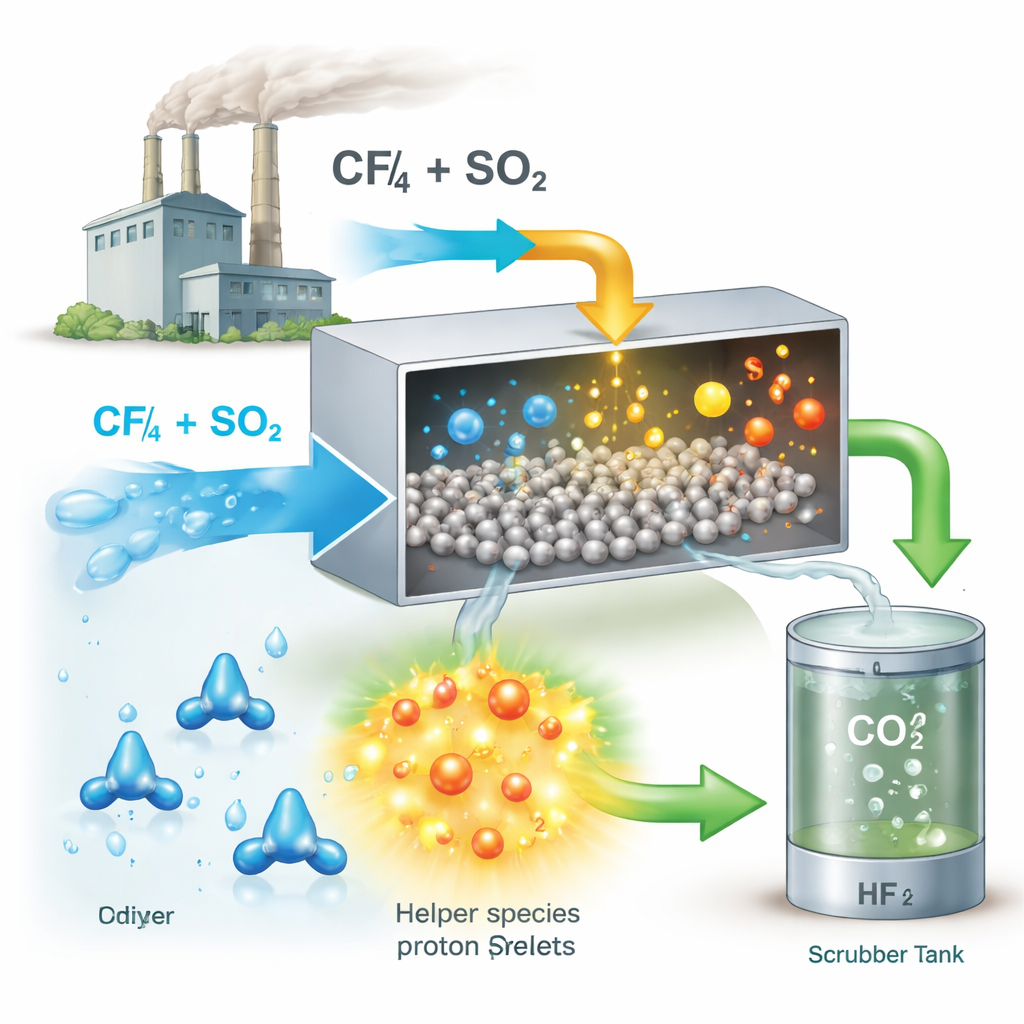
Turning a common pollutant into a helper
Surprisingly, the researchers found that another familiar pollutant, sulfur dioxide (SO2), can be used to solve the CF4 problem. SO2 is commonly released alongside CF4 from aluminum production. While SO2 usually harms catalysts by sticking to their surfaces, the team showed that under the right conditions it can do the opposite: it reshapes the catalyst surface so that water breaks apart more easily and supplies more reactive hydrogen ions (protons). These protons are essential for weakening the stubborn bonds in CF4 and for cleaning fluorine off the catalyst so it can keep working.
Building proton “fueling stations” on the surface
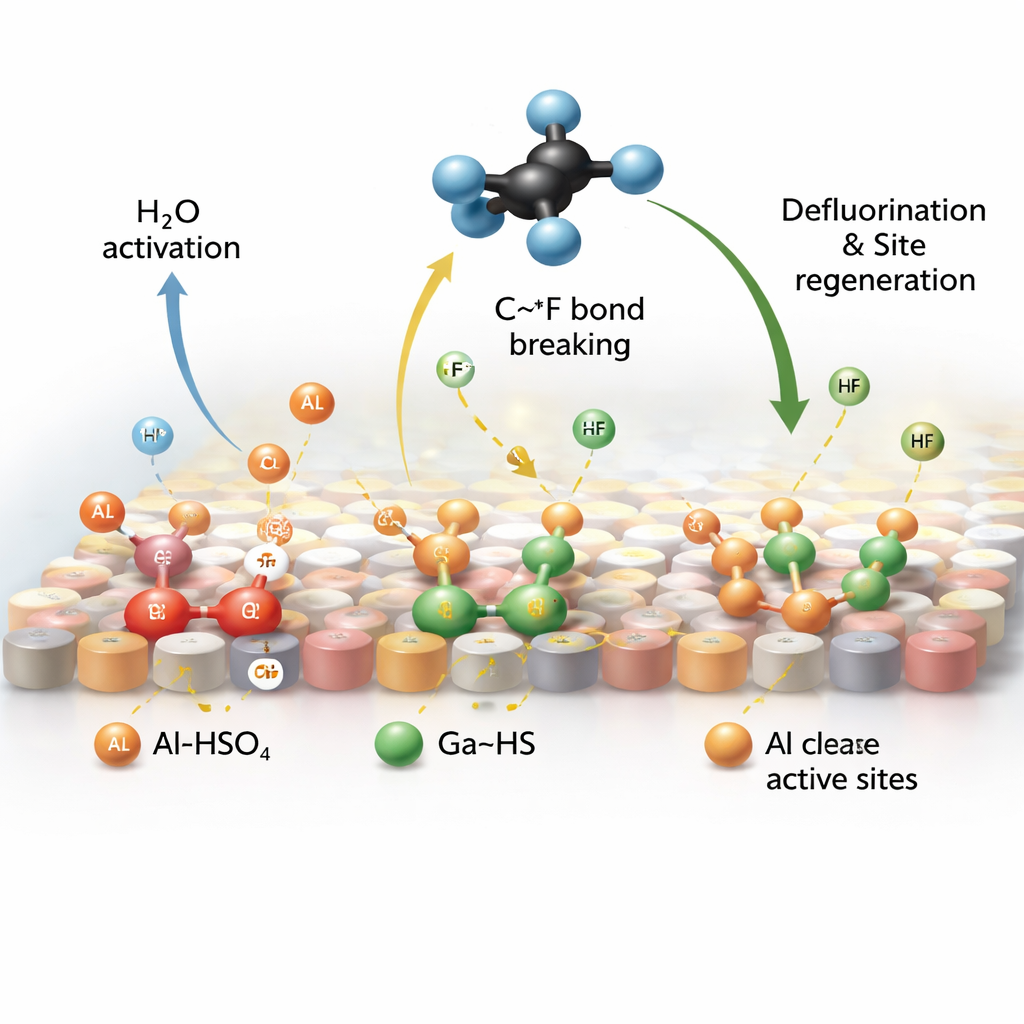
The key advance is the creation of special proton-rich sites directly on the catalyst, which is based on aluminum oxide doped with gallium. When SO2, water vapor, and CF4 flow over this material at high temperature, SO2 transforms into tightly bound acidic groups on the surface. Two families of such groups appear: one attached mainly to aluminum (Al–HSO4) and one to gallium (Ga–HS). Using sensitive spectroscopic tools and computer simulations, the authors show that the aluminum-based groups draw in CF4 and help split water to release protons, while the gallium-based groups use those protons to strip fluorine off poisoned sites and release it as hydrogen fluoride, restoring the catalyst’s activity.
Record performance under real-world conditions
Because these proton “fueling stations” are anchored strongly and remain stable at high temperatures, they supply reactive hydrogen far more effectively than traditional additives. The study finds that water activation is increased by about sixfold and proton availability by roughly tenfold compared with operation without SO2. As a result, complete breakdown of CF4 is achieved at 550 °C instead of the usual 700 °C, cutting the energy demand of the process. Just as important, the catalyst runs for more than 2,500 hours—over three months of continuous operation—without noticeable loss of performance, and it works across a wide range of SO2 levels matching those in industrial exhaust streams.
A new route to cleaning persistent air pollutants
For non-specialists, the outcome can be viewed as teaching a catalyst a new trick: using an unwanted gas (SO2) to build tiny, robust acid sites that feed it the protons it needs to chew through one of the toughest greenhouse gases known. By making both the destruction of CF4 easier and the catalyst longer-lived, this strategy points toward practical scrubbers that could be bolted onto smokestacks in aluminum and semiconductor plants. More broadly, the same concept of in-place proton regulation may be adapted to dismantle other gas-phase PFAS, offering a promising tool for reducing the long-term climate and environmental footprint of advanced manufacturing.
Citation: Zhang, H., Luo, T., Chen, Y. et al. Efficient and stable catalytic hydrolysis of perfluorocarbon enabled by SO2-mediated proton supply. Nat Commun 17, 597 (2026). https://doi.org/10.1038/s41467-026-68386-4
Keywords: tetrafluoromethane, PFAS destruction, catalytic hydrolysis, sulfur dioxide promotion, industrial emissions control