Clear Sky Science · en
Precise excision of expanded GGC repeats in NOTCH2NLC via CRISPR/Cas9 for treating neuronal intranuclear inclusion disease
Editing DNA to Tackle a Mysterious Brain Disorder
Neuronal intranuclear inclusion disease (NIID) is a rare but devastating brain disorder that can cause dementia, movement problems, and even early death. There is currently no cure. This study explores whether a powerful gene-editing tool, CRISPR, can be used to snip out a tiny but toxic stretch of DNA that drives NIID, offering a glimpse of how future treatments might rewrite our genes to combat brain disease. 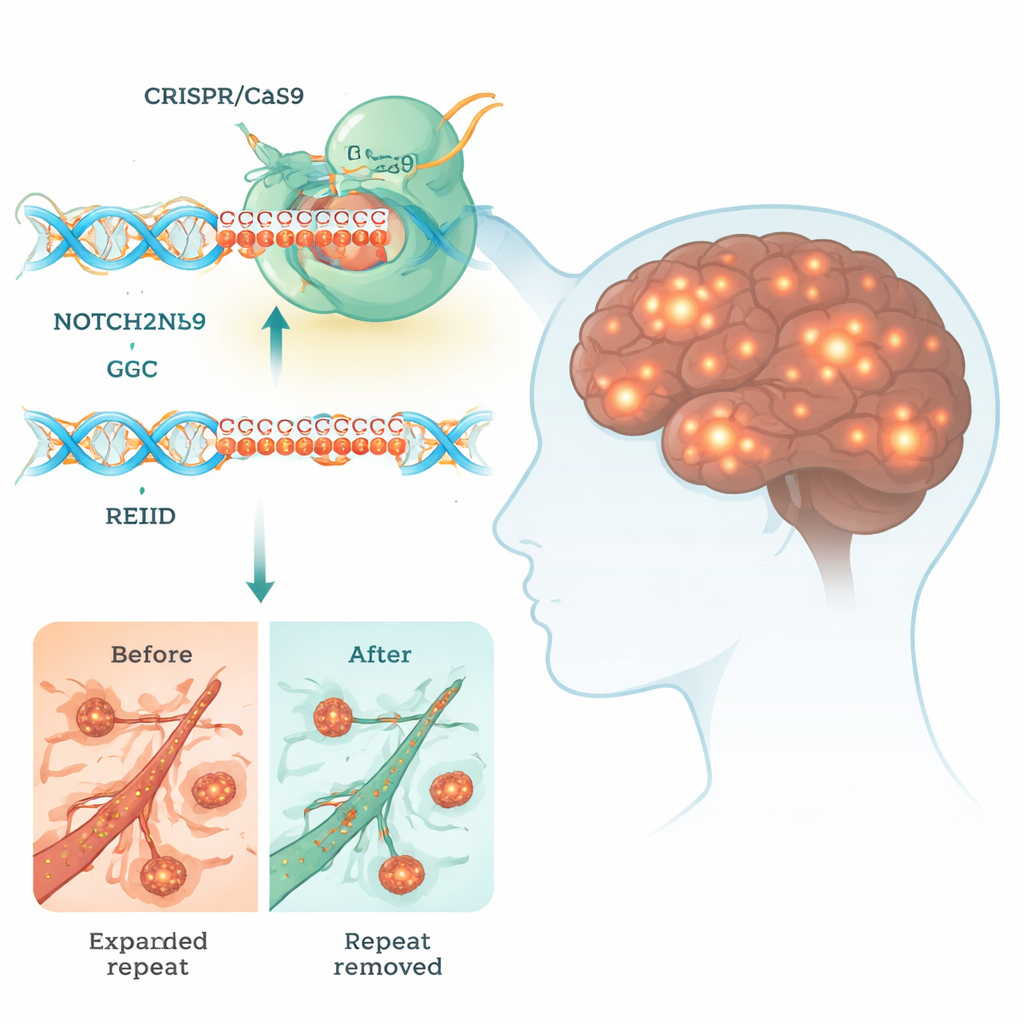
A Hidden Genetic Culprit
Many inherited brain disorders are caused by short DNA sequences that have grown far beyond their normal length. In NIID, the problem is an overgrown run of three-letter DNA units, "GGC," in a gene called NOTCH2NLC. Although this stretch sits in a region that does not directly code for a classic protein, it can still be read by the cell’s machinery to produce abnormal, polyglycine (polyG) chains. These sticky chains clump into blobs inside the nucleus of nerve cells and other tissues, disrupting vital processes such as energy production, RNA processing, and the traffic of molecules between nucleus and cytoplasm.
The Challenge of Precise Gene Surgery
Designing a gene-editing treatment for NIID is unusually hard because NOTCH2NLC has several nearly identical sister genes that are important for human brain development. If CRISPR cuts the wrong one, it could do more harm than good. The researchers carefully scanned the DNA around the GGC repeat and found tiny sequence differences that distinguish NOTCH2NLC from its relatives. They then designed pairs of CRISPR guide RNAs that direct the Cas9 "molecular scissors" to cut just before and after the expanded repeat. This dual-cut strategy lets the cell’s repair system remove the toxic segment while leaving the rest of the gene family untouched.
From Cells in a Dish to Patient-Derived Neurons
The team first tested their CRISPR design in standard human cells and in cells engineered to carry a large NOTCH2NLC repeat. In these models, the editor efficiently chopped out the repeat, and the levels of polyG clumps dropped by roughly half or more. They then moved to induced pluripotent stem cells (iPSCs) made from a patient with NIID and converted these cells into neural progenitor cells, an early stage of brain cells. Using CRISPR, they either deleted the expanded repeat or replaced it with a normal-length version. Detailed DNA and whole-genome sequencing showed that editing was highly accurate, with little evidence of unwanted cuts, and the edited cells still divided and matured normally. 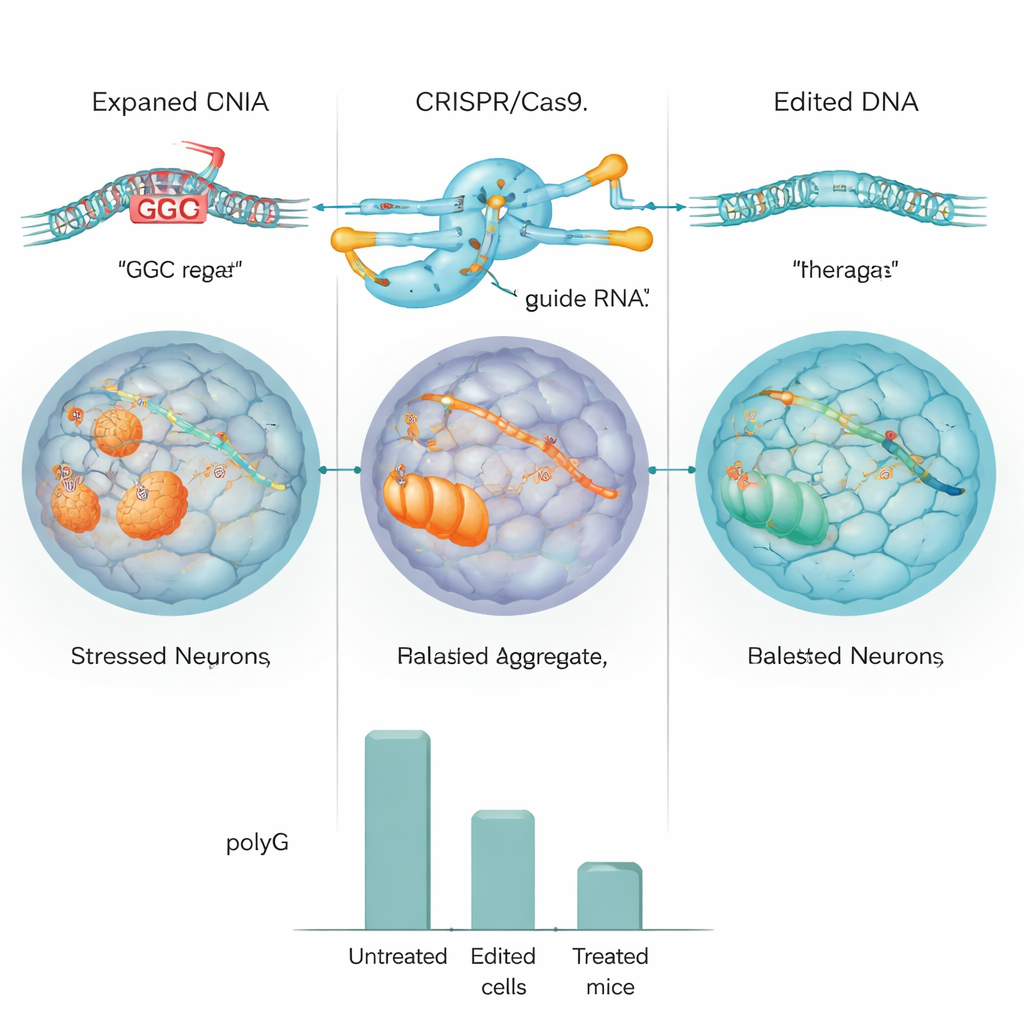
Rescuing Brain and Body in a Mouse Model
To see whether this approach could actually improve disease, the researchers turned to mice engineered to carry the human NOTCH2NLC repeat and to develop NIID-like features, including nuclear inclusions, movement problems, and shortened lifespan. They packaged the CRISPR system into a modified virus that can spread throughout the brain after a simple injection into the bloodstream of newborn mice. In treated animals, the expanded repeat was successfully removed in brain tissue. As a result, toxic polyG levels fell sharply, markers of nerve cell health improved, and abnormal activation of supporting brain cells eased. Behavior tests showed that treated mice moved more, balanced better, and survived longer than their untreated littermates. Similar benefits were seen in the heart, which also accumulates polyG in this model.
What This Could Mean for Future Treatments
To a layperson, the main message is that NIID appears to be driven largely by one specific overgrown DNA repeat, and precisely removing that repeat can reverse many signs of disease in cells and in mice. This work does not yet translate into a ready-to-use human therapy: researchers still need to refine long-term safety, improve delivery methods, and test in larger, more human-like animals. But the study offers a strong proof of concept that carefully targeted gene editing can safely silence a toxic repeat while sparing closely related genes. That strategy may one day be adapted not only for NIID, but also for other brain and muscle disorders caused by similar repeat expansions.
Citation: Xie, N., Pan, Y., Tong, H. et al. Precise excision of expanded GGC repeats in NOTCH2NLC via CRISPR/Cas9 for treating neuronal intranuclear inclusion disease. Nat Commun 17, 1683 (2026). https://doi.org/10.1038/s41467-026-68385-5
Keywords: CRISPR gene editing, neuronal intranuclear inclusion disease, repeat expansion disorders, NOTCH2NLC, neurodegeneration