Clear Sky Science · en
Tmem110 regulates the conformation of TRPML1 to maintain endolysosomal homeostasis and prevent mitochondrial DNA leakage and pathological self-DNA processing
Why spilled DNA after injury matters
When we suffer severe trauma, such as a major blow to the head, the damage does not stop at the injury site. Many patients go on to develop a dangerous whole‑body reaction in which the lungs, liver, kidneys, and other organs begin to fail. This study reveals how tiny structures inside cells—lysosomes, mitochondria, and immune sensors—talk to each other to decide whether the body quietly cleans up the debris or spirals into a self‑directed immune attack. Understanding this hidden cleanup system could open new ways to prevent organ failure after trauma and to treat autoimmune and inflammatory diseases.
From head injury to whole‑body crisis
Traumatic brain injury (TBI) can trigger a powerful inflammatory storm called multiple organ dysfunction syndrome (MODS), but how damage in the brain spreads to distant organs has been unclear. The authors focus on monocytes and macrophages, immune cells that patrol the blood and tissues and act as professional garbage collectors. After TBI, cells die in an uncontrolled way and release entire mitochondria—tiny energy factories that carry their own DNA—into the circulation. This mitochondrial DNA (mtDNA) looks suspiciously like bacterial DNA to the immune system and can switch on strong antiviral‑style defenses if not removed quickly. Normally, cells internalize these stray mitochondria into acidic compartments called lysosomes, where enzymes digest the DNA before it leaks into the cell fluid. 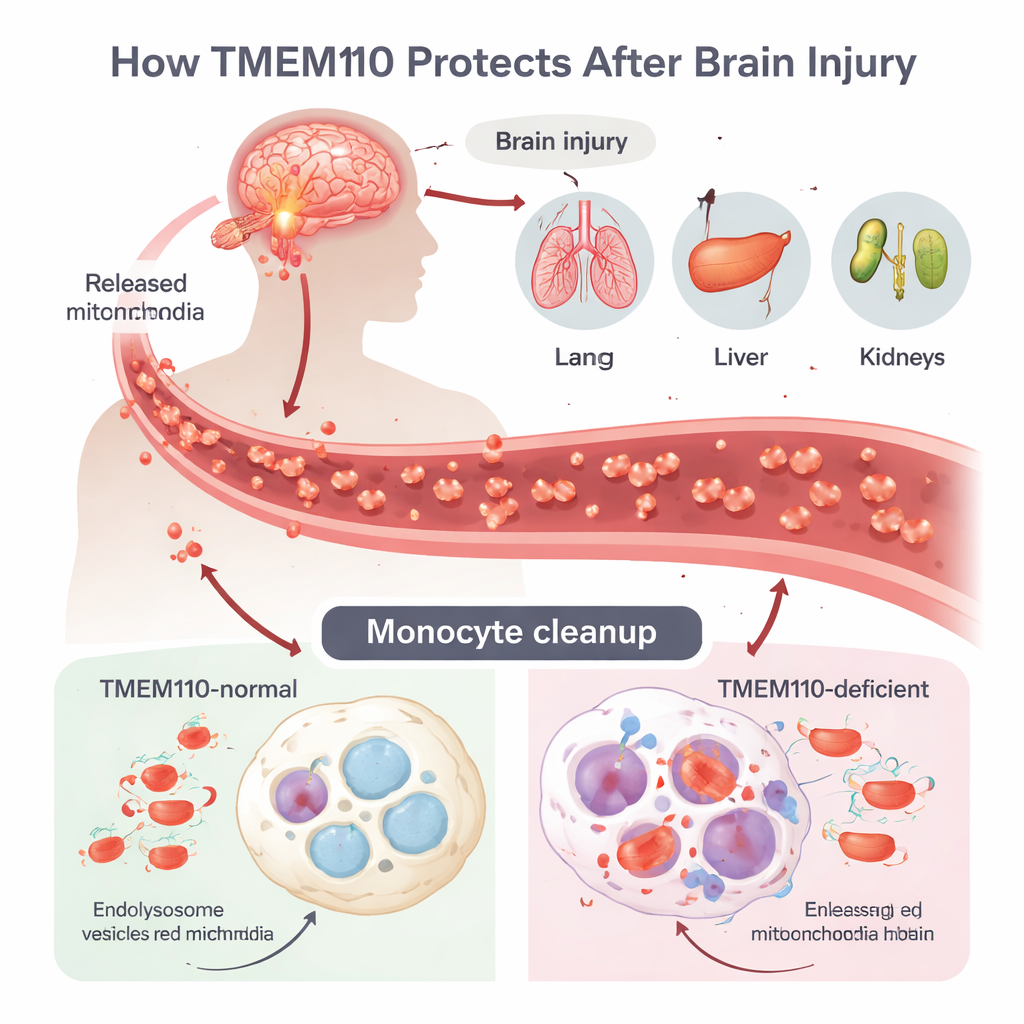
A gatekeeper protein that keeps lysosomes stable
The team identified TMEM110, a protein that spans the membrane of the endoplasmic reticulum (a major intracellular membrane network), as a crucial gatekeeper in this process. Using genetically engineered mice that lacked TMEM110 only in monocytes and macrophages, they created a controlled brain‑injury model. Compared with normal mice, TMEM110‑deficient animals developed much higher levels of type I interferons—potent antiviral signaling molecules—in their blood and cerebrospinal fluid. Their lungs, liver, kidneys, and lymph nodes showed more severe damage, and their long‑term survival after TBI was markedly worse. When the researchers blocked the receptor for type I interferons, or replaced the mutant bone marrow with normal cells, organ injury and death rates fell, showing that excessive interferon signaling from peripheral monocytes is a key driver of MODS in this setting.
How faulty ion handling lets DNA escape
Diving inside these immune cells, the authors found that TMEM110‑deficient lysosomes were chemically unbalanced: they were less acidic and overloaded with calcium. Under these conditions, the enzyme DNase II—which normally chops up DNA in lysosomes—lost activity, lysosomal membranes became fragile, and mtDNA escaped into the surrounding cytosol. Laboratory experiments with human monocytes engulfing labeled donor mitochondria confirmed that, without TMEM110, more mtDNA leaked into the cell fluid and triggered a surge in interferon‑stimulated genes. The study shows that TMEM110 works by controlling another protein, the lysosomal ion channel TRPML1. When TMEM110 is present, it physically contacts TRPML1 and shifts it into an “open” conformation that creates tiny calcium‑release hotspots at the lysosome surface. These controlled outflows of calcium help maintain the right acidity, support membrane repair, and keep mtDNA trapped and degraded inside. 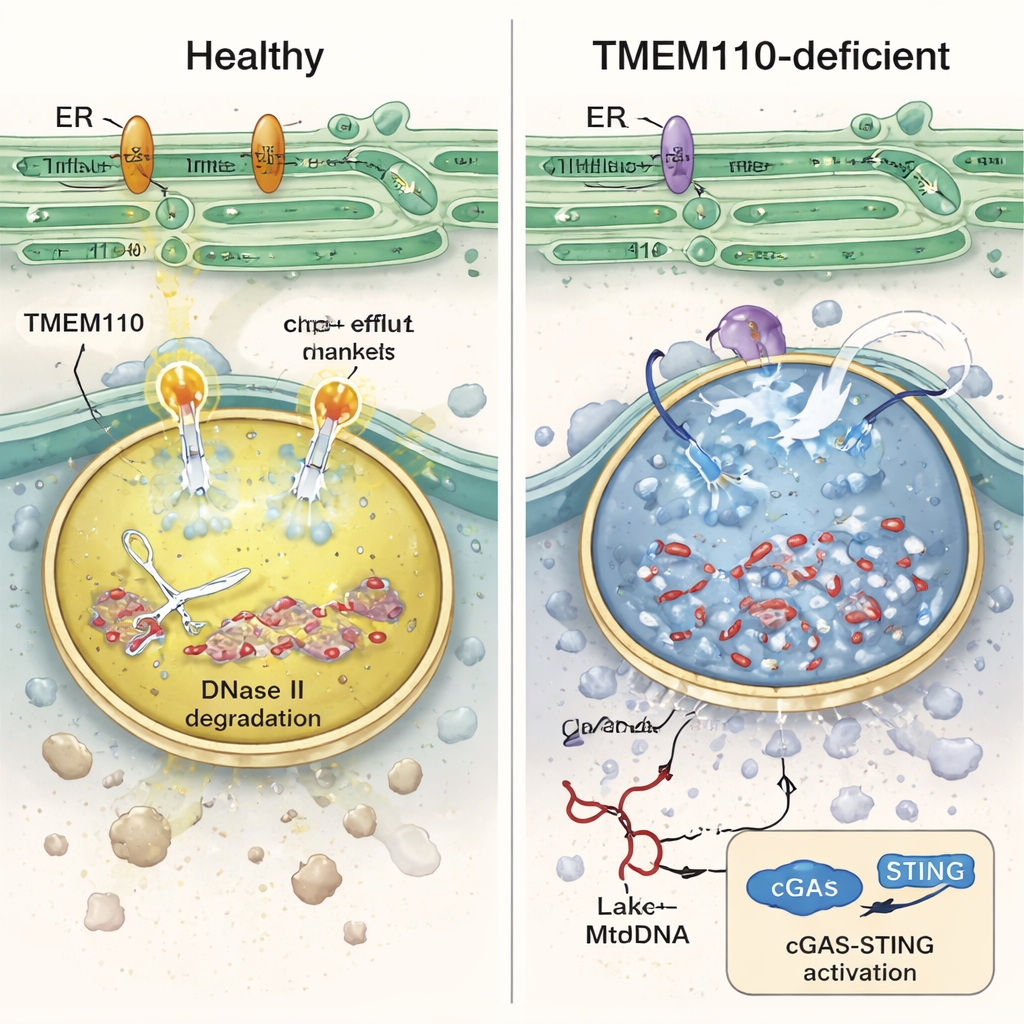
A feedback loop with the cell’s DNA alarm system
Once mtDNA spills into the cytosol, it is recognized by the cGAS–STING pathway, the cell’s main alarm system for stray DNA. Interestingly, TMEM110 also binds STING at rest and keeps it sequestered in the endoplasmic reticulum, preventing unnecessary alarm. When cytosolic DNA accumulates, STING becomes activated and loosens its grip on TMEM110. That, in turn, exposes a different region of TMEM110 that engages TRPML1 and boosts lysosomal calcium efflux, creating a feedback loop: sensing self‑DNA activates a mechanism that enhances DNA clearance. Disease‑linked mutations in STING found in human autoinflammatory syndromes disrupt this hand‑off, trapping TMEM110 in an inhibited state and contributing to chronic interferon activation. In mice carrying such a STING mutation, TBI led to worse organ damage and higher mortality, but delivering a small TMEM110 fragment to lysosomes using an mRNA–lipid nanoparticle therapy partially restored control of mtDNA and improved survival.
Clues from patients and paths to new treatments
The authors then turned to a clinical cohort of 143 people with trauma‑related brain injury and MODS. Patients whose monocytes had higher TMEM110 levels early after injury were more likely to recover organ function and survive. Those with lower TMEM110 showed more circulating mtDNA and decellularized mitochondria, stronger interferon responses, higher scores of organ failure, and broader autoantibody profiles, including antibodies against DNA and nuclear proteins. These patterns closely mirrored the findings in mice, supporting the idea that TMEM110‑driven control of lysosomal stability and mtDNA disposal is a shared mechanism in human disease.
What this means for patients with severe trauma
In everyday terms, this work suggests that after a major injury our immune system faces a delicate choice: silently digest the flood of self‑DNA released from dying cells, or interpret it as a viral invasion and mount an all‑out attack that can damage our own organs. TMEM110, by tuning a lysosomal ion channel and coordinating with the DNA sensor STING, helps cells choose the safer path. When this system fails—because TMEM110 is missing, STING is mutated, or lysosomes are overwhelmed—mtDNA escapes, the alarm keeps blaring, and multi‑organ damage follows. Targeting this TMEM110–TRPML1–STING axis, for example with lysosome‑directed mRNA therapies, could offer a new way to prevent or treat organ failure and autoimmune‑like complications after severe trauma.
Citation: Feng, Z., Pan, Y., Zhou, J. et al. Tmem110 regulates the conformation of TRPML1 to maintain endolysosomal homeostasis and prevent mitochondrial DNA leakage and pathological self-DNA processing. Nat Commun 17, 1678 (2026). https://doi.org/10.1038/s41467-026-68382-8
Keywords: traumatic brain injury, mitochondrial DNA, lysosomes, type I interferon, cGAS-STING pathway