Clear Sky Science · en
Variations in the Natural History of High-Risk HPV Types Following HPV-16/18 Bivalent Vaccination in Females Aged 18-45 Years
Why this study matters to everyday health
Cervical cancer is one of the most preventable cancers, thanks to vaccines and regular screening. Yet the virus that causes it—human papillomavirus, or HPV—comes in many types, and vaccines do not cover them all. This study followed more than 7,000 women in China for about ten years to see how an HPV vaccine targeting two major types (HPV‑16 and HPV‑18) changed not only those specific infections, but also the behavior of other risky HPV types. The findings hint that as vaccination becomes widespread, the rules for cervical screening may need to evolve too.
Following women over a decade
Researchers conducted a large randomized clinical trial of an HPV‑16/18 bivalent vaccine made using E. coli. Women aged 18–45 were randomly assigned to receive either the HPV vaccine or a control (hepatitis E) vaccine and were then followed for up to ten years, including an extension study. At regular visits, doctors collected cervical samples to test for high‑risk HPV types and to check for abnormal cells or precancerous changes known as CIN2+ (cervical intraepithelial neoplasia grade 2 or worse). Because women were randomly assigned, differences seen later could be confidently linked to vaccination rather than lifestyle or background factors.
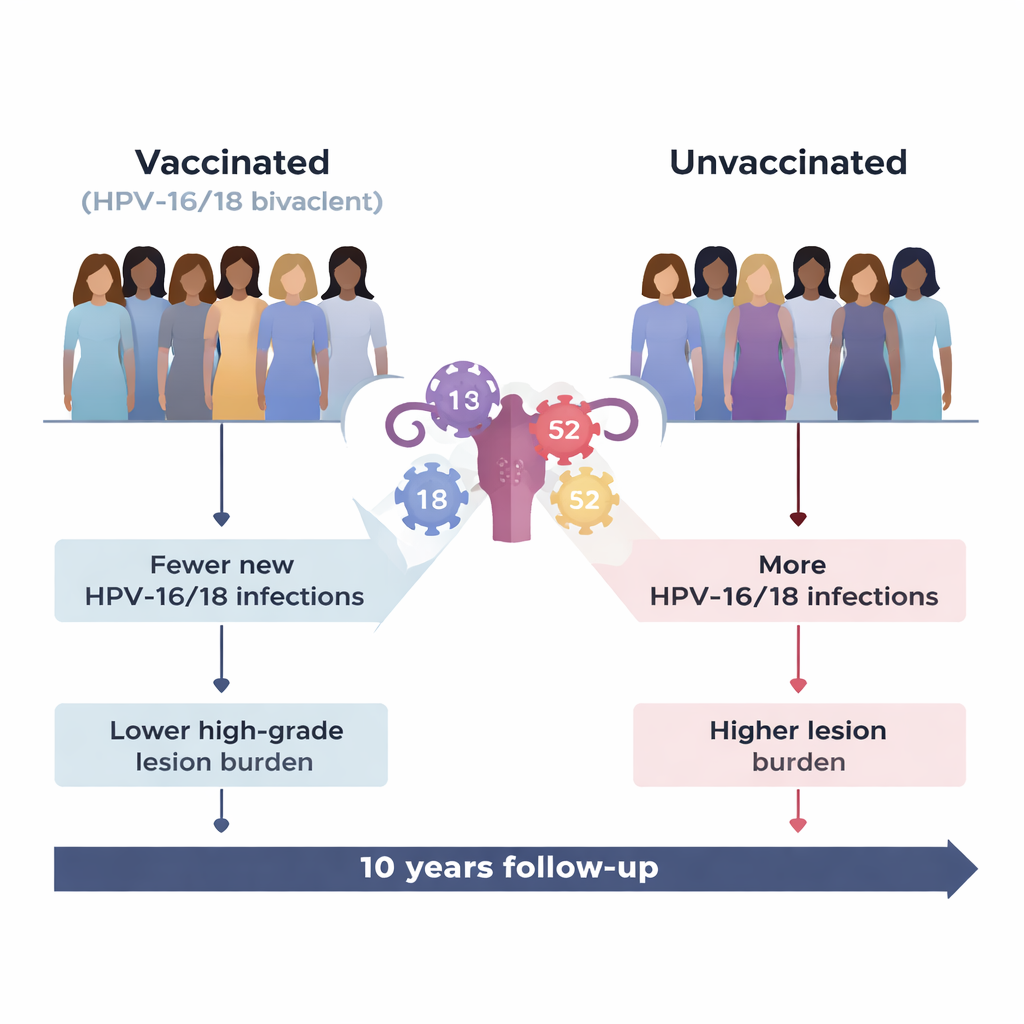
What the vaccine did as expected
The vaccine worked very well against the types it was designed for, HPV‑16 and HPV‑18, which together cause the majority of cervical cancers worldwide. Vaccinated women had far fewer new infections with these types and far fewer related high‑grade precancerous lesions compared with unvaccinated women. Even when a vaccinated woman did get a breakthrough HPV‑16/18 infection, that infection was more likely to clear on its own and less likely to progress to serious cervical changes. Overall, the burden of HPV‑16/18‑linked CIN2+ lesions in the vaccinated group was reduced by nearly 90 percent.
An unexpected rise of another risky type
When the researchers looked beyond the vaccine‑targeted types, a more complicated picture emerged. One non‑vaccine high‑risk type, HPV‑52, became relatively more important among vaccinated women. The overall chance of catching HPV‑52 in the first place was similar in both groups. However, once a woman was infected with HPV‑52, vaccination status appeared to change the course of that infection. In vaccinated women, HPV‑52 infections cleared less often, lingered longer, and were more likely to lead to high‑grade precancerous lesions than in unvaccinated women. Over the study period, HPV‑52 became the single most common cause of serious cervical lesions in the vaccinated group, accounting for nearly two in five new high‑grade cases.
Rethinking simple explanations
Scientists have long debated whether suppressing certain HPV types with vaccines might open an ecological “gap” that lets other types spread more easily, or whether interactions between types during co‑infection might change how they behave. This study did not find clear evidence that HPV‑52 infections became more frequent overall, nor that co‑infection with other types explained its behavior. Instead, the data suggest that vaccination may subtly influence how some non‑vaccine HPV types persist and progress, through mechanisms that are not yet understood. Importantly, despite this shift, the total number of serious lesions from all high‑risk types combined was still lower in vaccinated women than in controls.
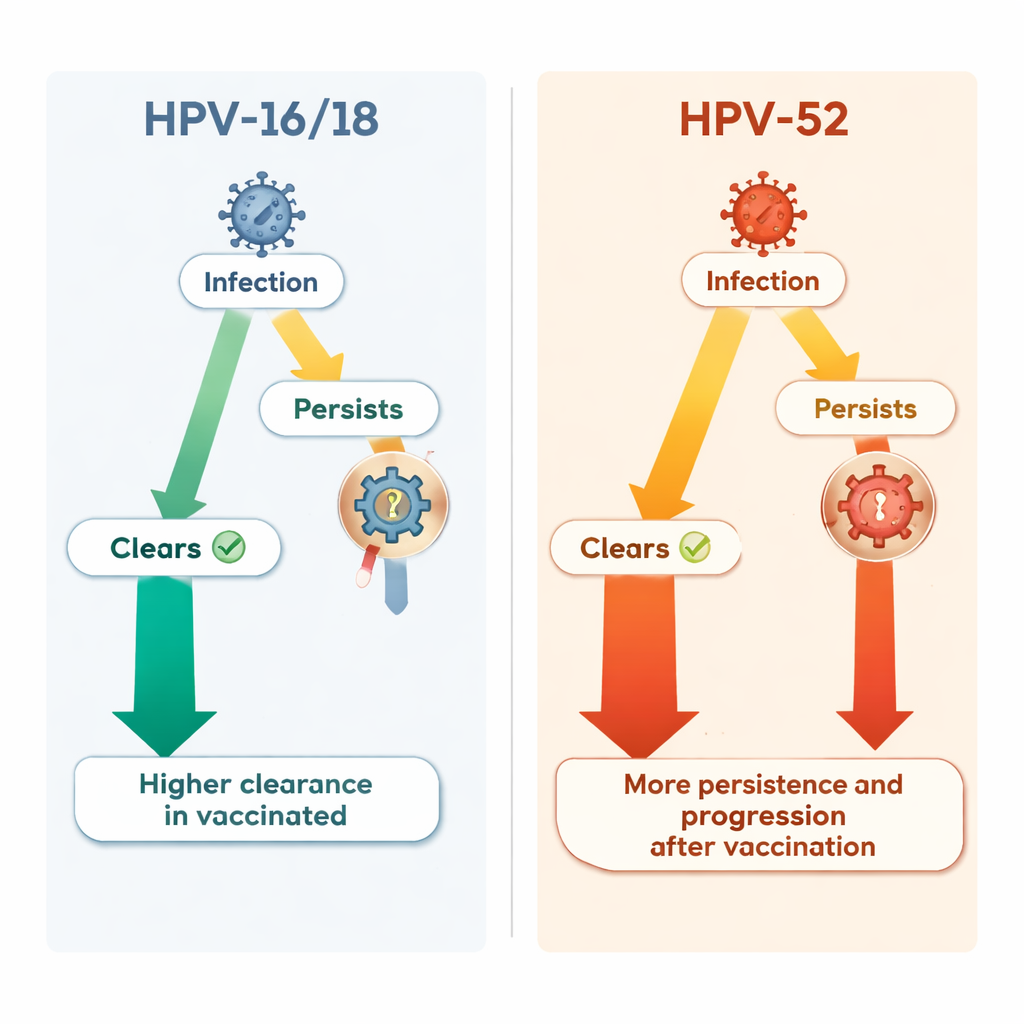
What this means for screening and vaccines
For people wondering what to do today, the message remains reassuring: vaccination against HPV‑16 and HPV‑18 provides major protection and clearly reduces the overall risk of dangerous cervical changes. However, as more vaccinated generations enter screening programs, tests that only flag HPV‑16/18 for urgent follow‑up may no longer be enough. Because HPV‑52 now plays a larger role in vaccinated women, guidelines for HPV testing and follow‑up may need to be updated so that high‑risk non‑vaccine types are recognized and managed appropriately. The authors argue that future vaccines should aim to cover more high‑risk HPV types, while health systems refine screening strategies to match the post‑vaccine reality.
Citation: Chen, Q., Quan, J., Zhu, K. et al. Variations in the Natural History of High-Risk HPV Types Following HPV-16/18 Bivalent Vaccination in Females Aged 18-45 Years. Nat Commun 17, 1677 (2026). https://doi.org/10.1038/s41467-026-68379-3
Keywords: HPV vaccination, cervical cancer screening, HPV-16/18 bivalent vaccine, HPV-52, cervical precancer