Clear Sky Science · en
Near-infrared light-driven nanomotors-based microneedles for the active therapy of bacterial infected acne
Why this acne research matters
Acne is often dismissed as a cosmetic nuisance, but for teenagers and adults alike it can be painful, scarring, and emotionally draining. Standard treatments, especially antibiotics, can cause side effects and drive drug‑resistant bacteria. This study describes a needle patch powered by harmless near‑infrared light that delivers tiny "nanomotors" directly into acne lesions. These smart particles generate their own oxygen, move actively through bacterial slime layers, and heat up just enough to kill germs and calm inflammation—offering a glimpse of future acne care that is more targeted and less reliant on antibiotics.
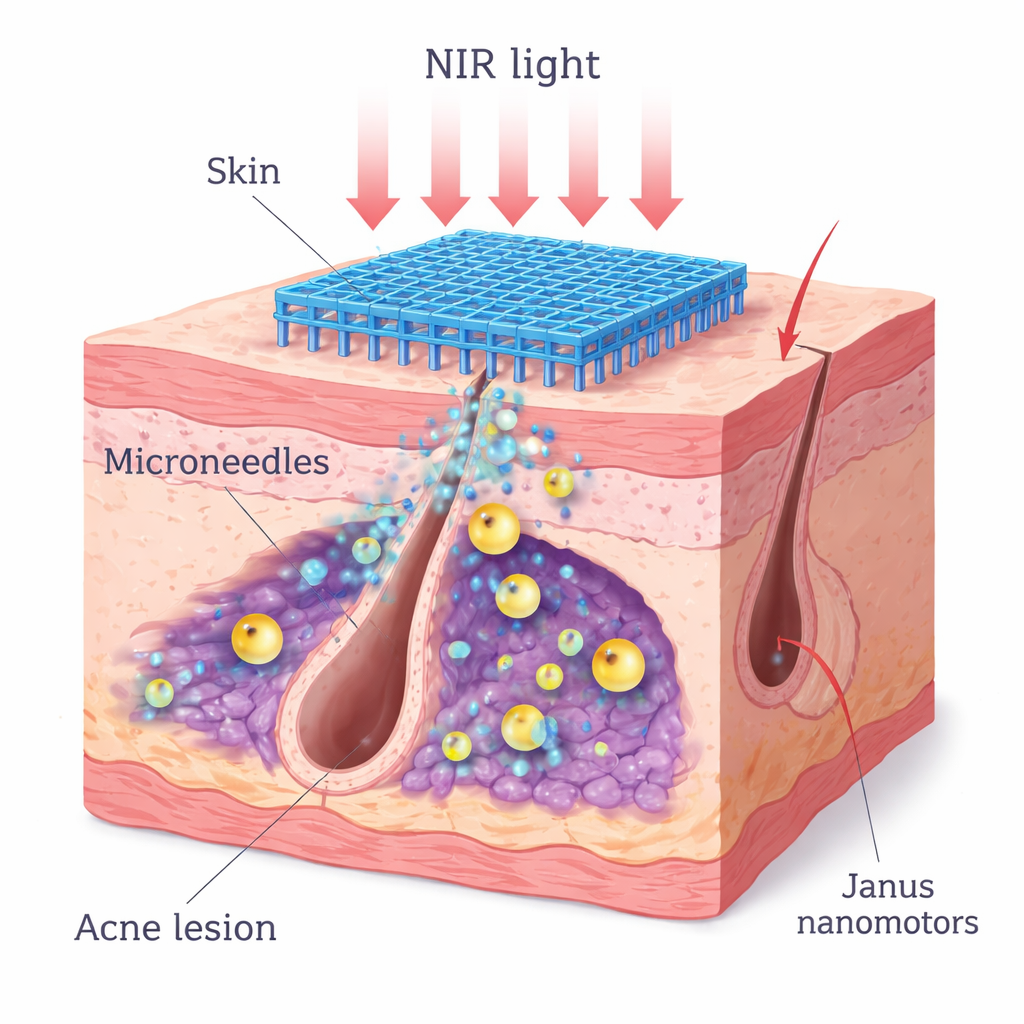
How acne turns into an inflamed bump
Most acne starts when pores and hair follicles become clogged with oil and dead skin cells. Inside these blocked follicles, a bacterium called Cutibacterium acnes (formerly Propionibacterium acnes) multiplies in the low‑oxygen environment. The microbes form protective biofilms—sticky communities wrapped in a dense matrix—that shield them from drugs. As they feed on skin oils, they release fatty acids that further reduce oxygen and irritate nearby cells. The skin responds by producing inflammatory signals such as TNF‑α and interleukins, and local immune cells that normally keep microbes in balance become depleted. Together, these changes drive the redness, swelling, and sometimes scarring seen in stubborn acne.
Why regular creams and pills often fall short
Topical creams and gels have trouble crossing the skin’s outer barrier, and oral antibiotics bathe the whole body in drug just to reach a small area, raising the risk of side effects and resistant strains. Inside a mature acne lesion, the thick biofilm around C. acnes further blocks penetration. Even photothermal therapy—using light‑absorbing particles to produce heat that kills bacteria—struggles, because the particles cannot move deeply into the biofilm, and the low‑oxygen, acidic environment around the lesion fuels chronic inflammation rather than healing.
A light‑activated microneedle patch with tiny engines
The researchers designed a dissolving microneedle patch that painlessly enters just below the skin surface and releases engineered nanomotors. Each nanomotor has a core of zinc peroxide that slowly breaks down in acidic conditions, releasing hydrogen peroxide, which is then converted into oxygen by a manganese dioxide shell. One side of the particle is coated with a light‑absorbing layer of polydopamine and manganese dioxide, creating a "Janus" (two‑faced) structure. When an 808‑nm near‑infrared laser shines on the skin, this asymmetric coating heats one side more than the other, creating a temperature gradient that drives the particle forward. This self‑propulsion helps the nanomotors spread through dense biofilms and follicle spaces while simultaneously delivering heat to weaken bacterial defenses.
From lab bench to mouse skin
In laboratory tests, the nanomotors heated efficiently under near‑infrared light, remained stable over repeated cycles, and released more hydrogen peroxide and oxygen under acidic, biofilm‑like conditions. Microneedle patches made from skin‑friendly hyaluronic acid were strong enough to pierce skin but dissolved within about half an hour, freeing the nanomotors into the dermis. Under the light beam, the particles showed clearly enhanced motion and penetrated deeper into both artificial biofilms and pig skin. In bacterial cultures of C. acnes and drug‑resistant Staphylococcus aureus, the combination of microneedles, nanomotors, and five minutes of near‑infrared exposure slashed biofilm mass and bacterial survival by more than 90 percent, and caused visible damage to microbial membranes and DNA.
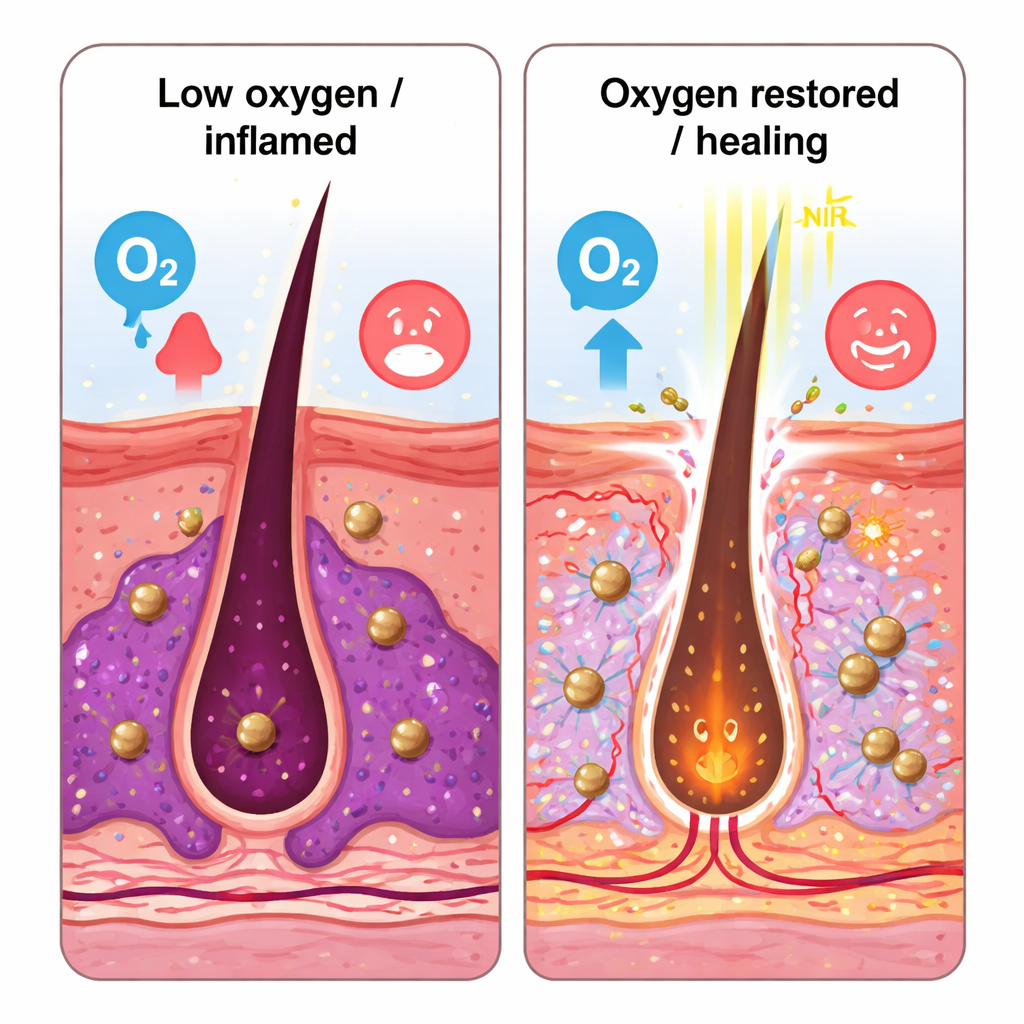
Calming inflammation and restoring balance
In a mouse model of acne, created by injecting C. acnes into the skin, the light‑activated patch reduced lesion size and bacterial counts as effectively as the antibiotic erythromycin, but without obvious tissue damage. Skin sections from treated mice showed fewer inflammatory cells, lower levels of inflammatory molecules (IL‑6, TNF‑α), and reduced activity of HIF‑1α, a marker of low oxygen. At the same time, markers of new blood vessel growth and wound repair rose, and key immune cells called ILC3, which had been suppressed by infection, rebounded and produced more of the healing factor IL‑22. The authors suggest that by supplying oxygen and physically breaking down biofilms, the nanomotors help normalize both the local microbiome and the skin’s immune environment.
What this could mean for future acne care
To a lay reader, the takeaway is that this microneedle‑nanomotor system acts like a smart, localized treatment: it gently opens a path through the skin, steers tiny engines into the heart of an acne lesion, and under a brief pulse of invisible light, delivers heat and oxygen exactly where they are needed. In mice, this approach cleared infection, eased inflammation, and supported tissue healing, matching antibiotic performance without bathing the whole body in drugs. While human trials and long‑term safety studies are still needed, the work points toward a new class of acne therapies that combine mechanical penetration, on‑demand activation, and self‑generated oxygen to tackle biofilm‑based infections more precisely and with fewer side effects.
Citation: Hu, Z., Gan, Y., Song, Y. et al. Near-infrared light-driven nanomotors-based microneedles for the active therapy of bacterial infected acne. Nat Commun 17, 1675 (2026). https://doi.org/10.1038/s41467-026-68376-6
Keywords: acne treatment, microneedle patch, nanomotors, near infrared therapy, bacterial biofilms