Clear Sky Science · en
Streamlined resource-efficient plasma amyloid-beta mass spectrometry assay has improved biomarker performance in preclinical Alzheimer’s disease
Why a Simple Blood Test for Alzheimer’s Matters
Alzheimer’s disease begins silently in the brain many years before memory problems appear. Today, confirming these early changes usually requires expensive brain scans or a spinal tap—procedures that are not widely available and can be uncomfortable. This study describes a greatly simplified blood test that can detect a key Alzheimer’s-related protein pattern in the blood with high accuracy, using less time, less blood, and less costly equipment. Such a test could make early detection and monitoring of Alzheimer’s more practical in everyday clinics and in large prevention trials.
From Complex Lab Test to Streamlined Tool
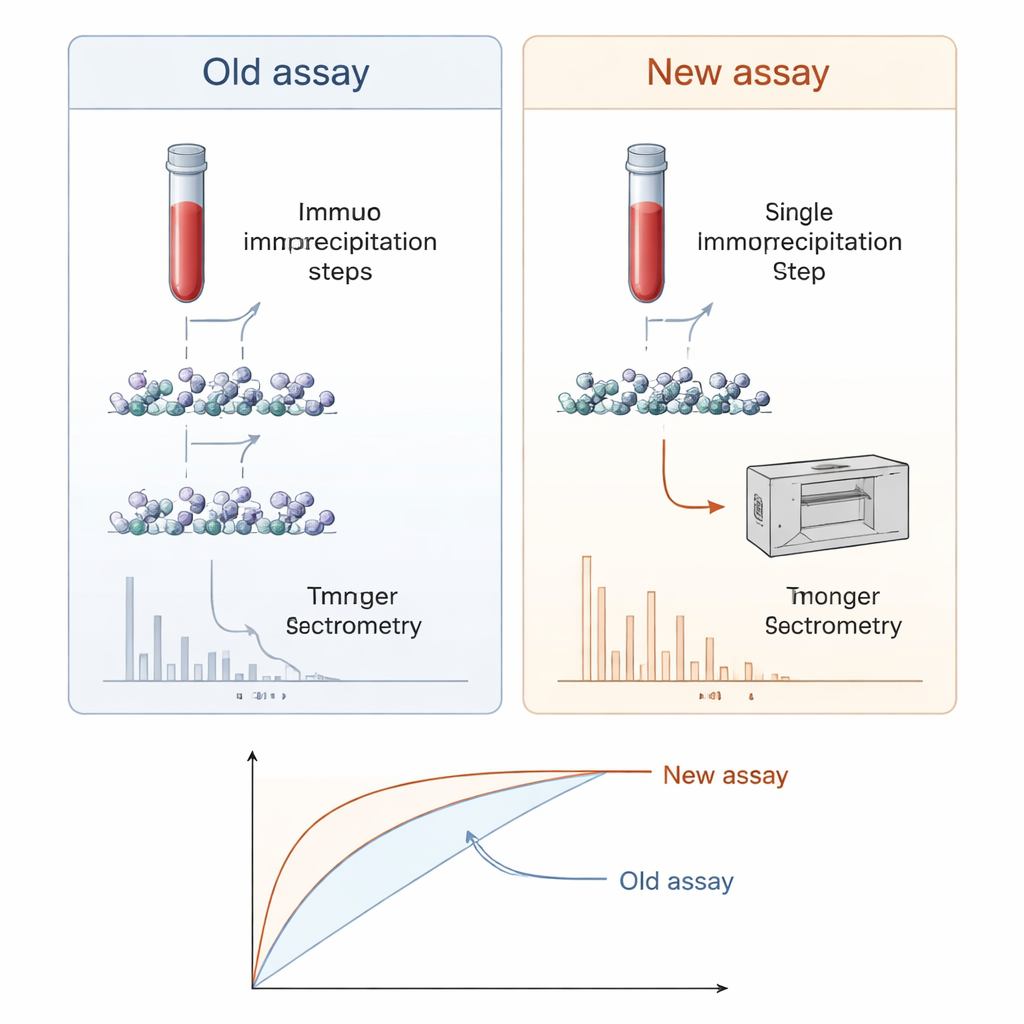
Researchers focused on amyloid-beta, a small protein that clumps into plaques in the brains of people with Alzheimer’s disease. A specific ratio of two forms, called Aβ1-42 and Aβ1-40, tends to fall in the blood when plaque builds up in the brain. A sophisticated method known as immunoprecipitation–mass spectrometry can measure these forms very precisely in plasma, but existing versions are slow, expensive, and require large blood volumes and highly specialized machines. The team had previously adapted a leading assay onto a more affordable tabletop mass spectrometer, but it still relied on two rounds of complex sample preparation, limiting how widely it could be used.
Making the Blood Test Faster, Cheaper, and Clearer
In the new work, the scientists redesigned the assay, creating what they call Pittsburgh Plasma Aβ version 2.0 (PAβ V2.0). They tested different detergents and blocking solutions to clean up the signal and reduce background “noise” from abundant blood proteins. A commercial cerebrospinal fluid buffer turned out to work best, allowing them to collapse the method down to a single antibody-capture step while sharply boosting the clarity of the mass spectrometry readout. With this optimized setup, they cut antibody use and sample volume requirements by about 75 percent, shortened preparation time from about six hours to three, and still achieved very precise measurements across a wide range of amyloid-beta concentrations.
Proving Reliability in Real People
To see if this streamlined test worked in practice, the team compared results from the new and old versions of the assay, as well as from a widely used commercial immunoassay, in several groups of older adults. Many of these volunteers were cognitively normal but had undergone amyloid PET brain scans, which show whether plaques are present. In a large U.S. exercise trial cohort of 317 people, the ratio Aβ1-42/Aβ1-40 measured with PAβ V2.0 aligned more closely with PET scan results than the old method and the immunoassay did. Statistically, the new test achieved a higher ability to distinguish people with abnormal PET scans from those without them, even when the researchers used as little as 100 microliters of plasma—about two drops of blood.
Seeing Changes Before Brain Scans Turn Positive
Interestingly, the improved blood test sometimes flagged individuals as having low Aβ1-42/Aβ1-40 even when their brain scans were still read as normal. The researchers interpret this as a sign that soluble amyloid-beta changes in blood may appear earlier in the disease process than the large, insoluble plaques detectable on PET images. This pattern mirrors previous findings from spinal fluid studies, suggesting that blood-based measures could offer a convenient early warning signal. Adding common clinical factors such as age, sex, and APOE genetic status brought only minor gains, implying that the assay itself already carries strong predictive power.
What This Could Mean for Patients and Trials
For non-specialists, the key message is that the scientists have turned a demanding, laboratory-heavy amyloid test into a faster, cheaper, and more accessible tool without sacrificing accuracy. The new assay uses small blood samples, works on a mass spectrometer model already found in many hospital labs, and costs an estimated $20 in reagents per test. This opens the door to using a simple blood draw to screen people for early brain changes, guide enrollment and monitoring in drug trials, and potentially support diagnosis and prognosis in routine care. While further comparisons with other blood tests are still needed, this streamlined method brings the promise of reliable, early Alzheimer’s detection much closer to everyday clinical reality.
Citation: Chen, Y., Zeng, X., Olvera-Rojas, M. et al. Streamlined resource-efficient plasma amyloid-beta mass spectrometry assay has improved biomarker performance in preclinical Alzheimer’s disease. Nat Commun 17, 1673 (2026). https://doi.org/10.1038/s41467-026-68372-w
Keywords: Alzheimer’s blood test, amyloid-beta, mass spectrometry, early detection, brain amyloid PET