Clear Sky Science · en
INCENP and CDCA8 predict neoadjuvant chemotherapy response and outcomes in esophageal squamous cell carcinoma
Why this research matters for cancer patients
Many people with cancers of the esophagus, breast, or lung receive strong chemotherapy before surgery to shrink their tumors. Yet only about half of these patients truly benefit; for the rest, the drugs bring side effects without stopping the cancer. This study asks a simple but crucial question: can we predict in advance who will respond to chemotherapy, and can we make the treatment work better for those who currently do not?
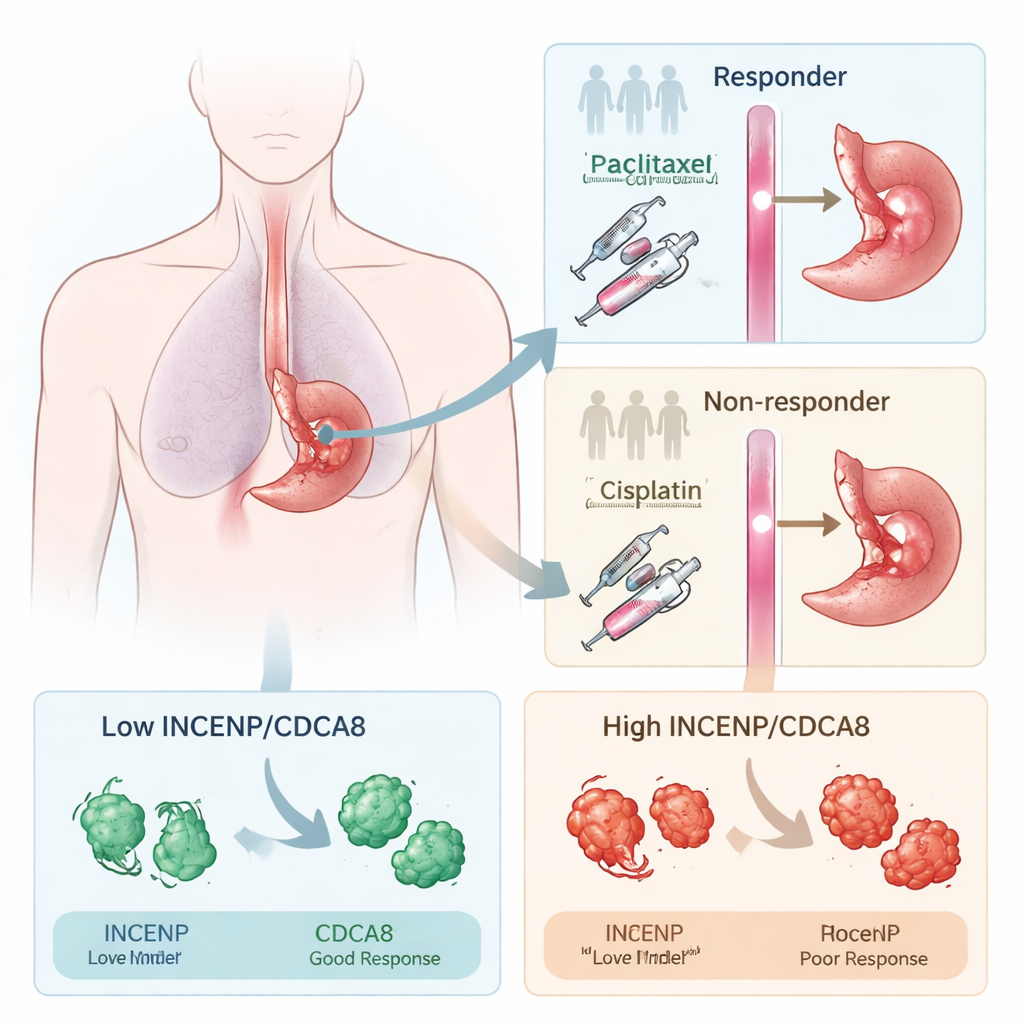
Seeing who responds to treatment
The researchers focused on a standard pre-surgery regimen that combines the drugs paclitaxel and cisplatin, used widely across several cancers. Working first with patients who had esophageal squamous cell carcinoma, they compared tumor samples from people whose cancers shrank strongly after this therapy (responders) with samples from those whose tumors barely budged (non-responders). By surveying which genes were turned up or down, they found that many differences involved how cells divide, a process chemotherapy is designed to disrupt. Two proteins that help control chromosome separation during cell division, INCENP and CDCA8, repeatedly stood out as being much higher in tumors that resisted treatment.
Two proteins that flag likely resistance
To test whether INCENP and CDCA8 were merely bystanders or active drivers of resistance, the team altered their levels in esophageal cancer cells grown in the lab. When they reduced either protein, the cancer cells became far more sensitive to paclitaxel and cisplatin, forming fewer colonies and showing much lower drug doses needed to kill them. When they forced cells to make extra INCENP or CDCA8, the opposite happened: the drugs became less effective. In mice implanted with human esophageal tumors, knocking down these proteins made chemotherapy shrink tumors more strongly. Patients whose tumors contained high levels of INCENP and CDCA8 before treatment were also more likely to have disease progression and shorter survival, reinforcing their value as warning signals.
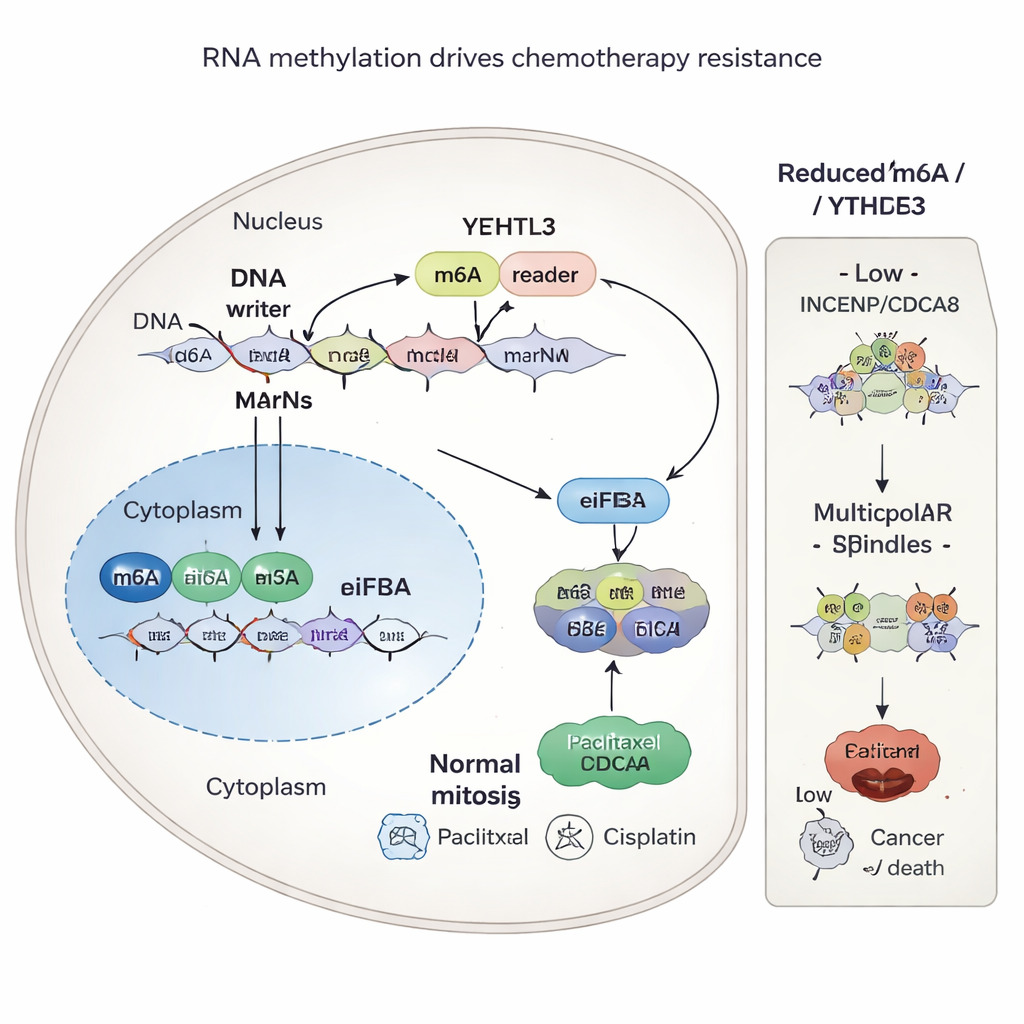
A chemical tag on RNA that fuels resistance
The study then asked why INCENP and CDCA8 are so abundant in resistant tumors. The answer traced back to a subtle chemical mark on RNA called m6A, which acts like a tuning knob for how efficiently genetic messages are read into protein. An enzyme called METTL3 adds these m6A marks to specific spots on the RNA blueprints for INCENP and CDCA8. Another protein, YTHDF3, recognizes these marks and helps recruit the cell’s protein-making machinery, boosting production of the two proteins. When the scientists reduced METTL3 or YTHDF3, or blocked YTHDF3 with a small molecule called Ebselen, the m6A marks were lost or ignored, INCENP and CDCA8 protein levels fell, and cancer cells became more vulnerable to chemotherapy.
Disrupting cell division to help chemotherapy
INCENP and CDCA8 are part of a larger machine called the chromosomal passenger complex that ensures chromosomes are evenly divided between daughter cells. In resistant cells, high levels of these proteins help maintain neat, two-poled (bipolar) spindles during cell division, allowing the cancer to survive the damage caused by paclitaxel and cisplatin. When INCENP and CDCA8 are suppressed, cells often form faulty, multi-poled spindles. This chaos in chromosome segregation pushes cancer cells toward death, amplifying the killing power of chemotherapy. Crucially, the ability of INCENP and CDCA8 to protect cells depended on those specific m6A sites in their RNA; mutating these sites removed the resistance.
Beyond one cancer type
Because paclitaxel plus cisplatin is also used for lung and breast cancers, the team examined biopsy samples from patients with these tumors as well. In all three cancer types, people whose tumors had low levels of INCENP, CDCA8, and YTHDF3 before therapy were far more likely to respond well to treatment. Statistical analyses showed that combining these three markers gave particularly good power to distinguish likely responders from non-responders, suggesting a practical test panel that could be used across multiple cancers.
What this means for patients and doctors
In plain terms, this work identifies a molecular “fingerprint” that helps explain why some tumors shrug off powerful chemotherapy. High levels of INCENP and CDCA8, driven by an m6A-based control system involving METTL3 and YTHDF3, keep cancer cells dividing cleanly even under drug attack. Measuring these proteins in a biopsy could help doctors predict whether paclitaxel plus cisplatin is likely to help a given patient, sparing others from ineffective treatment. At the same time, drugs that block this pathway could be combined with standard chemotherapy to tip resistant tumors into fatal division errors, potentially improving survival for people with several major cancers.
Citation: Wang, X., Wang, T., Wang, K. et al. INCENP and CDCA8 predict neoadjuvant chemotherapy response and outcomes in esophageal squamous cell carcinoma. Nat Commun 17, 1672 (2026). https://doi.org/10.1038/s41467-026-68371-x
Keywords: neoadjuvant chemotherapy, esophageal squamous cell carcinoma, chemotherapy resistance, INCENP and CDCA8 biomarkers, RNA m6A methylation