Clear Sky Science · en
Loss of Fsr quorum sensing promotes biofilm formation and worsens outcomes in enterococcal infective endocarditis
Silent Saboteurs on the Heart’s Valves
Infective endocarditis is a life‑threatening infection of the heart’s valves, and it is becoming more common worldwide. This study focuses on a major culprit, the gut bacterium Enterococcus faecalis, and asks why some infections are especially hard to treat. By uncovering how these bacteria talk to each other and build protective biofilms on heart valves, the researchers reveal why certain strains trigger more severe disease and resist antibiotics, and why one bacterial communication system, called Fsr, turns out to be a surprising brake rather than an accelerator of damage.
How Bacteria Build Fortresses in the Heart
Heart valves normally withstand some of the fastest blood flow in the body, but if the valve surface is damaged, platelets and clotting proteins can form a tiny clot, or vegetation. This clot becomes a landing pad for bacteria in the bloodstream. Once attached, the bacteria multiply and weave themselves into a biofilm—a slimy, structured community that shields them from immune cells and drugs. The team used microfluidic devices to mimic blood flow in the lab, and a rat model to recreate endocarditis in living animals, allowing them to watch how E. faecalis adapts as the infection progresses from a few scattered cells to thick, mature biofilms. 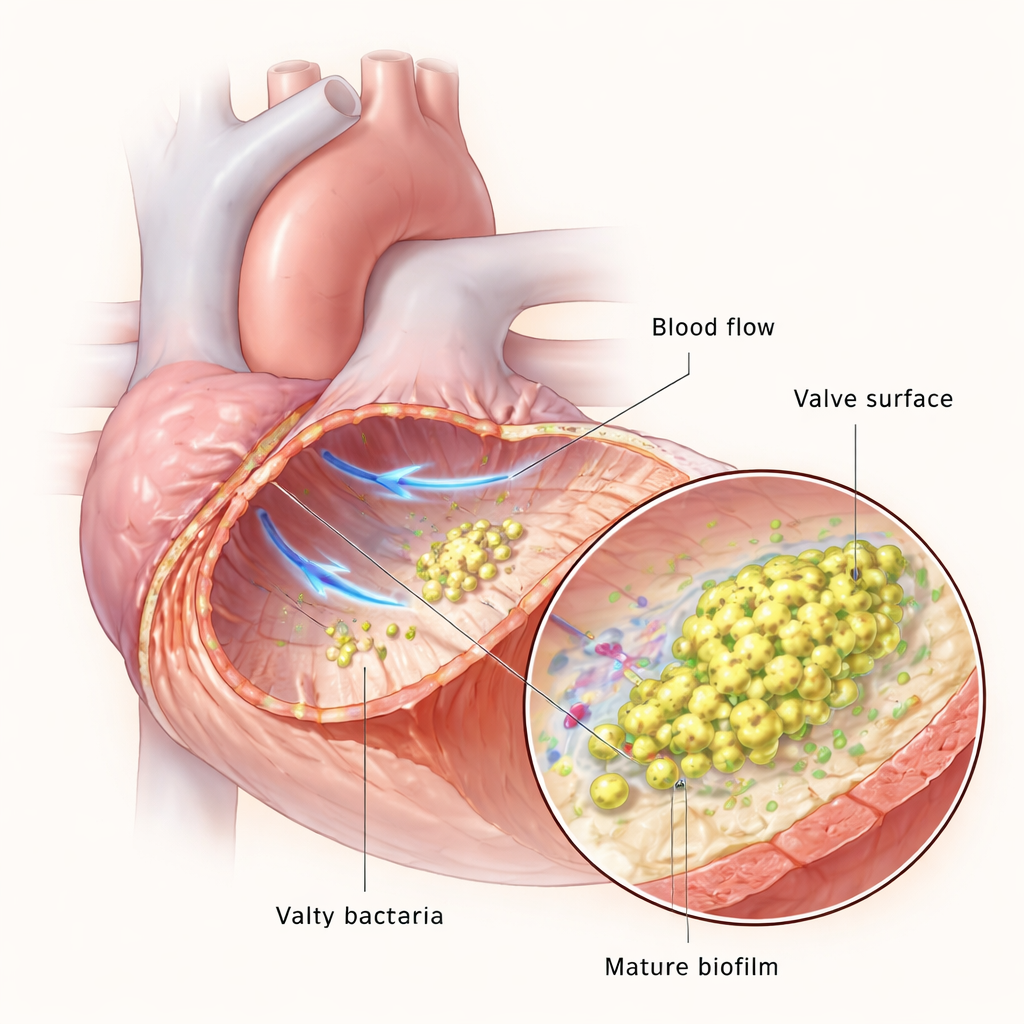
When Fast Blood Flow Turns Off Bacterial “Chatter”
Bacteria often coordinate their behavior using quorum sensing, a chemical “voting” system that switches genes on or off once enough cells are present. Fsr is the quorum sensing system used by E. faecalis. Surprisingly, the researchers found that strong, moving fluid actually washes away the signaling molecules needed for Fsr to turn on. In early infection, when bacteria are stuck on the exposed surface of the vegetation and feel the full force of blood flow, Fsr is largely silent. Only later, when microcolonies grow larger and become buried deeper inside the vegetation—shielded from flow—does Fsr activate. This means that in the real heart, quorum sensing is governed not just by how many bacteria are present, but also by where they sit within the three‑dimensional structure.
Turning Off Fsr Lets Biofilms Overgrow
To test what Fsr actually does for the infection, the team compared normal bacteria with mutants lacking the entire Fsr system. Early on, both versions colonized the valves equally well. But by three days, animals infected with Fsr‑deficient strains had larger vegetations and many more bacteria packed into them. Detailed imaging showed that their biofilms covered more of the clot’s surface and their microcolonies were bigger. Losing Fsr also shut down two key secreted enzymes, GelE and SprE, which normally help shape the biofilm and may limit overgrowth. Animals infected with mutants missing just these enzymes also developed bulkier biofilms, although not as extreme as in Fsr‑null strains, suggesting other Fsr‑controlled factors are also at work. 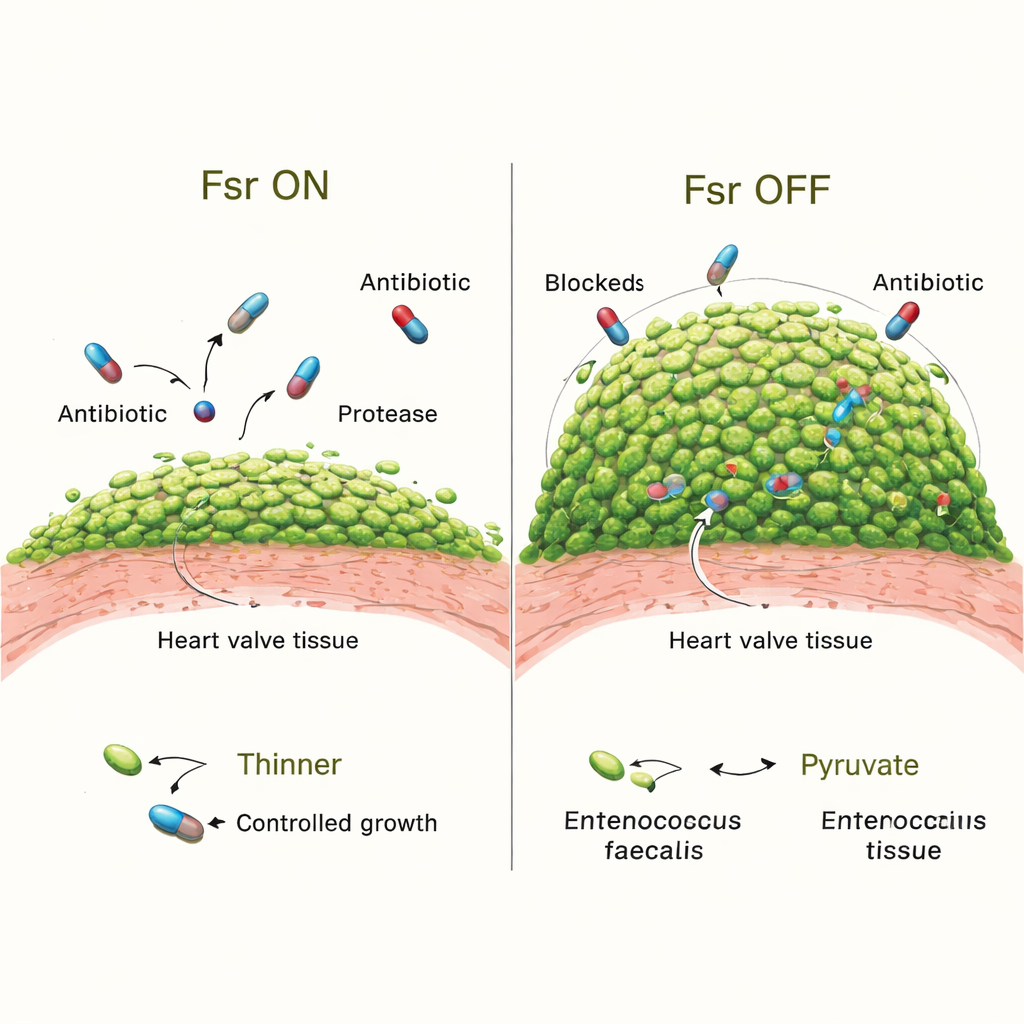
Metabolic Rewiring and a Stronger Grip on Antibiotics
Gene‑expression profiling of bacteria inside infected valves revealed that Fsr does more than control a few enzymes. Without Fsr, hundreds of genes changed their activity, especially those involved in sugar uptake and energy production. One pair of genes, lrgA and lrgB, stood out: they were strongly switched on in Fsr‑deficient bacteria and helped the cells better exploit pyruvate, a small energy‑rich molecule present in blood. When the researchers deleted lrgAB along with Fsr, the excess biofilm growth vanished, pointing to a key role for this metabolic pathway in powering the overgrown infection. Notably, Fsr‑deficient biofilms were also tougher to treat. In rats given the antibiotic gentamicin, infections with normal bacteria shrank and platelet counts—a marker of disease severity—improved. In contrast, Fsr‑deficient infections barely responded, even though the bacteria were no more drug‑resistant in standard lab tests.
A Human‑Specific Twist in Inflammation
The study also uncovered a potential way these bacteria may stir up inflammation in people. The GelE enzyme can slice the human precursor of the immune signal IL‑1β into an active form that triggers inflammatory responses. Intriguingly, GelE cut rat IL‑1β differently, breaking it down rather than activating it, and the precise “activation” cut was missing from rat and mouse proteins. This species‑specific effect suggests that in humans, GelE may help focus inflammation right at the biofilm’s edge, possibly worsening tissue damage during endocarditis.
From Bench to Bedside: Why Fsr Loss Matters for Patients
To see whether these experimental findings mattered in real life, the researchers analyzed E. faecalis isolates from 81 patients with infective endocarditis in Switzerland and the United States. Nearly half of the strains naturally lacked the Fsr system. Patients infected with these Fsr‑negative strains had longer‑lasting bacteremia—more days with bacteria detectable in their blood—and were more likely to fall into a high‑severity category that included intensive care, prolonged hospital stays, major heart procedures, or death. Other virulence genes could not explain this pattern, pointing directly to Fsr loss as a key marker of more aggressive disease.
Why This Matters for Future Treatments
Together, these findings overturn the simple idea that blocking bacterial communication will always weaken infections. In E. faecalis endocarditis, the Fsr system actually restrains biofilm expansion and makes bacteria more vulnerable to antibiotics. When Fsr is missing, bacteria grow denser biofilms, tap into blood nutrients more efficiently, and help drive stubborn, prolonged infections. For patients, this means that strains lacking Fsr may predict harder‑to‑treat disease, and that therapies which blindly shut down quorum sensing could backfire in this context. Instead, carefully targeting metabolic pathways like pyruvate use, or monitoring Fsr and GelE status as prognostic markers, may offer better ways to manage these dangerous heart infections.
Citation: Antypas, H., Schmidtchen, V., Staiger, W.I. et al. Loss of Fsr quorum sensing promotes biofilm formation and worsens outcomes in enterococcal infective endocarditis. Nat Commun 17, 1668 (2026). https://doi.org/10.1038/s41467-026-68366-8
Keywords: infective endocarditis, bacterial biofilms, quorum sensing, Enterococcus faecalis, antibiotic tolerance