Clear Sky Science · en
Interfacial evaporation-induced localized multi-field coupling enables efficient co-recovery of freshwater and nitrates
Turning Pollution into a Resource
Nitrate is a double‑edged sword. It is an essential ingredient in fertilizers and industry, but when it leaks into rivers, lakes, and groundwater it pollutes drinking water and fuels algal blooms that threaten food and water security. This study presents a sunlight‑powered device that can both clean water and salvage valuable nitrate at the same time, offering a way to treat contamination while also recycling a key ingredient for agriculture and energy.
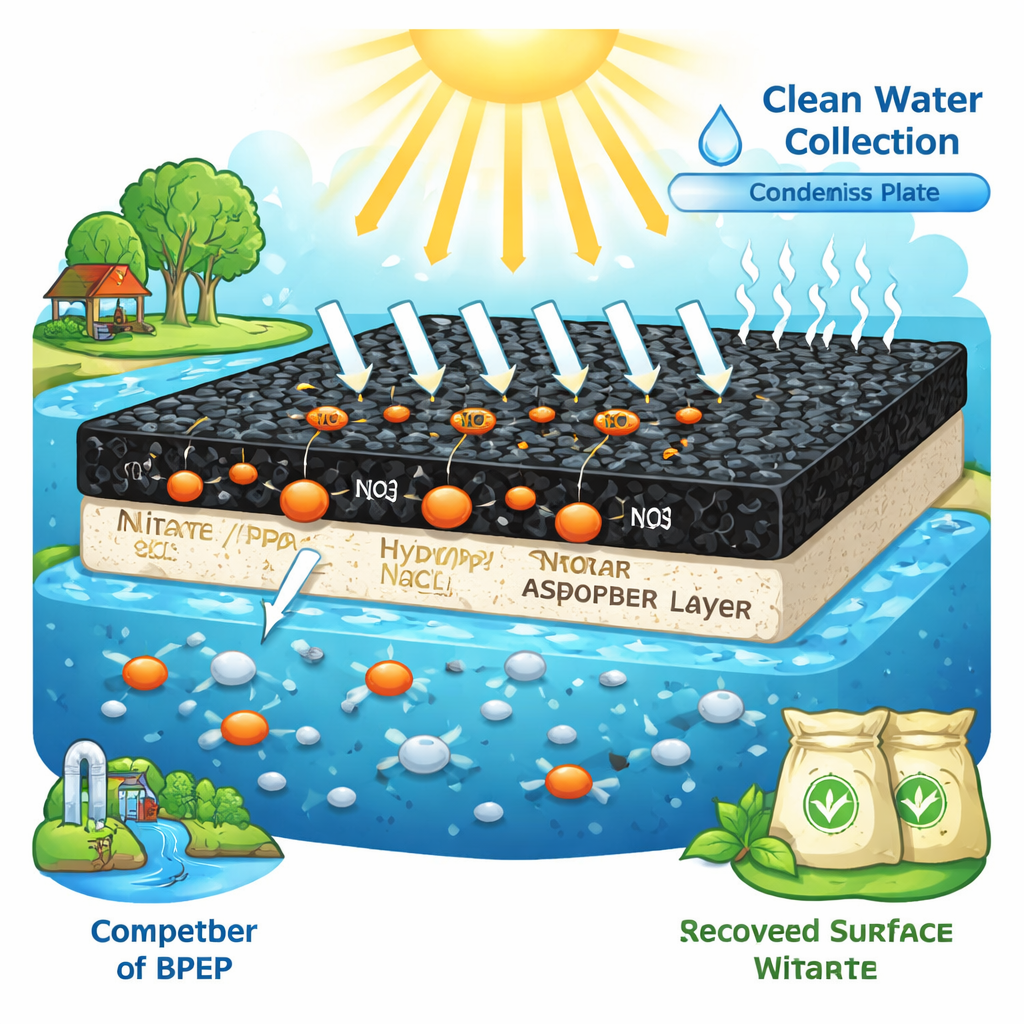
Why Nitrate in Water Matters
Across the globe, shortages of freshwater and reliable food supplies are closely linked. Nitrate, widely used in fertilizers and chemicals, often washes off fields and industrial sites into surface waters. At low levels it is hard to capture, yet it can still harm ecosystems and human health. Making new nitrate usually relies on energy‑hungry, high‑temperature industrial processes that emit greenhouse gases. If we could instead pull nitrate back out of polluted water and reuse it, we would tackle water pollution and reduce the energy cost of producing fertilizers and chemicals.
Using Sunlight to Drive Evaporation
The researchers built a bio‑inspired photothermal evaporation platform, or BPEP, that sits on the surface of water. Its heart is a thin hydrogel made from bacterial cellulose coated with polypyrrole, a black, light‑absorbing polymer. When sunlight shines on this layer, it heats up strongly while the underlying water stays relatively cool, because the device is insulated from below. This concentrated heating at the water’s surface causes rapid evaporation, producing clean vapor that can be condensed into freshwater. At the same time, the dark coating attracts nitrate ions from the water, acting like a sponge that prefers nitrate over many other dissolved salts.
How the Device Boosts Nitrate Capture
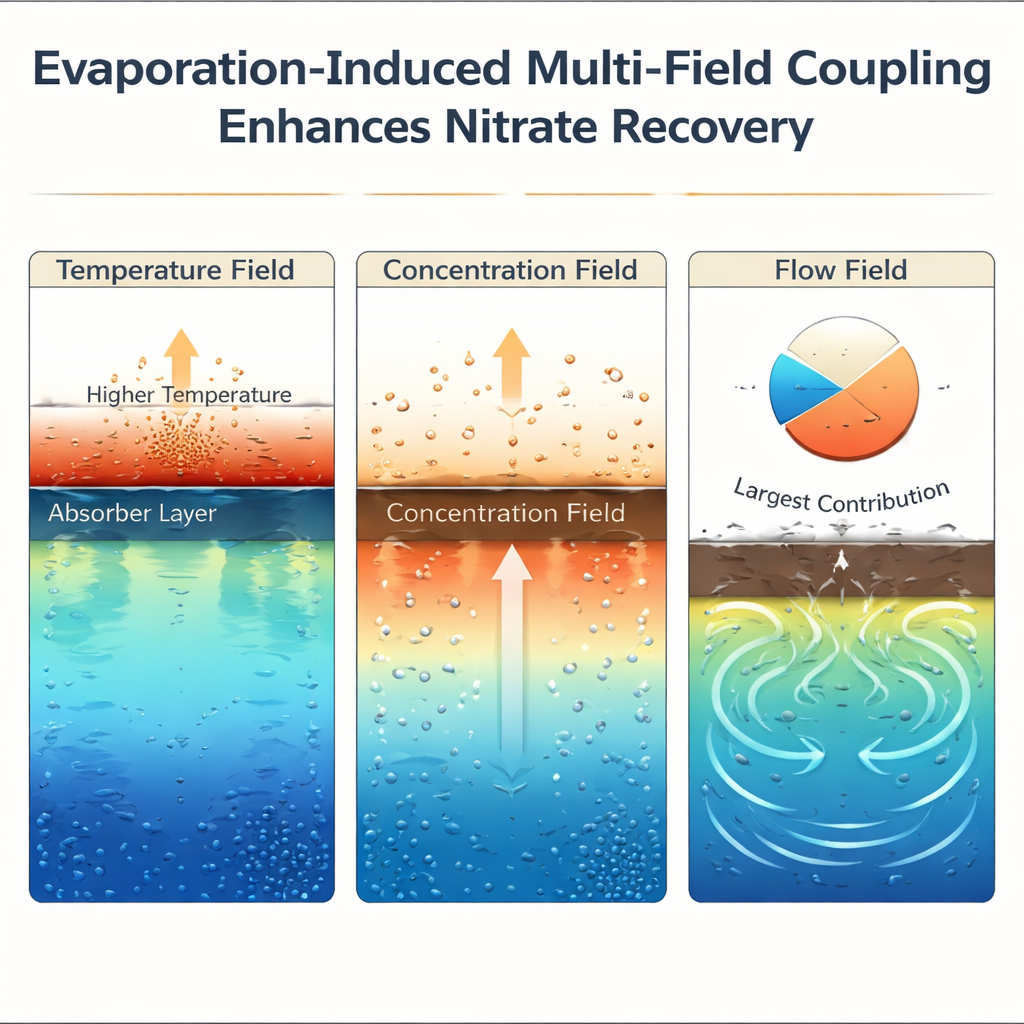
Evaporation does more than just make steam. As water molecules escape, nitrate and other ions are left behind and become more concentrated near the hot surface. The temperature, concentration, and movement of the liquid all change locally, and these three “fields” reinforce one another. The warmer top layer slightly improves how readily nitrate sticks to the coating, the higher local nitrate concentration favors adsorption, and the constant flow created by evaporation transports ions quickly to the active sites. Simulations and experiments show that this flow effect is the main driver, responsible for roughly three‑quarters of the improvement in nitrate capture compared with a still, unheated system.
Performance with Real Water
In the laboratory under standard sunlight, the BPEP evaporated water much faster than plain water and captured nitrate at a surface‑based capacity several times higher than in the dark. The material maintained most of its performance over repeated use, and common competing ions in natural waters had only a modest impact at typical salinities. Outdoor tests with urban river water showed that the device could lower nitrate levels from moderately polluted values down to near‑safe limits in a single day, while simultaneously providing a steady output of clean water. The same platform can also desalinate seawater and purify industrial wastewater, removing more than 99 percent of key contamination indicators.
From Waste to Fertilizer and Fuel
Captured nitrate is not discarded. It can be rinsed out of the device and converted into useful products. The authors demonstrate that the recovered nitrate can be broken down biologically into harmless nitrogen gas, or transformed electrochemically into ammonia, a major fertilizer and energy carrier. When plants were irrigated with ammonia made from the recovered nitrate, they grew taller than plants given only pure water, confirming its practical value. By concentrating nitrate before these conversion steps, the solar device makes downstream chemical and biological processes more efficient.
A Sun‑Powered Tool for Sustainable Water and Food
In simple terms, this work shows that it is possible to build a floating, sunlight‑driven “still” that not only turns dirty water into drinkable water, but also harvests dissolved nitrate that would otherwise be wasted or cause pollution. By boosting nitrate capture through smart control of heat, flow, and concentration at the water’s surface, the system turns a common contaminant back into a resource. If scaled and integrated with existing treatment plants and fertilizer production, this approach could help communities move toward more sustainable water use and more efficient fertilizer cycles.
Citation: Yu, Z., Shi, L., Ning, R. et al. Interfacial evaporation-induced localized multi-field coupling enables efficient co-recovery of freshwater and nitrates. Nat Commun 17, 1667 (2026). https://doi.org/10.1038/s41467-026-68365-9
Keywords: nitrate recovery, solar water purification, photothermal evaporation, water and fertilizer recycling, sustainable agriculture