Clear Sky Science · en
Engineered VPg saRNA achieves cap-independent, low-immunogenic and precise encoding of therapeutic proteins in vivo
Rewriting the Rules for RNA Medicines
Vaccines based on messenger RNA (mRNA) helped turn the tide against COVID-19, but using similar molecules to replace or supplement missing proteins in chronic disease is much harder. The body tends to destroy foreign RNA quickly and mount strong immune reactions, and many existing self-amplifying RNA designs make errors as they copy themselves. This study describes a new kind of self-amplifying RNA—built from parts of human norovirus—that is engineered to be long-lasting, less inflammatory, and unusually accurate, opening the door to more precise and durable RNA medicines.
A Smarter Self-Copying RNA
Conventional mRNA medicines carry a single "recipe" for a protein and are read once by the cell before they are broken down. Self-amplifying RNA (saRNA) includes extra viral machinery that lets the RNA copy itself inside cells, greatly boosting output from a small dose. The best-studied saRNA systems come from alphaviruses, but they are bulky, trigger strong antiviral defenses, and copy their sequences with relatively low accuracy. The authors instead turned to human norovirus, a common cause of stomach flu. They isolated a compact section of norovirus genetic material and rebuilt it as a carrier for therapeutic genes. At its core is VPg, a small viral protein that is chemically attached to the front end of the RNA. VPg replaces the usual "cap" structure found on human mRNAs and allows the RNA to be translated by a different set of cell factors that are less sensitive to certain stress signals. 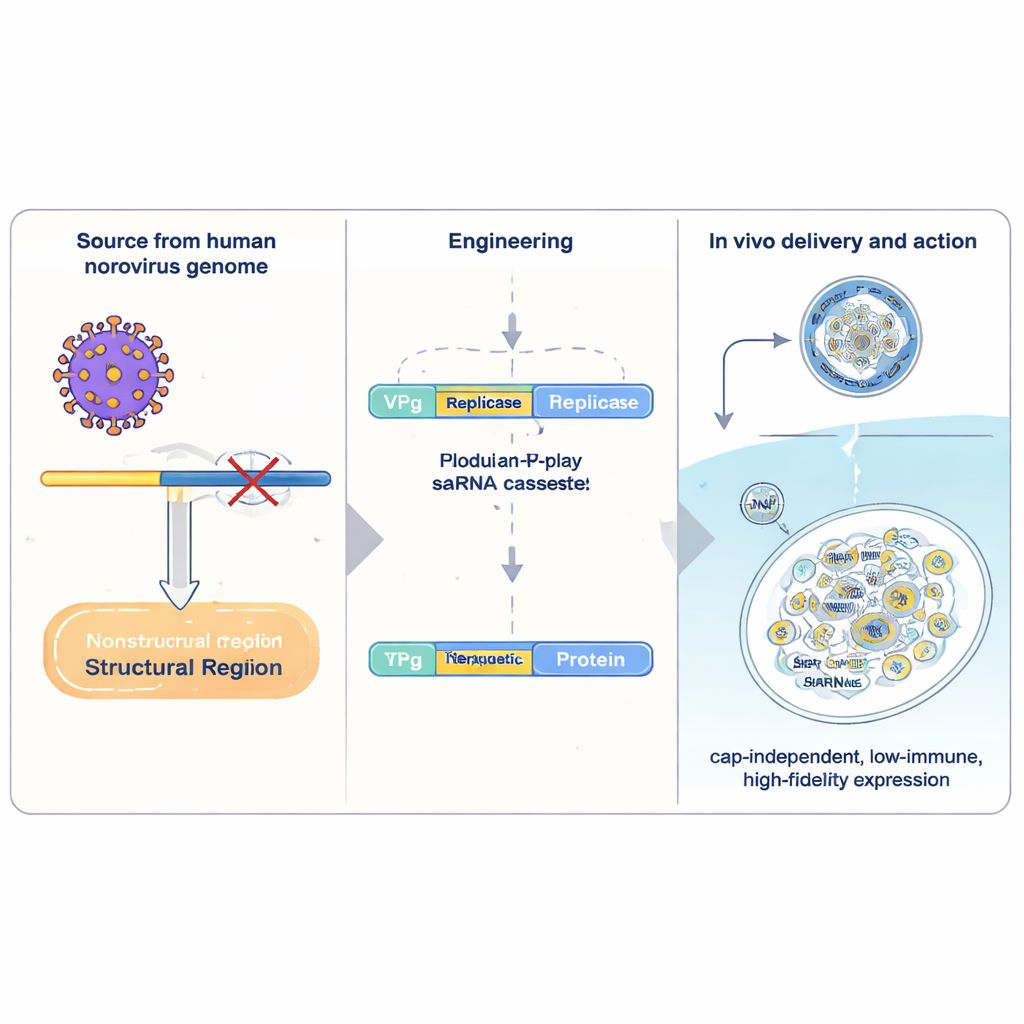
High Accuracy Without Stirring Up Immunity
For protein therapies, accuracy matters: even small copying errors can change a drug’s behavior or create toxic byproducts. The team showed that when norovirus VPg binds to the virus’s RNA-copying enzyme (a polymerase), it slows the rate at which incorrect building blocks are added, sharply reducing the error rate. They pinpointed specific amino acids in VPg that contact a key control region of the polymerase and demonstrated that disrupting this interface makes copying less accurate. At the same time, classic saRNA designs create large amounts of double-stranded RNA, a molecular alarm bell that activates antiviral defenses and shuts down protein production. By redesigning the polymerase to favor one particular VPg-linked starting form and by rearranging the genome so that only the therapeutic strand is efficiently produced, the authors greatly reduced double-stranded RNA formation. They further tuned a tail region of the RNA so that VPg attaches at the correct site, preventing short, unintended fragments that might translate into stray peptides.
Working Where Standard mRNA Fails
The new VPg-based saRNA was then tested in demanding disease settings where ordinary capped mRNA struggles. In cancer-associated cachexia—a wasting syndrome in which muscles and fat melt away—key translation factors that recognize the standard mRNA cap are switched off, making cap-dependent mRNAs inefficient. Because VPg uses an alternate entry point into the cell’s translation machinery, it continued to drive production of the appetite and metabolism hormone ghrelin in a mouse model of pancreatic cancer. Monthly low-dose injections helped preserve muscle and fat, improved food intake and metabolism, and modestly extended survival, without obvious toxicity or signs of chronic immune activation.
Precision Cancer Attack and Calmer Autoimmunity
To test whether high-fidelity copying really matters, the researchers loaded the VPg vector with a complex oncolytic construct, GSDMDENG, designed to punch holes in cancer-cell mitochondria and stimulate tumor-specific immune responses. When the same construct was placed on a conventional alphavirus saRNA, mutations accumulated over repeated rounds of copying and weakened its selective cancer-killing and immune-priming functions. In contrast, the VPg system preserved the intended sequence, produced strong tumor cell killing, and triggered durable T cell responses that slowed or prevented pancreatic cancer in mouse models. Finally, they applied VPg saRNA to a different challenge: chronic graft-versus-host disease, where immune cells attack the lungs after bone marrow transplantation. A VPg saRNA encoding activated protein C reduced harmful antibody formation, prevented lung scarring, and improved breathing in mice, while a comparable alphavirus-based construct provoked stronger innate immunity and worsened outcomes.
Why This Matters for Future RNA Drugs
Taken together, the work outlines a compact, "plug-and-play" RNA platform that couples three desirable traits: it copies itself accurately, it is designed to minimize immune alarms, and it bypasses some of the bottlenecks that restrict standard mRNA translation. Because the therapeutic gene is simply swapped into a fixed norovirus-based backbone, many different proteins—from hormones to clotting regulators to oncolytic agents—can be tested on the same chassis. While much work remains before use in people, this VPg saRNA approach suggests a path toward RNA medicines that act more like stable protein infusions: low-dose, longer-lasting, and tailored to operate even in diseased tissues where ordinary mRNA messages cannot be reliably read. 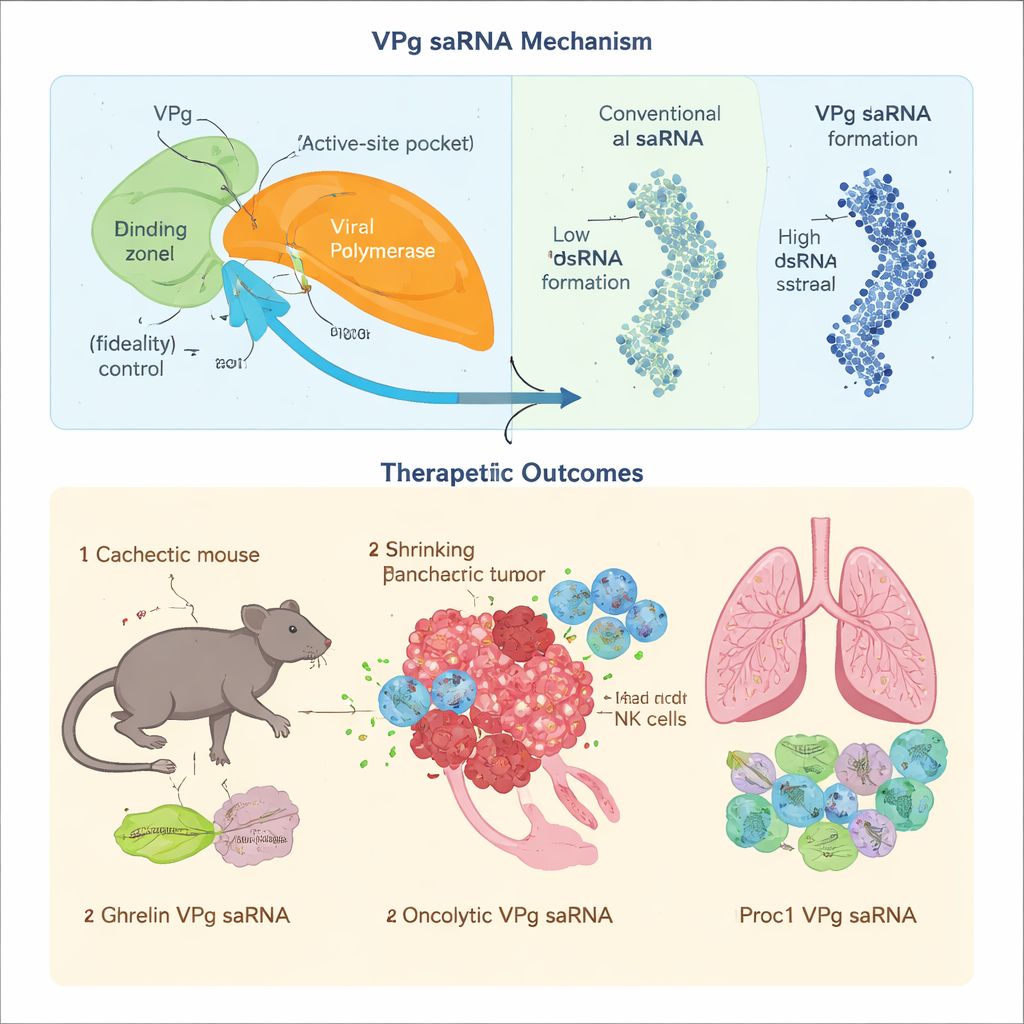
Citation: Feng, Z., Chu, L., Li, Q. et al. Engineered VPg saRNA achieves cap-independent, low-immunogenic and precise encoding of therapeutic proteins in vivo. Nat Commun 17, 1666 (2026). https://doi.org/10.1038/s41467-026-68364-w
Keywords: self-amplifying RNA, mRNA therapeutics, norovirus VPg, cancer cachexia, graft-versus-host disease