Clear Sky Science · en
Reversible bismuth reduction-driven dark photoelectrochemistry
Why light-off chemistry matters
Solar-powered sensors and catalysts usually work better when the lights are on. This study turns that idea on its head by creating a system where the signal actually grows stronger in the dark. The researchers show how a special bismuth-based material can store the effect of light and then release it later, allowing it to tell apart very similar chemical molecules with unusual precision. This counterintuitive "dark-enhanced" behavior could inspire new ways to build chemical sensors, batteries, and energy devices that keep working after the lights go out.
A new twist on light-powered sensors
Most photoelectrochemical devices rely on semiconductors that convert light into electric signals or drive chemical reactions. In today’s designs, shining light on an electrode generally boosts the flow of electrical current as charges move across the solid–liquid boundary. Different molecules in the solution are then recognized mainly by how big a current they cause. That approach often struggles with selectivity: molecules that behave similarly, such as many common biological or environmental chemicals, can be hard to tell apart. Traditional fixes like adding enzymes or complex coatings improve selectivity but add cost and can be unstable.
Turning the usual behavior upside down
The team focused on a material called bismuth oxybromide (BiOBr), shaped into tiny nanosheets and used as a light-sensitive cathode. When they tested it in water containing dissolved oxygen, they saw something surprising: the cathode produced a larger current in the dark than under illumination. In other words, turning the light on reduced the current instead of increasing it. This “reverse photocurrent” only appeared under normal air conditions; it disappeared when the solution was saturated with oxygen or stripped of it with nitrogen. Changes in the electrode’s color during testing hinted that bismuth atoms near the surface were cycling between different chemical states in step with the light–dark switches. 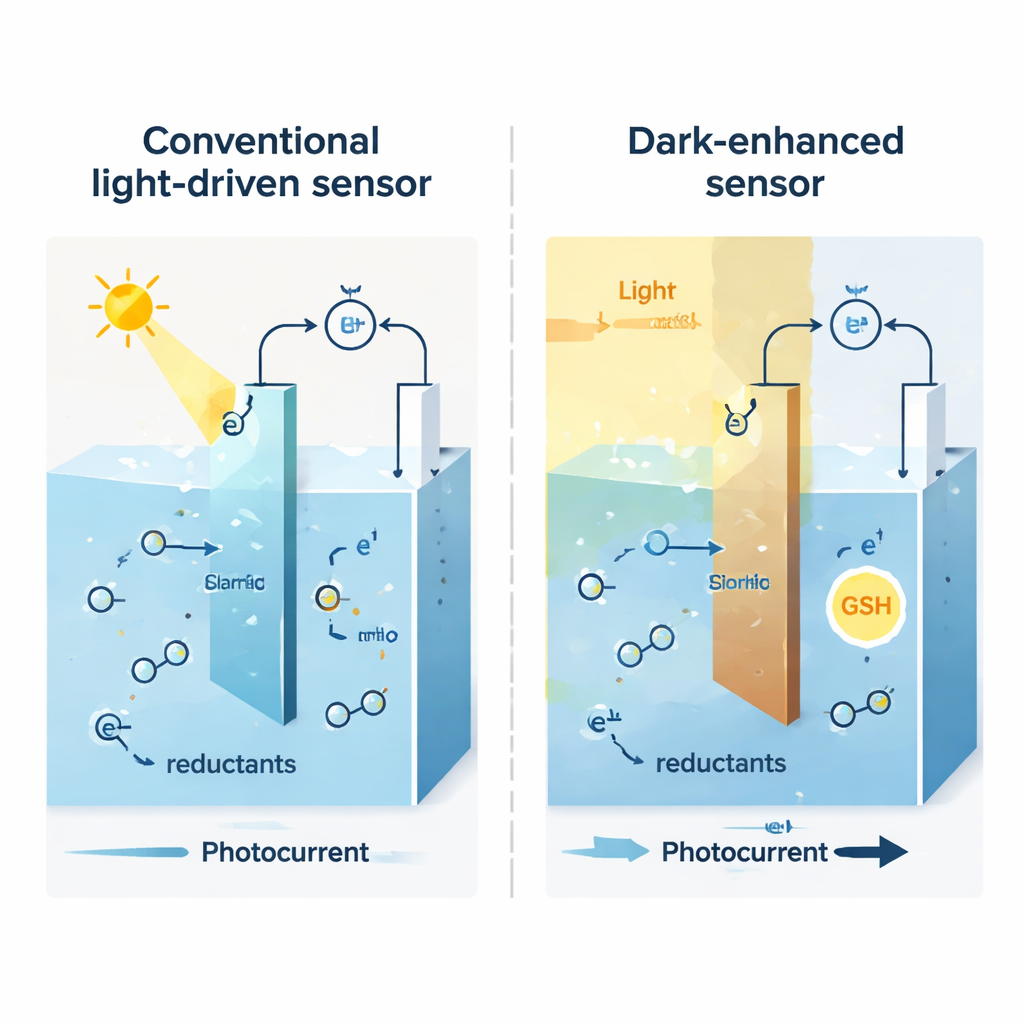
How the material stores and releases light’s effect
Detailed measurements of the electrode’s structure and electronic behavior revealed what was happening. Under illumination, BiOBr partially reduces some of its bismuth ions, creating a slightly lower-valent form that traps extra electrons and darkens the surface. These trapped electrons passivate, or “turn off,” the usual reaction where oxygen is reduced at the surface, so the current goes down when the light is on. When the light is switched off, dissolved oxygen in the water re-oxidizes these bismuth sites, restoring their original state and reactivating oxygen reduction. As a result, the current jumps up in the dark. This reversible bismuth redox cycle effectively builds a new energy level into the material that only exists after illumination, allowing the electrode’s chemistry to be different in light versus dark.
Selective recognition of a key biological molecule
The researchers then asked whether this unusual dark behavior could be used to distinguish similar reducing molecules. They compared many candidates, including the antioxidant ascorbic acid and the tripeptide glutathione (GSH), an important defender against oxidative stress in living cells. Only GSH dramatically amplified the reverse, dark-enhanced current. Spectroscopic tests showed that GSH binds directly to bismuth atoms, forming Bi–S bonds and enabling the bismuth to cycle between several oxidation states more easily. During illumination, the BiOBr surface effectively acts like a tiny “pseudo-anode,” pulling electrons from GSH and creating more reduced bismuth sites. When the light is turned off, these extra sites are rapidly re-oxidized by oxygen while GSH and its oxidized form interconvert, greatly boosting the dark current. Ascorbic acid, which does not bind in the same way, cannot trigger this enhanced cycle. 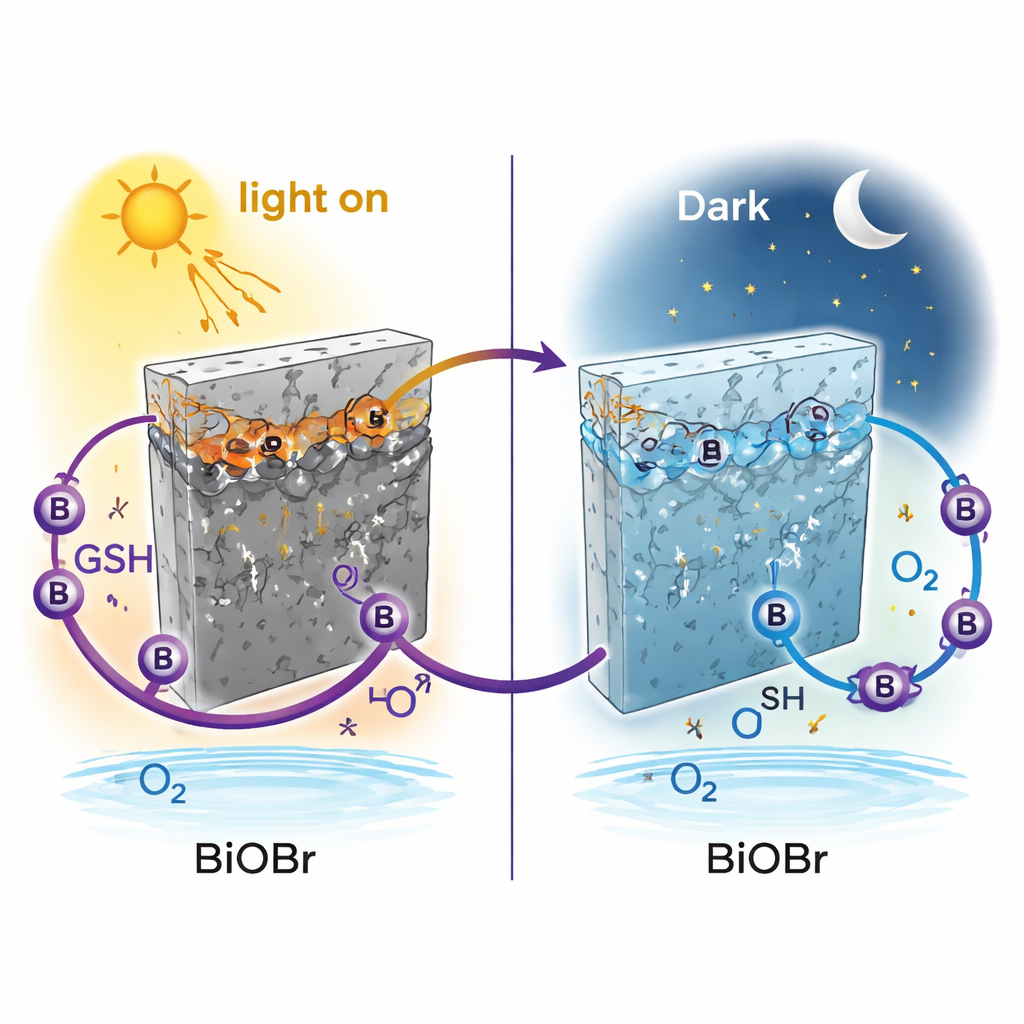
From laboratory curiosity to practical sensor
By harnessing this dark-enhanced effect, the team built a highly selective sensor for glutathione. The device produced a clear, linear change in dark current for a wide range of GSH concentrations, with very low detection limits. It showed strong discrimination against other common biological and thiol-containing molecules and worked well in real samples taken from vegetables such as onions, spinach, and broccoli. Compared with conventional light-boosted sensors, this dark-based approach offered a better detection range, higher sensitivity, and improved selectivity.
What this means for future technologies
To a non-specialist, the key message is that the authors have discovered a way to make a light-activated material whose most useful signal appears when the light is off. By carefully tuning how bismuth atoms in BiOBr gain and lose electrons, and by exploiting a special partnership with glutathione, they created a surface that can “remember” light exposure and then use that memory to distinguish one molecule from many look-alikes. This fresh view of how light, oxygen, and surface chemistry interact at an electrode could guide the design of next-generation sensors and energy devices that are both more selective and more versatile in real-world conditions.
Citation: Qin, Y., Chen, Y., Wan, H. et al. Reversible bismuth reduction-driven dark photoelectrochemistry. Nat Commun 17, 1640 (2026). https://doi.org/10.1038/s41467-026-68359-7
Keywords: photoelectrochemistry, bismuth oxybromide, dark photocurrent, glutathione sensing, electrochemical biosensor