Clear Sky Science · en
Bioadaptive Ni single atoms unlock high rate microbial electrosynthesis of isopropanol from CO2
Turning Waste Gas into Useful Alcohol
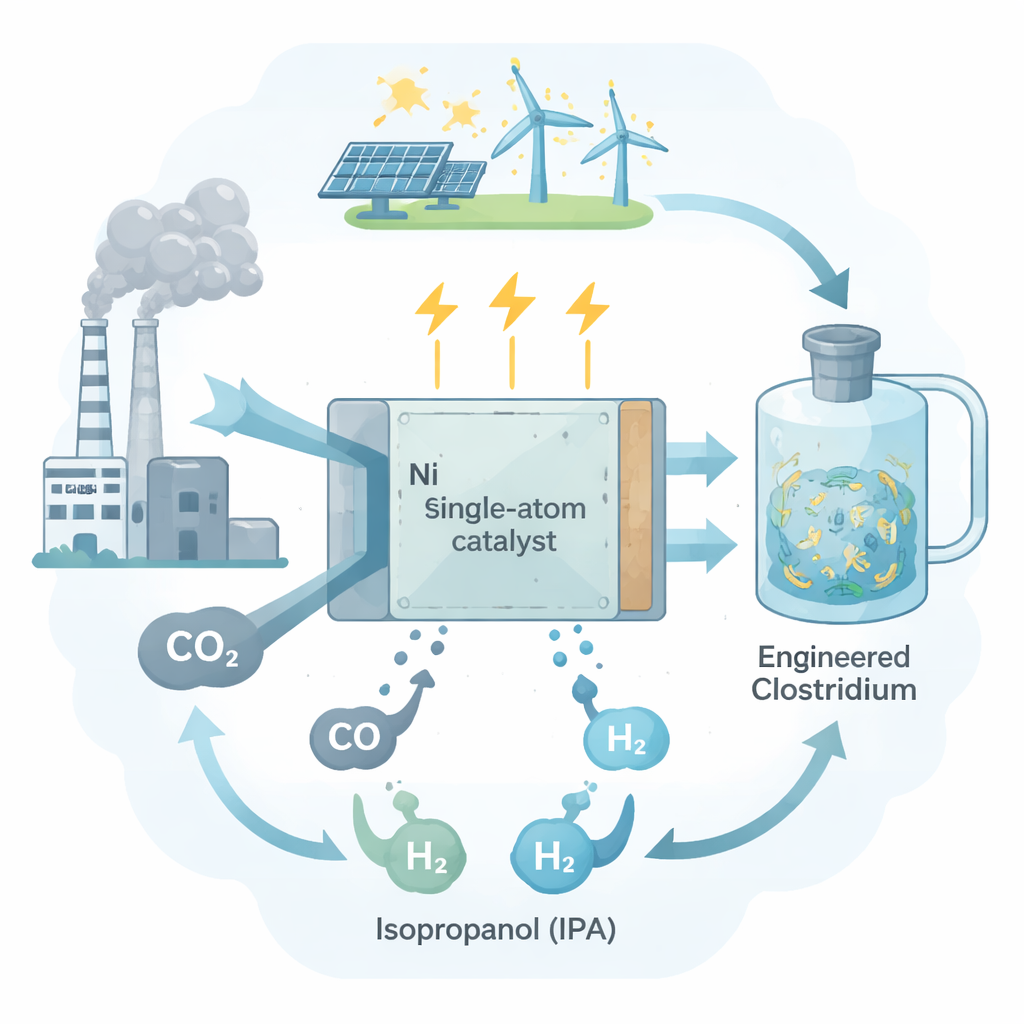
Isopropanol—the familiar ingredient in rubbing alcohol and electronics cleaners—is mostly made today from fossil fuels in energy-hungry factories. This study explores a different path: using electricity and living microbes to turn waste carbon dioxide (CO2) into isopropanol at room temperature. The researchers show how a specially designed nickel-based catalyst can survive in a soup of nutrients and cells, making it possible to link clean electricity, CO2 captured from industry, and engineered bacteria into one continuous process.
Why Isopropanol and CO2 Matter
Isopropanol is a workhorse chemical used in disinfectants, fuel additives, and especially in cleaning semiconductor chips, a market that is booming with the rise of AI and advanced electronics. Global demand is already worth billions of dollars and is projected to grow. Today, almost all isopropanol is made from petroleum-derived propylene or from acetone using high temperatures, high pressures, and fossil hydrogen. These routes emit CO2 and require difficult separations. If CO2 itself could be the starting material, powered by renewable electricity, the same chemical could be produced with a far smaller carbon footprint—and potentially even make use of CO2 that would otherwise be vented to the atmosphere.
Microbes as Tiny Chemical Factories
The team builds on recent advances in “gas fermentation,” where certain microbes eat simple gases such as CO2, carbon monoxide (CO), and hydrogen (H2) and use them to grow and make multi‑carbon products. Here they work with a genetically engineered strain of the bacterium Clostridium ljungdahlii that can produce isopropanol from gas mixtures. Careful fermentation tests revealed that CO plays a crucial role: when the microbes received only H2 and CO2, they barely made any isopropanol and grew poorly. When CO was added, isopropanol levels rose by about 140‑fold, and production of other products like ethanol and acetate also increased strongly. CO not only provides carbon, but also delivers the energy-rich electrons the cells need to power their metabolism, making it a more effective fuel than H2 alone.
The Catalyst Problem in Living Media
To supply CO from CO2 on demand, the system relies on an electrochemical cell—essentially a device that uses electricity to push CO2 to react at an electrode. In simple salt solutions, silver is a well-known catalyst for turning CO2 into CO. But in real microbial growth media, which contain amino acids, vitamins, and many other organic molecules, silver performs poorly: its CO output drops by one to two orders of magnitude. Using advanced spectroscopy, the authors show that on silver surfaces these organic molecules crowd onto the electrode, blocking CO2 from reaching reactive sites. Even when higher voltages are applied and some organics desorb, hydrogen gas formation takes over, wasting electrons and undermining the goal of steady CO production for the microbes.

Nickel Single Atoms That Play Nicely with Biology
The core innovation of this work is a “bioadaptive” catalyst made of isolated nickel atoms anchored in a nitrogen-doped carbon support. This nickel single-atom catalyst keeps its structure as tiny, separated sites rather than large metal particles. In standard electrolytes it already shows excellent efficiency for making CO. Crucially, in the complex microbial medium it maintains almost the same CO selectivity—up to about 92%—and much higher activity than silver. Measurements of the catalyst’s surface vibrations and local atomic environment indicate that, unlike silver, it does not strongly bind the organic components of the growth medium. Computer simulations back this up: typical medium molecules like amino acids and nucleic acid bases stick readily to silver but are thermodynamically disfavored on the nickel single sites. As a result, CO2 can still approach and react at these nickel centers even in the crowded biological environment.
A Working Hybrid System and What It Means
With a reliable CO source in hand, the researchers built a full hybrid reactor that links the nickel electrode to a culture of engineered C. ljungdahlii. Under continuous operation at body-like temperature (37 °C), the system held a stable electrical current and gas composition for four days. During this time, the microbes converted the electrochemically produced CO (and some H2) into a mix of isopropanol, ethanol, and acetate. After accounting for evaporation, the isopropanol production rate reached about 161 milligrams per liter per day at a current density around 10.8 amperes per square meter—competitive with or better than earlier systems that relied only on H2 as the electron source. Importantly, structural checks after long operation showed that the nickel single-atom catalyst remained intact and did not significantly leach metal into the broth.
From Lab Demonstration to Greener Chemicals
In plain terms, this study shows that it is possible to directly feed CO2 and electricity into a system containing living microbes and still run an efficient chemical reaction, as long as the catalyst is designed to tolerate biological complexity. The nickel single-atom catalyst acts like a selective gatekeeper: it keeps its focus on CO2 even while immersed in nutrient-rich media, delivering a steady stream of CO that microbes then upgrade into isopropanol. While engineering challenges remain—such as matching gas production rates to microbial uptake, preventing flooding of gas-diffusion layers, and simplifying product recovery—this work outlines a promising route toward cleaner, electricity-driven manufacture of everyday chemicals from waste CO2.
Citation: Zhou, G., Humphreys, J.R., Cheng, D. et al. Bioadaptive Ni single atoms unlock high rate microbial electrosynthesis of isopropanol from CO2. Nat Commun 17, 1639 (2026). https://doi.org/10.1038/s41467-026-68358-8
Keywords: CO2-to-chemicals, microbial electrosynthesis, single-atom catalysts, isopropanol production, nickel electrocatalyst