Clear Sky Science · en
Operando insights into stability of perovskite-based solar water splitting devices
Turning Sunlight and Water into Clean Fuel
Imagine making clean hydrogen fuel from nothing but sunlight and water, using devices as thin and elegant as modern solar panels. This paper explores a new way to make those devices last much longer in real operating conditions, by cleverly using invisible near‑infrared (NIR) light to gently heat tiny catalysts without harming the light‑absorbing material. The work tackles one of the biggest barriers to practical, low‑cost solar hydrogen: keeping high‑efficiency perovskite devices stable for hundreds of hours in water.
Why Perovskites Excite Energy Researchers
Perovskites are a family of crystalline materials that absorb sunlight extremely well and move electric charges efficiently. In just over a decade, they have rivaled traditional silicon in solar cells and are now being adapted to split water into hydrogen and oxygen. These perovskite-based systems have already passed a key commercialization benchmark, converting more than 10% of incoming sunlight into chemical energy stored in hydrogen. However, unlike conventional solar panels, these devices must also drive relatively slow electrochemical reactions in a liquid. That mismatch in timescales—fast generation of charges vs. slower use of those charges at the catalyst—can cause charges to pile up inside the device, triggering chemical changes that gradually break it down.
A Smart Architecture with Localized Heating
The authors designed a perovskite “photocathode” that sits in water but is sealed against moisture by a conductive epoxy and metal contacts. Physically separated from the light‑absorbing stack is a platinum-on-carbon catalyst, which actually touches the water and produces hydrogen gas. Crucially, this catalyst can be selectively heated by a NIR laser that passes harmlessly through the water and glass. Because the epoxy is a good thermal insulator, the catalyst warms up while the perovskite layer remains cool and protected. Under standard sunlight, the device already delivers very high photocurrent; when NIR light is added, the current and operating voltage both improve, and the perovskite maintains over 90% of its initial performance for 310 hours—far longer than similar systems without this approach. 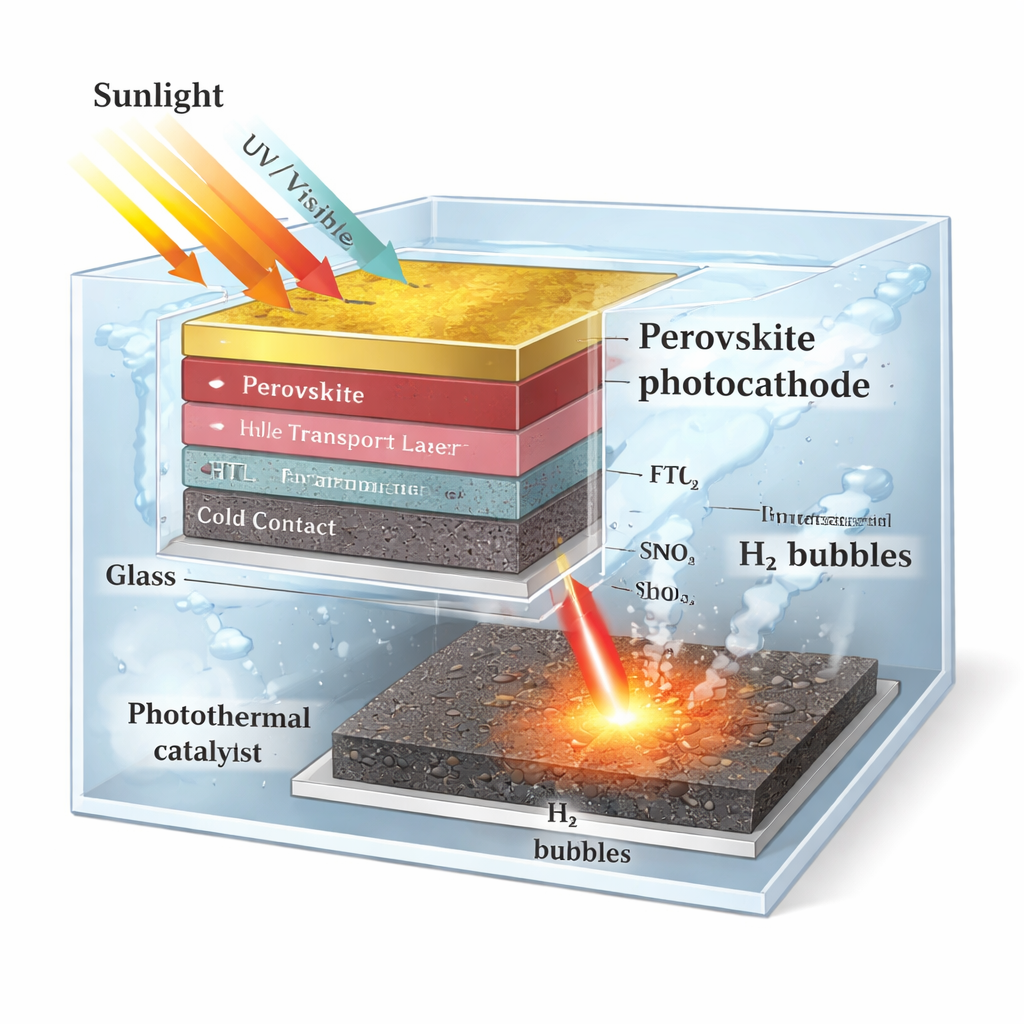
How Warm Catalysts Calm Down the Device
By watching the device as it runs, the team shows that the gently heated catalyst speeds up the hydrogen evolution reaction at its surface. Faster reaction rates mean photogenerated electrons are consumed more quickly, so fewer charges accumulate inside the perovskite stack. Advanced measurements of current and voltage fluctuations reveal that with NIR heating, electrons and holes recombine less and move more cleanly through the multilayer structure. Over longer operation, the perovskite in the unheated devices develops more defects, shows signs of ion migration—especially iodine drifting toward the hole‑transport layer—and accumulates chemical damage. In contrast, the NIR‑assisted devices show fewer new traps, weaker signs of ion migration, and much less structural degradation, indicating that stable, rapid charge extraction is key to preserving the material.
Keeping Bubbles and Catalysts Under Control
Hydrogen bubbles forming on the catalyst can also destabilize the system by blocking active sites and physically stressing the catalyst particles. High-speed videos show that without NIR heating, large bubbles grow and cling to the catalyst surface before finally detaching, increasing the chance that platinum particles are pulled off. When the catalyst is gently heated, bubbles form and detach more quickly and at smaller sizes. Simulations suggest that tiny temperature gradients in the water induce local fluid motion, helping sweep bubbles away in a kind of built‑in micro‑stirring. This behavior reduces current fluctuations and slows the mechanical degradation of the catalyst, complementing the electronic benefits of faster reaction kinetics. 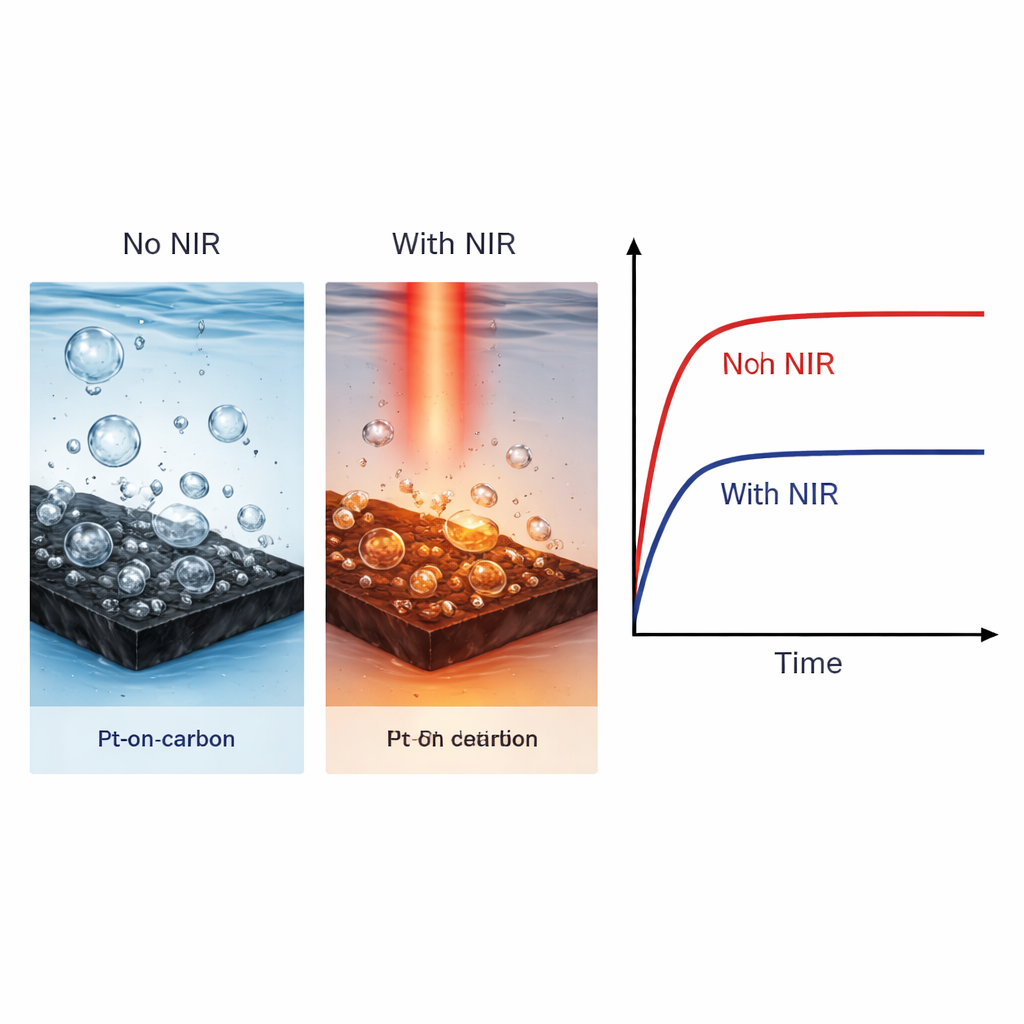
Toward Practical Solar Hydrogen Devices
Finally, the researchers combine their improved perovskite cathode with a perovskite-based anode that produces oxygen, arranging both in a side‑by‑side, light‑sharing configuration. Without any external voltage, the full system reaches a solar‑to‑hydrogen efficiency of about 15% and maintains 70% of its initial output for 115 hours. For a layperson, the bottom line is that this work shows how subtle control of temperature—focused on the catalyst rather than the fragile light absorber—can dramatically extend the lifetime of high‑efficiency solar water splitting devices. It points to a future where compact, perovskite-based “artificial leaves” could generate clean hydrogen fuel reliably and cheaply, helping decarbonize sectors that are hard to electrify directly.
Citation: Jeong, CS., Jeong, W., Yun, J. et al. Operando insights into stability of perovskite-based solar water splitting devices. Nat Commun 17, 1638 (2026). https://doi.org/10.1038/s41467-026-68357-9
Keywords: solar water splitting, perovskite, hydrogen fuel, photocatalysis, renewable energy