Clear Sky Science · en
Interphase electron redistribution induced by confined transformation in PtPdBiSn nanoplates for efficient ethanol oxidation electrocatalysis
Turning Alcohol into Clean Power
Liquid fuels like ethanol are appealing for future clean energy: they are easy to store, can be made from biomass, and fit naturally into existing fuel systems. But our current catalysts—the materials that help turn ethanol into electricity in fuel cells—waste much of its energy and degrade too quickly. This paper describes a new way to redesign tiny metal particles so they move electrons more efficiently inside themselves, dramatically boosting how well they power ethanol fuel cells.
Why Ethanol Fuel Cells Need Better Helpers
Direct ethanol fuel cells use ethanol as a liquid fuel to generate electricity with high energy density and low emissions. Their weak point is the anode catalyst, usually based on platinum. To fully use ethanol’s energy, several strong carbon–carbon and carbon–hydrogen bonds must be broken in a precise sequence, while avoiding the buildup of poisonous by-products such as carbon monoxide on the catalyst surface. Traditional strategies tweak the mix of metals and the surface composition of tiny particles, but keep their internal crystal structure fixed. That limits how much the electrons inside these particles can be redistributed to create truly ideal reaction sites.
Rebuilding Nanoplates from the Inside Out
The authors start with carefully engineered hexagonal nanoplates made from four metals: platinum, palladium, bismuth, and tin. These plates have a layered structure: an ordered inner region and a surrounding shell of a different crystal type. Pt and Pd supply the main activity for ethanol oxidation, while Bi and Sn help bind oxygen-containing species that clean off poisons. The key twist is that the team then deliberately transforms the crystal structure of the core using gentle electrochemical cycling in an alkaline ethanol solution. During this “electrochemical reconstruction,” some tin dissolves away and the initially ordered core becomes a more open, disordered hexagonal arrangement, while the outer shell keeps its original form and the overall hexagonal shape is preserved. 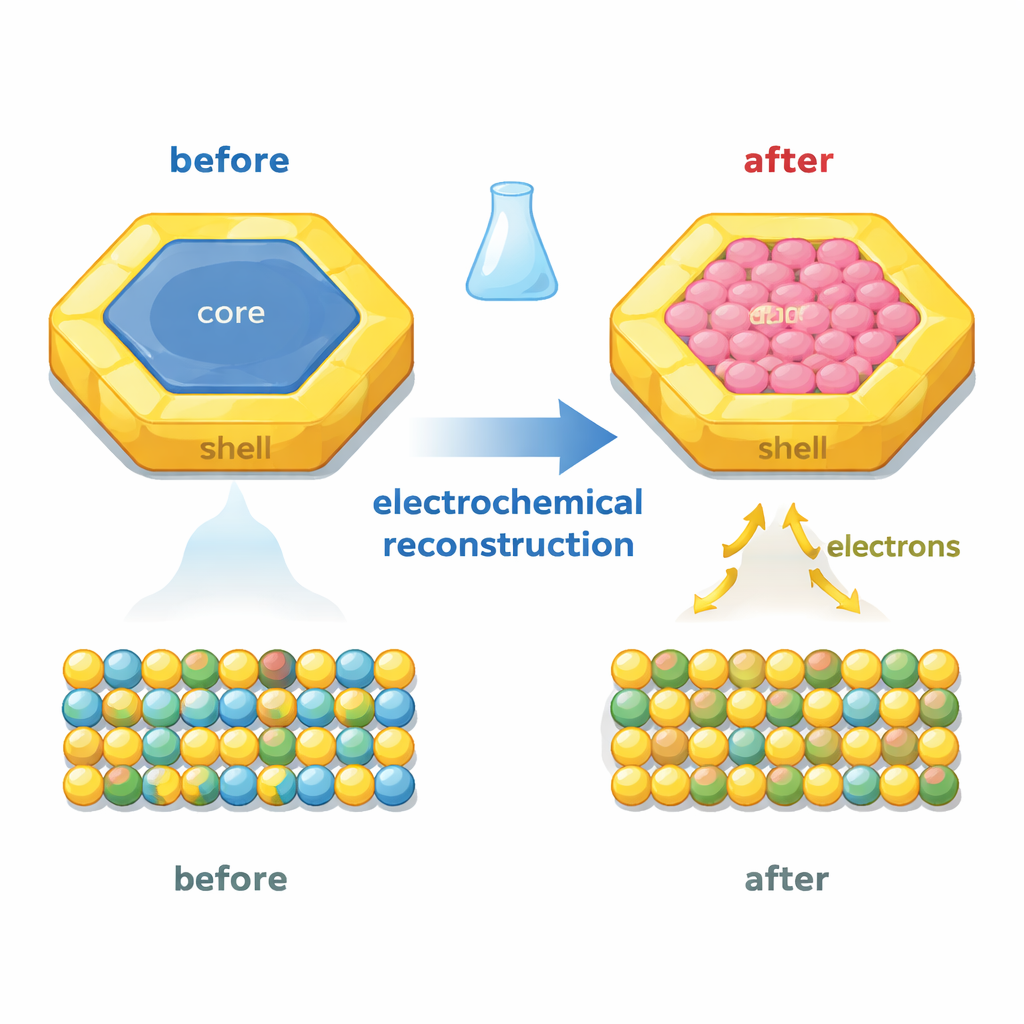
Making the Shell Electron-Rich
With advanced electron microscopy and X-ray methods, combined with quantum mechanical calculations, the researchers show that this internal restructuring changes how electrons are shared between the core and shell. In the original particles, electrons tend to flow from the shell into the core. After reconstruction, the direction reverses and the flow becomes much stronger: electrons now move from the bismuth-rich core out toward the platinum–palladium shell. This makes the shell electron-rich, which weakens the grip on poisoning molecules like carbon monoxide while still holding oxygen-containing species strongly enough to help oxidize reaction leftovers. Electronic structure analyses reveal that the coupling between the orbitals of Bi, Pt, and Pd is strengthened, and key energy levels shift closer to the ideal range for catalytic reactions.
Catalyst that Stays Fast and Resists Poisoning
These rebuilt nanoplates deliver exceptionally high performance for ethanol oxidation in alkaline solution. When supported on carbon, the new catalyst shows mass activity about 18 times higher than a commercial platinum-on-carbon benchmark and specific activity about 26 times higher. It also holds onto roughly 80% of its initial activity even after 20,000 operating cycles, far outlasting standard catalysts. Detailed spectroscopic studies indicate that the catalyst steers ethanol toward the so-called C1 pathway, where ethanol is fully oxidized to carbon dioxide, rather than stopping at partially oxidized products. At the same time, it shows greatly reduced buildup of surface carbon monoxide, thanks to both the discontinuous pattern of Pt sites and the presence of oxygen-loving Sn at the surface, which helps bring in hydroxyl groups that rapidly remove CO. 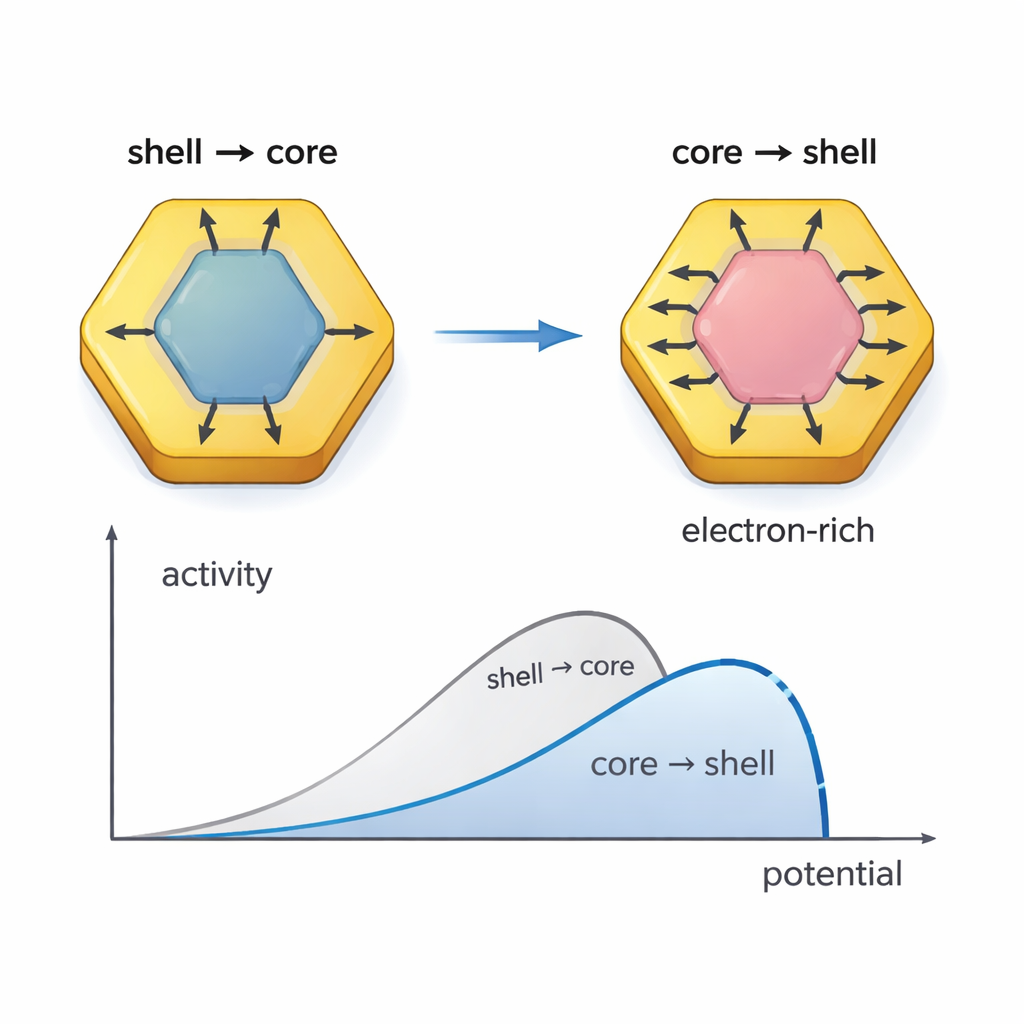
From Lab Discovery to Practical Devices
To test real-world potential, the team built complete direct ethanol fuel cells. Using their new nanoplates as the anode and a standard platinum cathode, they achieved a much higher power output than a cell using platinum on both sides, while using far less noble metal. The improved device also ran steadily over many hours, reflecting the structural stability of the reconfigured particles. The authors’ calculations support the experiments, showing that the new core–shell structure lowers the energy barriers for breaking ethanol’s bonds and for splitting the carbon–carbon bond, all while reducing the tendency to bind CO too strongly.
A New Knob for Tuning Tiny Catalysts
In simple terms, this work shows that how atoms are arranged in the heart of a nanoparticle can be as important as which elements are present at the surface. By carefully transforming the inner crystal structure while keeping the outer shell intact, the researchers created a controlled flow of electrons from core to shell, turning the shell into an especially effective reaction zone. This design principle—using “confined transformation” inside core–shell particles to reshape internal electron distribution—could guide the creation of many new catalysts, not only for ethanol fuel cells but also for other clean energy and chemical processes.
Citation: Shao, M., Wang, A., Fu, H. et al. Interphase electron redistribution induced by confined transformation in PtPdBiSn nanoplates for efficient ethanol oxidation electrocatalysis. Nat Commun 17, 1635 (2026). https://doi.org/10.1038/s41467-026-68352-0
Keywords: ethanol fuel cells, electrocatalysis, nanoparticles, core–shell catalysts, clean energy