Clear Sky Science · en
Molecular heterogeneity of the non-human primate cochlea
Why the inner ear’s hidden world matters
Hearing seems effortless, but it depends on an astonishingly intricate machine buried deep in the skull: the cochlea of the inner ear. When this delicate structure fails, the result is often permanent hearing loss. Most of what we know about cochleas comes from mice, yet treatments must ultimately work in humans. This study opens a rare window into the cochlea of a non-human primate, the macaque, whose hearing system is much closer to ours. By cataloging tens of thousands of individual cells, the researchers reveal how the primate cochlea is built, how it resembles the mouse ear, and where it crucially differs—especially in cell types linked to inherited deafness and future gene therapies.
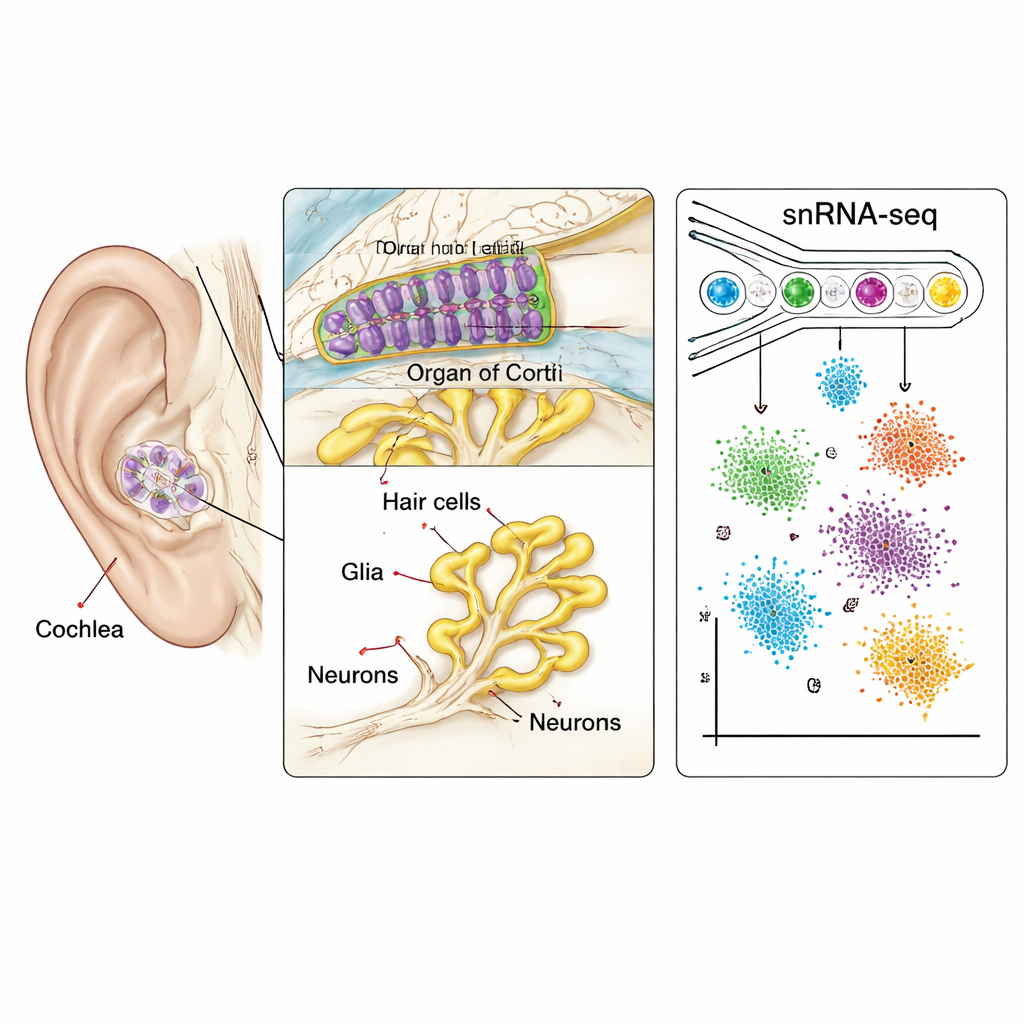
Mapping every cell in a tiny spiral
The cochlea is a fluid-filled spiral that converts sound vibrations into electrical signals. Inside it sits a strip of tissue called the organ of Corti, lined with sensory hair cells, supporting cells, and neurons that carry information to the brain. Because these cells are few, fragile, and encased in bone, they have been extremely hard to study in primates. Here, scientists used single-nucleus RNA sequencing, a high-throughput method that reads which genes are active inside individual cell nuclei. Working with cochleas from juvenile and adult macaques, they profiled more than 36,000 nuclei taken from nearly all major regions: the sensory epithelium, the nerve cluster called the spiral ganglion, and the surrounding tissues that regulate fluid and blood supply.
A conserved blueprint for hearing
Each nucleus was grouped into cell types based on its gene activity, creating a “cell atlas” of the macaque cochlea. When this atlas was compared with similar data from mice, the overall blueprint looked surprisingly familiar. The sensory hair cells that detect sound, and the spiral ganglion neurons that send signals to the brain, showed highly conserved molecular signatures. Key genes that distinguish inner hair cells (which send information) from outer hair cells (which amplify sound) were turned on in similar patterns in both species. Even specialized motor proteins like prestin, which lets outer hair cells physically change length in response to electrical signals, were present and functional in macaques as in mice. This suggests that the core machinery of hearing has been strongly preserved across mammalian evolution.
Glia and neurons show primate twists
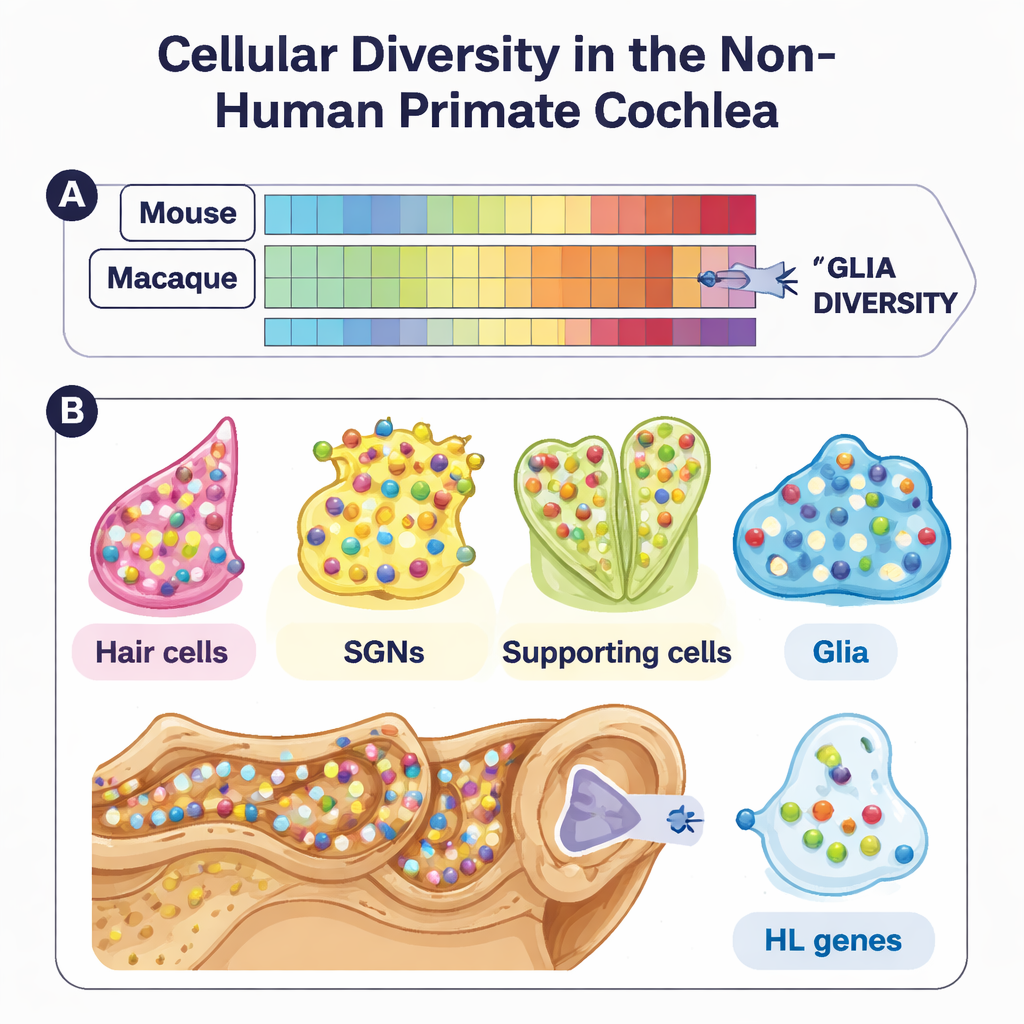
Beneath this shared blueprint, however, the study uncovered important primate-specific twists. In particular, glial cells—support cells that wrap, nourish, and communicate with neurons—were much more diverse in macaques than in mice at the molecular level. Although their shapes and locations looked similar under the microscope, their gene expression patterns diverged, especially for genes involved in ion balance, debris clearance, and chemical signaling. Spiral ganglion neurons also showed fine-grained diversity. The team identified subtypes of these sensory neurons and found that some marker genes used to define neuron classes in mice did not line up cleanly in macaques. One transcription factor, PBX3, emerged as a primate-enriched regulator in certain neuron subtypes, hinting at evolutionary refinements in how primate ears encode sound.
A genetic roadmap for hearing loss
To link this atlas to human disease, the researchers overlaid hundreds of known deafness genes—drawn from clinical genetics databases—onto their macaque cell map. Many of these genes turned out to be sharply restricted to particular cell types, such as hair cells, supporting cells, or lateral wall cells that help generate the ear’s internal voltage. Others were concentrated in glia or neurons. The overall distribution closely mirrored what is seen in mice, reinforcing that macaques are a relevant model for human hearing. By pinpointing where each deafness gene is normally active, the atlas provides a kind of “disease map” that can guide targeted gene therapies and drug delivery strategies, especially as such treatments move from mouse models into larger animals.
What this means for future hearing therapies
For a non-specialist, the key message is that scientists have, for the first time, drawn a detailed, cell-by-cell map of the primate cochlea and compared it directly with the well-studied mouse ear. The take-home conclusion is reassuring and cautionary at once: the main hearing machinery is strongly conserved, which supports using mouse studies to design treatments, but important differences—especially in glial cells and certain neurons—could influence how therapies work in humans. This macaque cochlear atlas now serves as a crucial bridge between basic research in rodents and clinical advances for people living with hearing loss.
Citation: Chen, X., Che, Y., Qi, J. et al. Molecular heterogeneity of the non-human primate cochlea. Nat Commun 17, 1633 (2026). https://doi.org/10.1038/s41467-026-68350-2
Keywords: cochlea, single-cell atlas, hearing loss, non-human primate, gene expression