Clear Sky Science · en
From TDP-43/RNA complex formation to disease-linked TDP-43 aggregation through a structural and cellular approach
Why this brain protein matters
Neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS) and frontotemporal dementia often feature clumps of misfolded proteins inside nerve cells. One of the most common culprits is a protein called TDP-43, which normally helps process RNA—the working copy of our genes. This study asks a crucial question: what does TDP-43 do in healthy cells, and what goes wrong that turns a useful RNA helper into a toxic aggregate-forming protein?
How TDP-43 works in healthy nerve cells
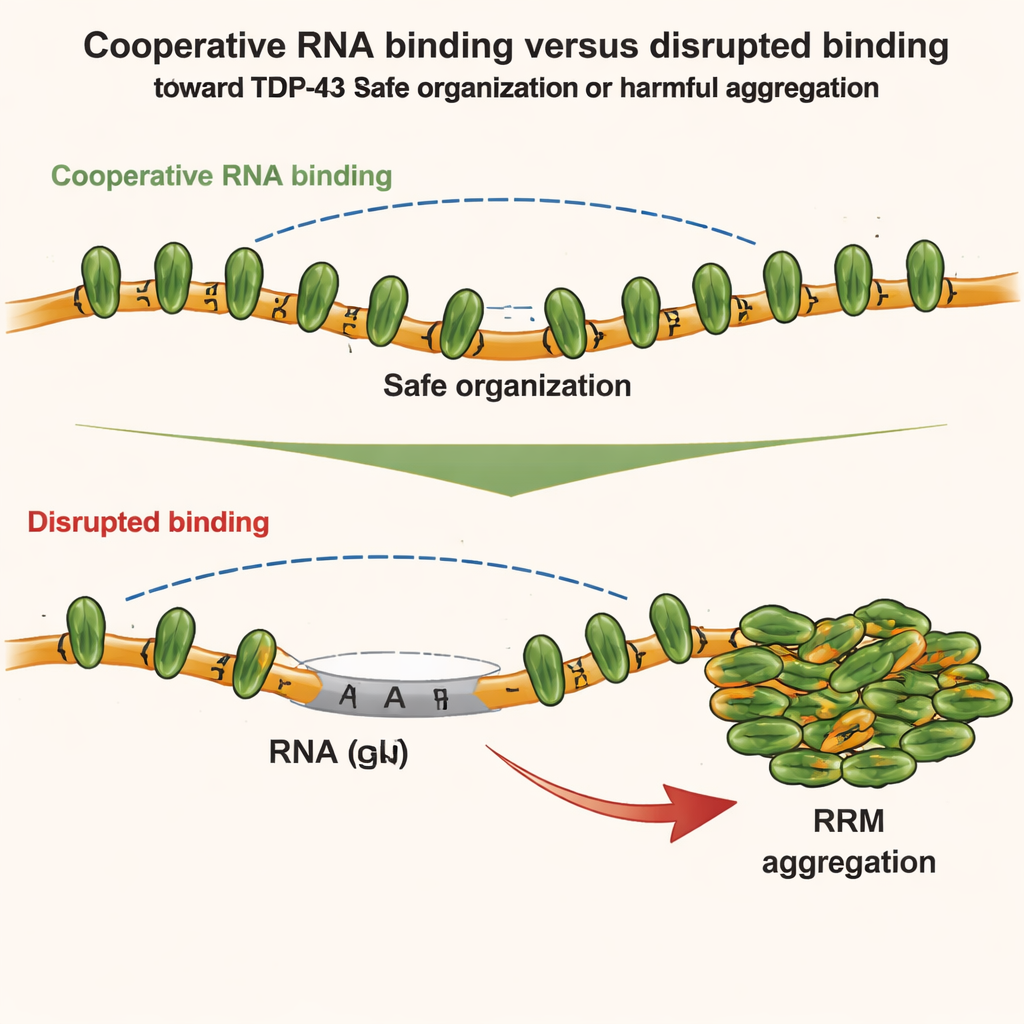
TDP-43 usually resides in the cell nucleus, where it binds long stretches of RNA rich in repeated “GU” building blocks. Using a combination of structural biology and cell experiments, the authors show that TDP-43 molecules line up cooperatively along these GU-rich regions, forming smooth, continuous chains on the RNA. When arranged this way, the front end of the protein—its N-terminal domain—stays physically separated between neighboring molecules. As a result, these N-terminal segments remain available to reach across and connect distant GU-rich sections of the same RNA, gently looping and compacting long introns (the non-coding stretches inside genes) without clumping.
A built-in tug-of-war inside the protein
The team then explored how different parts of TDP-43 compete or cooperate with each other. They focused on two regions: the N-terminal domain, which can stick to other N-terminal domains, and the central RNA-binding modules, which drive cooperative binding along GU-rich RNA. Using sensitive techniques such as NMR spectroscopy, calorimetry, and small-angle X-ray scattering, they found that when TDP-43 binds RNA cooperatively, the geometry of the chain actually weakens direct N-terminal-to-N-terminal contacts between adjacent molecules. In other words, strong cooperative RNA binding keeps local N-terminal interactions in check. But when this cooperativity is broken—for example, by inserting non-GU stretches into the RNA or by mutating TDP-43—N-terminal regions on neighboring proteins move close together and can now latch onto each other.
From helpful compaction to harmful clumping
These structural insights were tested in living cells. The authors used engineered cell systems to track how different TDP-43 mutants mix or separate inside artificial RNA-rich compartments. They found that both cooperative RNA binding and N-terminal interactions help organize TDP-43 into higher-order assemblies, but in different ways. Cooperative binding strings proteins along GU-rich RNA, while N-terminal contacts link distant clusters together. Under mild oxidative stress, however, the picture shifts. A chemical stressor (arsenite) causes specific chemical changes—acetylation—on the central RNA-binding motifs, making them prone to sticking together. If cooperative binding to RNA is intact, TDP-43 remains mostly soluble. If cooperativity is weakened, N-terminal links between nearby TDP-43 molecules act like a scaffold that brings the acetylated cores together, encouraging irreversible aggregation.
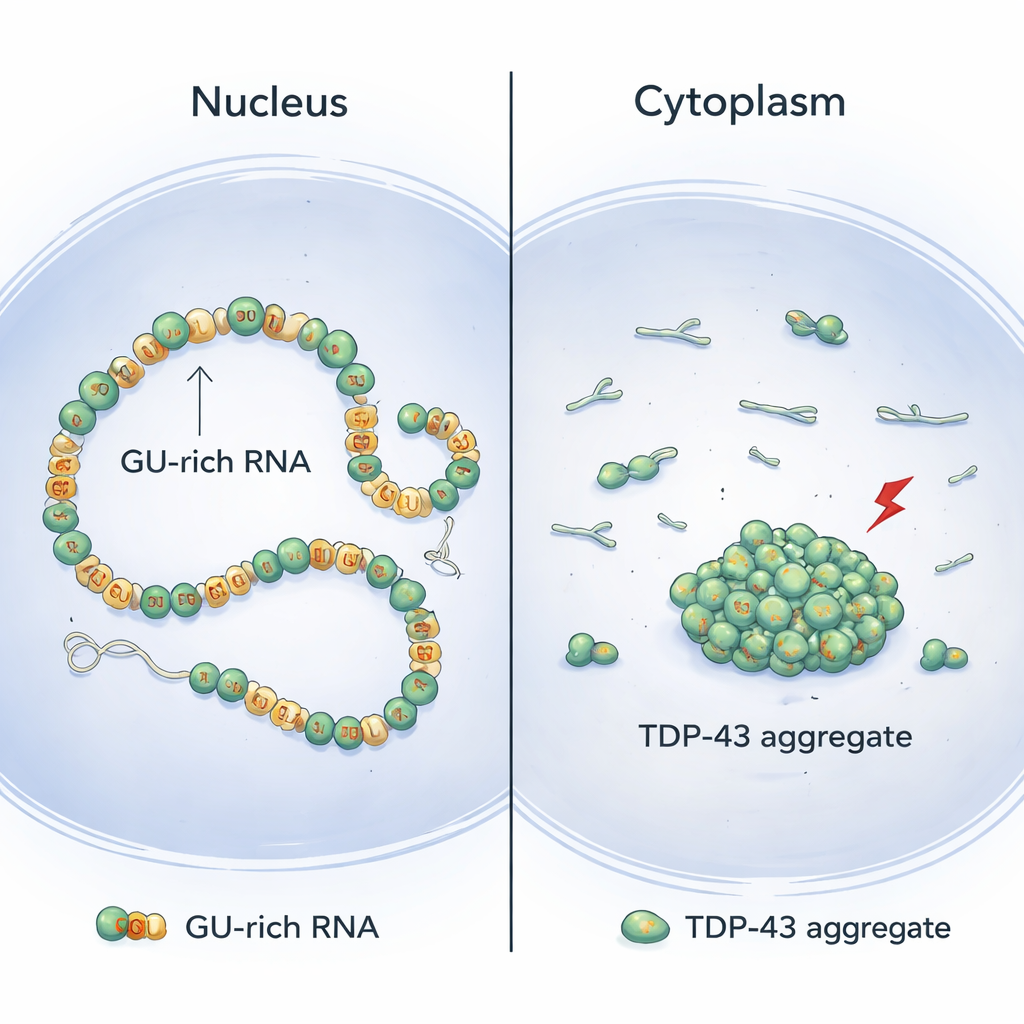
Why TDP-43 clumps more in the cytoplasm
The model emerging from this work helps explain why TDP-43 aggregates are often found in the cytoplasm of diseased neurons rather than in the nucleus. In the nucleus, long introns with many GU repeats are common, favoring continuous, cooperative binding that keeps risky N-terminal contacts between neighbors low and allows safe long-range looping instead. In the cytoplasm, by contrast, mature RNAs have far fewer GU-rich stretches. This makes TDP-43 binding more patchy and less cooperative, increasing the chances that neighboring N-terminal domains will interact and set the stage for aggregation once stress-induced chemical changes occur. Supporting this view, artificially supplying extra GU-rich introns in the nucleus reduced TDP-43 aggregation under stress.
What this means for future therapies
Put simply, the study paints TDP-43 as a protein balanced between helpful RNA organization and harmful clumping, with cooperative binding to GU-rich RNA acting as a safety mechanism. When that safety net fails—because of altered RNA sequences, protein mutations, or cellular stress—the same domains that normally compact long RNA segments can instead drive formation of toxic aggregates. For non-specialists, the key takeaway is that maintaining the right kind of interactions between TDP-43 and RNA may be a promising strategy to prevent or slow the protein clumping seen in ALS and related brain diseases.
Citation: Feng, Y., Joshi, V., Pankivskyi, S. et al. From TDP-43/RNA complex formation to disease-linked TDP-43 aggregation through a structural and cellular approach. Nat Commun 17, 1631 (2026). https://doi.org/10.1038/s41467-026-68346-y
Keywords: TDP-43 aggregation, RNA-binding proteins, ALS, protein–RNA interactions, neurodegeneration