Clear Sky Science · en
Atomically precise ligand engineering of gold nanoparticles via interphase mass transfer
Why tiny gold particles matter for health
Medical imaging increasingly relies on nanoparticles—tiny particles a few thousand times smaller than a human hair—to light up organs and tumors deep inside the body. This study shows how scientists can now fine-tune the outer coating of ultrasmall gold particles almost atom by atom, so that the same glowing core can be steered toward different organs, especially shifting from the liver and spleen to the kidneys. Such control could help design safer imaging agents and drug carriers that go exactly where doctors want them and exit the body more cleanly.
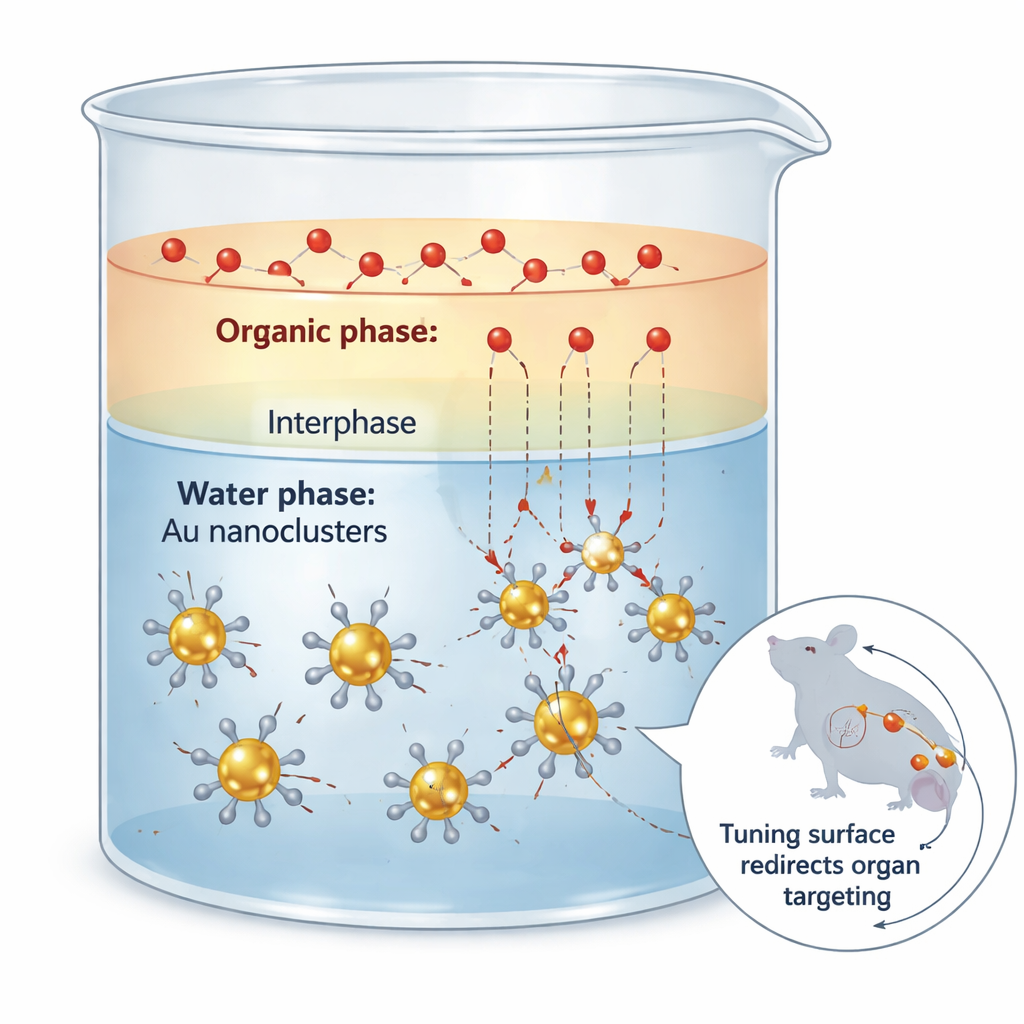
The challenge of dressing up tiny gold clusters
Gold nanoclusters are ultra-small gold particles that behave more like large molecules than metal grains. They can emit light in the near-infrared II window, which penetrates deeply into tissue and gives sharp images with low background. However, almost everything about how these clusters behave in the body—where they travel, how long they circulate, and how they are cleared—depends not on the gold itself, but on the organic ligands, or "shell" molecules, attached to their surface. Traditional ways of changing this shell often work like rough surgery: the reaction goes too fast, the coating ends up uneven, and the gold core can be partially eaten away, ruining both structure and optical properties.
Slowing the traffic between two liquids
The researchers tackled this problem by exploiting a simple physical idea: mass transfer resistance at the boundary between two liquids that do not fully mix. They placed the gold clusters in water and the incoming sulfur-based ligands in an organic solvent such as ethyl acetate. At the thin region where the two liquids partially mix, ligands slowly diffuse toward the clusters and exchange with the original shell. Careful kinetic measurements showed that the unwanted "etching" of the gold core is extremely sensitive to ligand concentration, while the desired exchange is much less so. By using the two-phase setup to keep free ligand levels low but continuously replenished, the team suppressed etching by roughly sixty-fold while preserving most of the useful exchange rate.
Building a precise molecular toolkit
With this interphase-assisted method, the authors could replace surface ligands on a model gold cluster, Au25, in a highly controlled way. They started from clusters protected by a negatively charged sulfonic acid ligand and swapped in a variety of new thiol ligands carrying carboxylic acids, amines, hydroxyls, nitro groups, and hydrophobic aromatic rings. Mass spectrometry revealed clean, well-defined mixtures such as Au25 clusters bearing specific numbers of each ligand type, rather than broad, messy distributions. The method also worked for other cluster sizes and ligand families, suggesting it is a general strategy for crafting robust, tailor-made gold nanoclusters for different applications.
Guiding nanoparticles from liver to kidneys
The most striking test of this precision came from in vivo imaging experiments in mice. By gradually introducing a positively charged ligand, p-aminothiophenol, into the otherwise negatively charged shell, the team created a series of clusters whose overall surface charge shifted from strongly negative to near-zwitterionic (balanced positive and negative groups). All versions glowed similarly in lab tests, but their biodistribution in mice differed dramatically. Purely negative clusters mainly accumulated in liver and spleen. As more positive ligands were added, less signal appeared in these organs and more in the kidneys and bladder, indicating enhanced renal clearance. When a neutral hydroxyl ligand was used instead of the amine, this shift did not occur, underscoring the special role of positive charges in redirecting the particles.
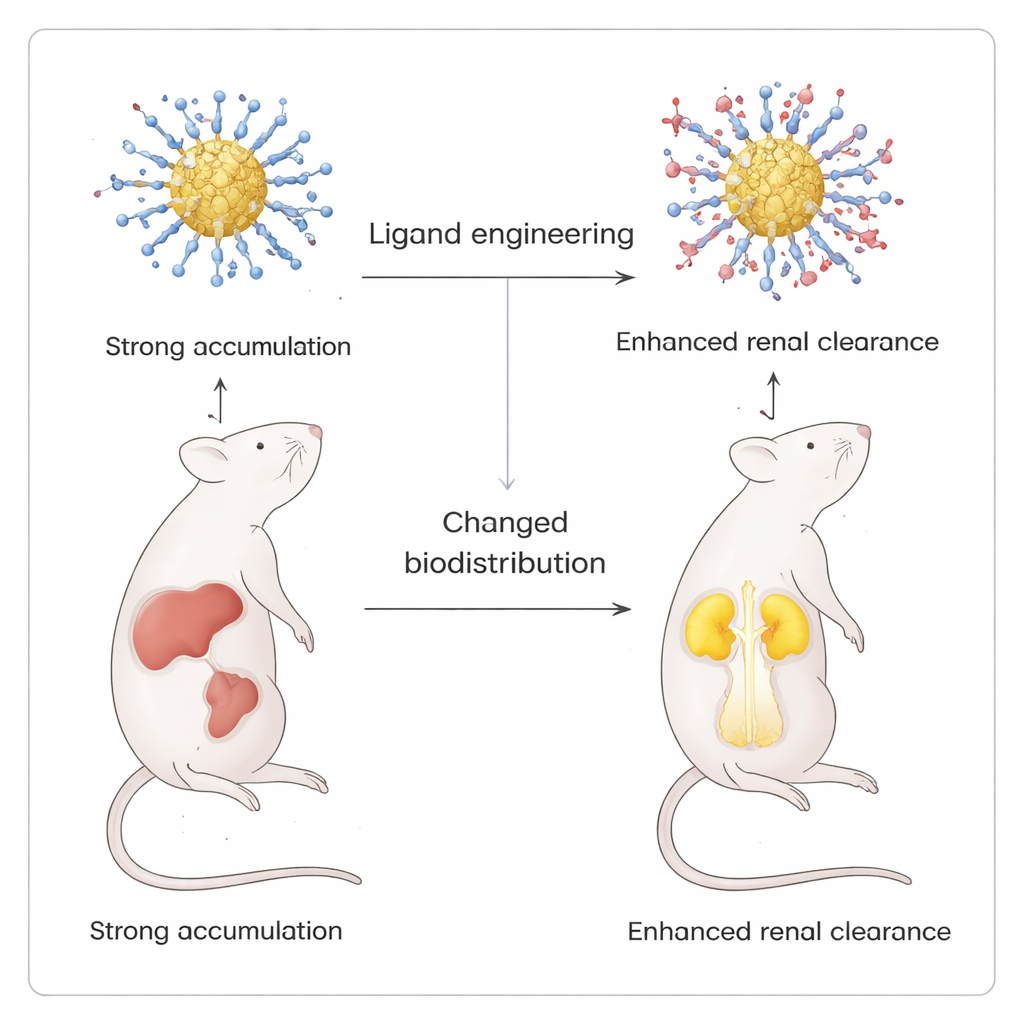
What this means for future medicines
For non-specialists, the key takeaway is that scientists have found a way to “dress” glowing gold nanoclusters with molecular coats that can be tuned almost one molecule at a time, without damaging the particle underneath. By using a gentle two-liquid setup, they can control both the chemistry and the ultimate fate of these particles inside the body. This level of control makes it possible to design imaging agents and potential drug carriers that light up specific organs and then leave through the kidneys rather than lingering in the liver and spleen, paving the way for safer and more predictable nanomedicine.
Citation: Zhang, B., Xiao, F., Song, X. et al. Atomically precise ligand engineering of gold nanoparticles via interphase mass transfer. Nat Commun 17, 1630 (2026). https://doi.org/10.1038/s41467-026-68345-z
Keywords: gold nanoclusters, ligand engineering, bioimaging, nanomedicine, biodistribution