Clear Sky Science · en
Cross-species evidence for a developmental origin of adult hypersomnia with loss of synaptic adhesion molecules beat-Ia/CADM2
Why Too Much Sleep Can Be a Serious Problem
Many people envy heavy sleepers, but for those with idiopathic hypersomnia, overwhelming sleepiness can wreck work, school, and relationships. This condition leaves people exhausted despite a full night of rest, and doctors still do not fully understand why. This study combines human genetics with experiments in fruit flies and zebrafish to trace excessive sleepiness back to how brain circuits are wired during development, pointing to a potential new treatment strategy.
Finding Sleepiness Genes in Human DNA
The researchers began by mining massive genetic studies of hundreds of thousands of people who reported excessive daytime sleepiness or frequent napping. Instead of assuming that the closest gene to each risk variant was important, they looked across larger 3D neighborhoods of the genome called "topological domains" to gather all plausible genes. They then used computational tools to find matching genes in fruit flies, ending up with more than 200 fly genes to test. Systematically turning these genes down in all neurons, they looked for flies that slept far more than normal. Among the strongest hits were genes from the “beaten path” family, counterparts of the human gene CADM2, which encodes a molecule that helps nerve cells stick and connect at synapses. 
Sleepy Flies and Sleepy Fish
When the fly version of CADM2, called beat-Ia, was reduced in neurons, the flies slept much longer both day and night. They were not sluggish when awake; instead, their sleep bouts were longer, they were harder to wake, and they fell back asleep more quickly after lights came on—features that closely mirror human hypersomnia. The team went on to test CADM2 in zebrafish, a small vertebrate whose sleep can be tracked by video. Disrupting the fish gene cadm2b made the fish sleep more without reducing their movement when awake, supporting a conserved role for this molecule in keeping animals properly alert.
How Early Brain Wiring Shapes Lifelong Sleep
A key insight was that beat-Ia matters most during brain development, not adulthood. By turning gene knockdown on only before or only after flies emerged from their pupal stage, the researchers showed that disrupting beat-Ia early in life was enough to cause life-long excessive sleepiness, whereas turning it down only in adults had little effect. They traced beat-Ia’s action to a small set of neurons that make neuropeptide F (NPF), the fly counterpart of vertebrate neuropeptide Y (NPY). In normal flies, NPF neurons send dense synaptic projections into a brain region called the suboesophageal zone, where they connect onto specific inhibitory (GABA-producing) neurons that help stabilize wakefulness. In flies lacking beat-Ia, the large clusters of synapses in this region failed to form, even though the nerve fibers physically reached the area. This suggests that faulty synapse formation, rather than gross miswiring, can tilt the balance toward excessive sleep.
From Miswired Circuits to a Drug Target
Using a detailed wiring map of the fly brain, the team identified a handful of downstream neurons in the suboesophageal zone that receive input from NPF cells and are predicted to be GABAergic. Silencing these cells increased sleep, while activating them promoted wakefulness, matching the idea that NPF normally keeps people awake by driving a local inhibitory network. The researchers then asked whether they could compensate for lost CADM2-like function by boosting NPY signaling. In cadm2b-deficient zebrafish, bathing the larvae in a drug that activates one NPY receptor subtype (similar to the fly NPF receptor) brought their excessive sleep back to normal levels, without strongly affecting normal fish. This cross-species result suggests that when synaptic adhesion molecules go awry during development, enhancing NPY pathways in adulthood might help restore proper sleep–wake balance. 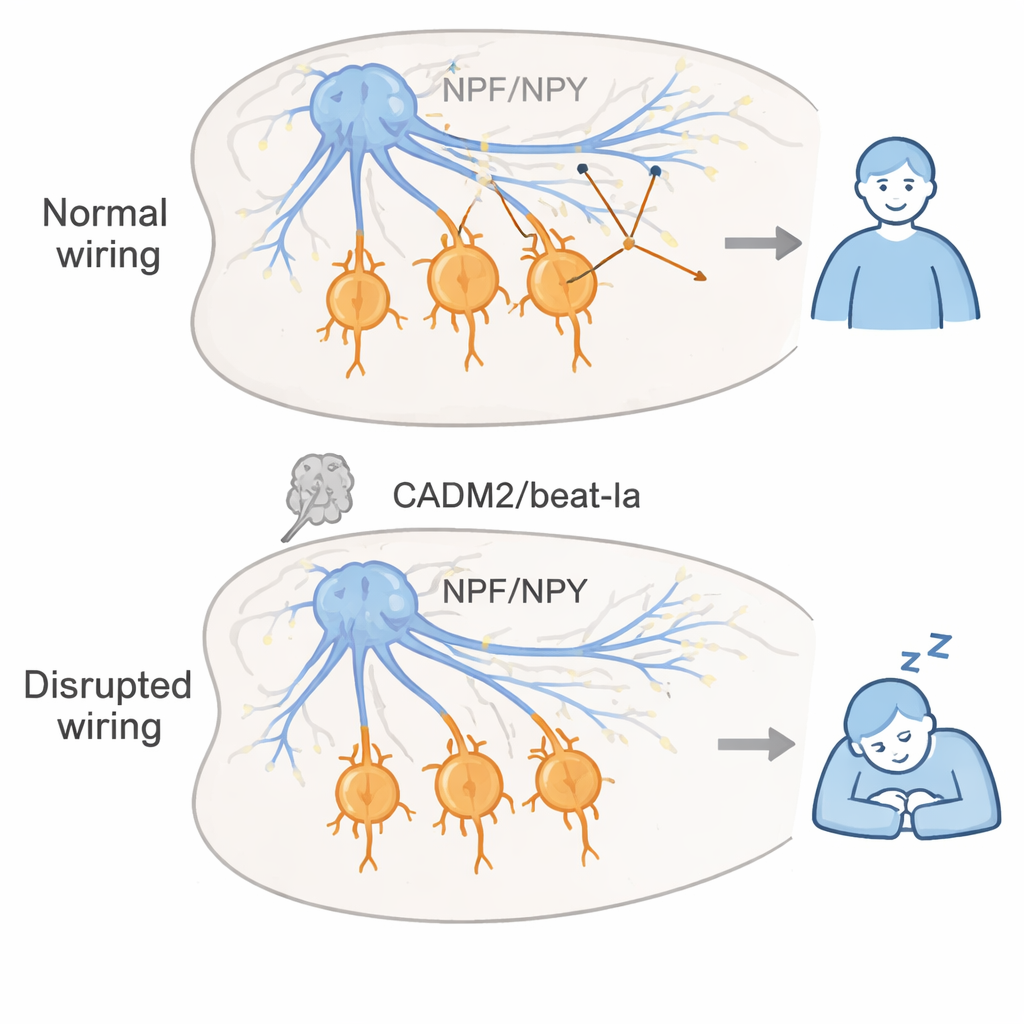
What This Means for People Who Can’t Stay Awake
Altogether, the work proposes that some forms of idiopathic hypersomnia may stem from subtle mistakes in how wake-promoting brain circuits are wired during early life, involving CADM2 and related adhesion molecules. These changes do not destroy the brain but reconfigure how strongly certain sleep and arousal pathways talk to each other. Importantly, the study also shows that even if the wiring problem begins in development, its effects may still be treatable later by targeting conserved neuropeptide systems like NPY. For patients, this raises the possibility that future drugs designed to fine-tune these signaling pathways could offer more precise relief from disabling daytime sleepiness.
Citation: Mace, K., Zimmerman, A., Chesi, A. et al. Cross-species evidence for a developmental origin of adult hypersomnia with loss of synaptic adhesion molecules beat-Ia/CADM2. Nat Commun 17, 1628 (2026). https://doi.org/10.1038/s41467-026-68343-1
Keywords: idiopathic hypersomnia, sleep genetics, synaptic adhesion, neuropeptide Y, brain development