Clear Sky Science · en
A water-soluble copolymer for storage and electron conversion in photocatalytic on-demand hydrogen evolution
A New Way to Bottle Sunshine
Modern societies need huge amounts of energy, but sunshine does not always shine when we need it. This research explores a clever way to "bottle" solar energy in a liquid material and release it later as clean hydrogen fuel on demand. Instead of big metal batteries, the authors use a special water‑soluble plastic that can soak up electrons under light and then give them back later to make hydrogen gas, a potential green fuel for industry and transport.
Turning a Plastic into a Temporary Battery
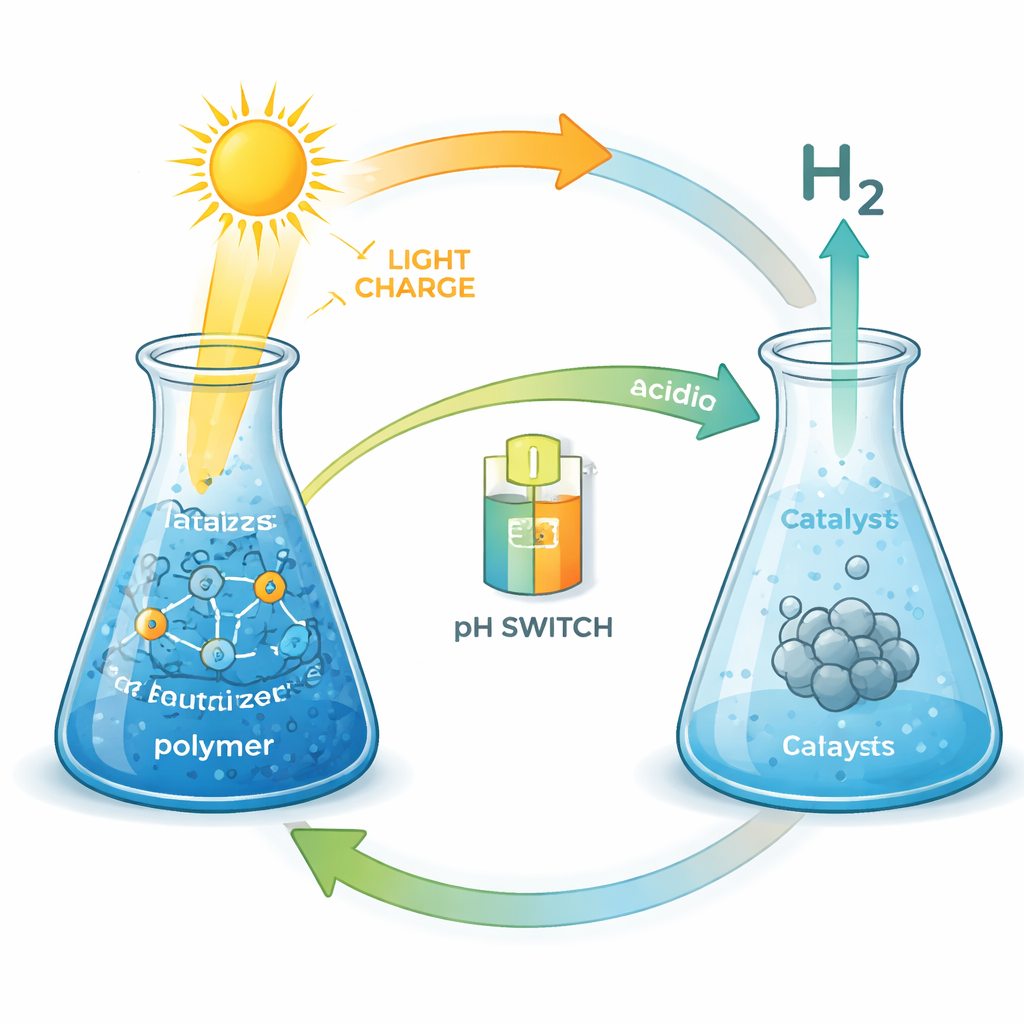
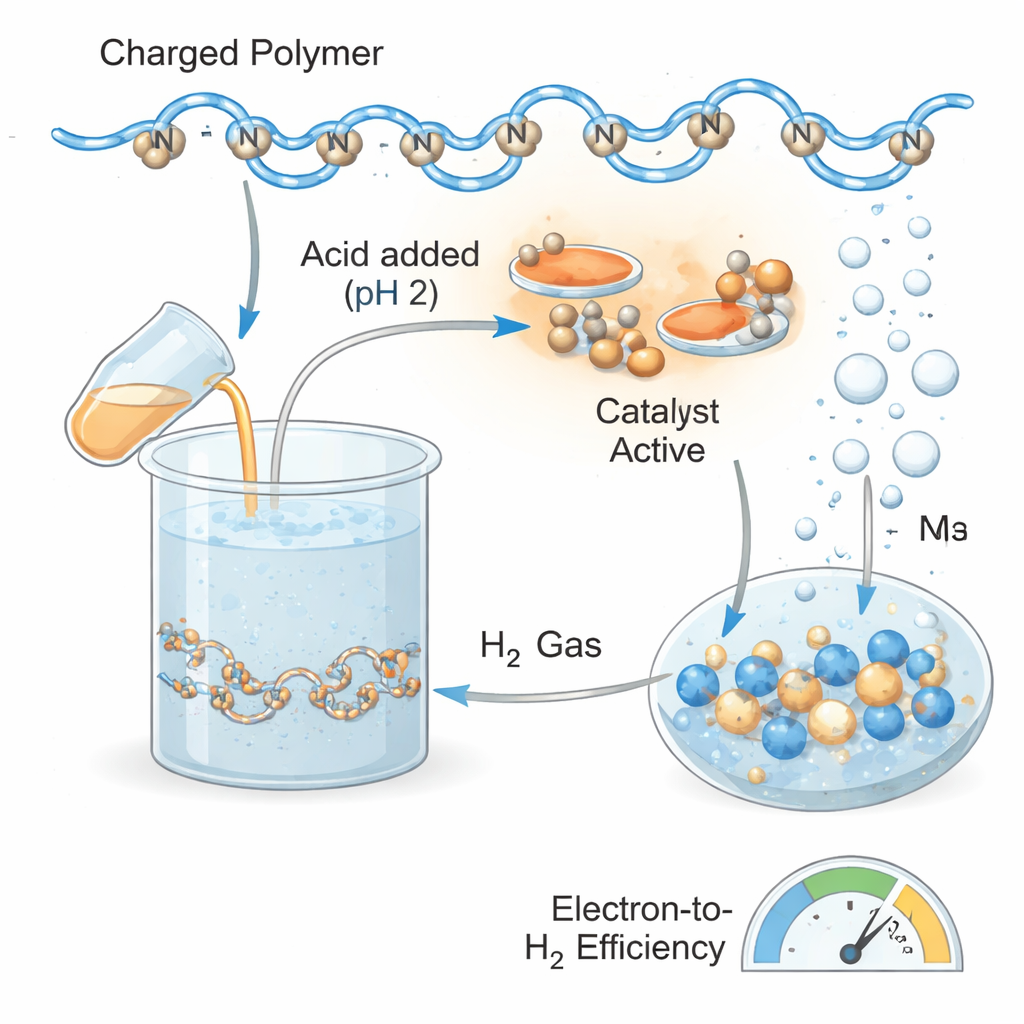
At the heart of the study is a tailor‑made copolymer, a long chain molecule built from two types of building blocks. One part keeps the material easily dissolved in water; the other part contains so‑called viologen units that behave a bit like tiny rechargeable battery cells. When the solution is lit with visible light in the presence of a ruthenium dye and a simple sacrificial additive, electrons are moved from the additive onto the polymer. In effect, light “charges” the polymer, filling many of its viologen sites with stored electrons.
Charging Up with Light and Storing for Days
The team first asked how efficiently this soft material could be charged by light. Using the ruthenium complex as a light‑absorbing helper and triethylamine as the electron source, they showed that up to about 80 percent of the available storage sites on the polymer could be filled. Careful measurements of how the solution absorbed light at specific colors allowed them to track this state of charge over time. Once charged, the violet‑colored solution remained essentially unchanged in the dark for at least three days, corresponding to a stored electrical charge of about 101 coulombs per gram of polymer—far above some recently reported solid frameworks designed for the same purpose. By comparison, a related simple viologen molecule lost a large fraction of its charge within the first day, highlighting the stabilizing effect of the polymer environment.
Releasing Clean Fuel on Demand
Charging the polymer is only half the story; the real payoff is turning the stored electrons into hydrogen gas whenever it is needed. To trigger this release, the researchers added acid to lower the solution to pH 2 and introduced different hydrogen‑producing catalysts based on platinum or rhodium. Under these conditions, the charged viologen units handed their electrons to the catalysts, which combined them with protons from the acidic solution to form molecular hydrogen. Colloidal platinum nanoparticles turned out to be the top performer: they rapidly “discharged” the polymer and converted up to about 72 percent of the stored electrons into hydrogen, a remarkably high efficiency for such a soft, water‑based system. Rhodium complexes were also effective but generally slower or less efficient, depending on how easily their metal centers could accept electrons.
Storing, Waiting, Then Fuel—Again and Again
Because the polymer and the light‑absorbing dye remain intact over the pH range used, the same solution can be used repeatedly. After hydrogen production at low pH, simply neutralizing the mixture allows it to be re‑charged with light. The authors demonstrated at least four cycles of charging and on‑demand hydrogen evolution without ever isolating or replacing the polymer. While the catalysts gradually lost some activity—partly because of chemical changes under acidic conditions and repeated pH swings—the polymer itself continued to store and release charge reliably. When the output from all cycles is added up, the reusable system delivers more than twice as much hydrogen as even a perfect single‑use system would, underscoring the benefit of recyclability.
What This Means for Future Energy Systems
For non‑experts, the key message is that this work shows a realistic pathway toward liquid “solar fuels” that can bridge the gap between when the sun shines and when energy is needed. A simple, fully water‑soluble plastic can act as a temporary energy tank: it is charged by sunlight via a dye, holds onto that energy for days without noticeable loss, and then, when triggered by an acid and a suitable catalyst, releases it as hydrogen gas at high efficiency. The whole process can be repeated multiple times using the same solution, controlled by something as simple as a pH switch. While still a laboratory system, it points toward flexible, scalable ways to store renewable energy as clean fuel for energy‑intensive processes such as future green steel production.
Citation: Hartkorn, M., Kampes, R., Müller, F. et al. A water-soluble copolymer for storage and electron conversion in photocatalytic on-demand hydrogen evolution. Nat Commun 17, 1141 (2026). https://doi.org/10.1038/s41467-026-68342-2
Keywords: solar energy storage, hydrogen fuel, photocatalysis, redox polymer, renewable energy