Clear Sky Science · en
Intranasal unadjuvanted LcrV boosts parental Yersinia OMV primed lung immunity against pneumonic plague in mice
A Two-Step Defense Against a Famous Killer
Plague may sound like a disease from medieval history, but its lung infection form, pneumonic plague, can still kill quickly and spread through the air. Antibiotics help, yet drug‑resistant strains and the threat of deliberate release keep plague on modern watch lists. This study in mice explores a new two‑stage vaccination strategy that aims not just to raise antibodies in the blood, but to train long‑lasting immune “guards” that live right inside the lungs, ready to stop a lethal infection before it takes hold.
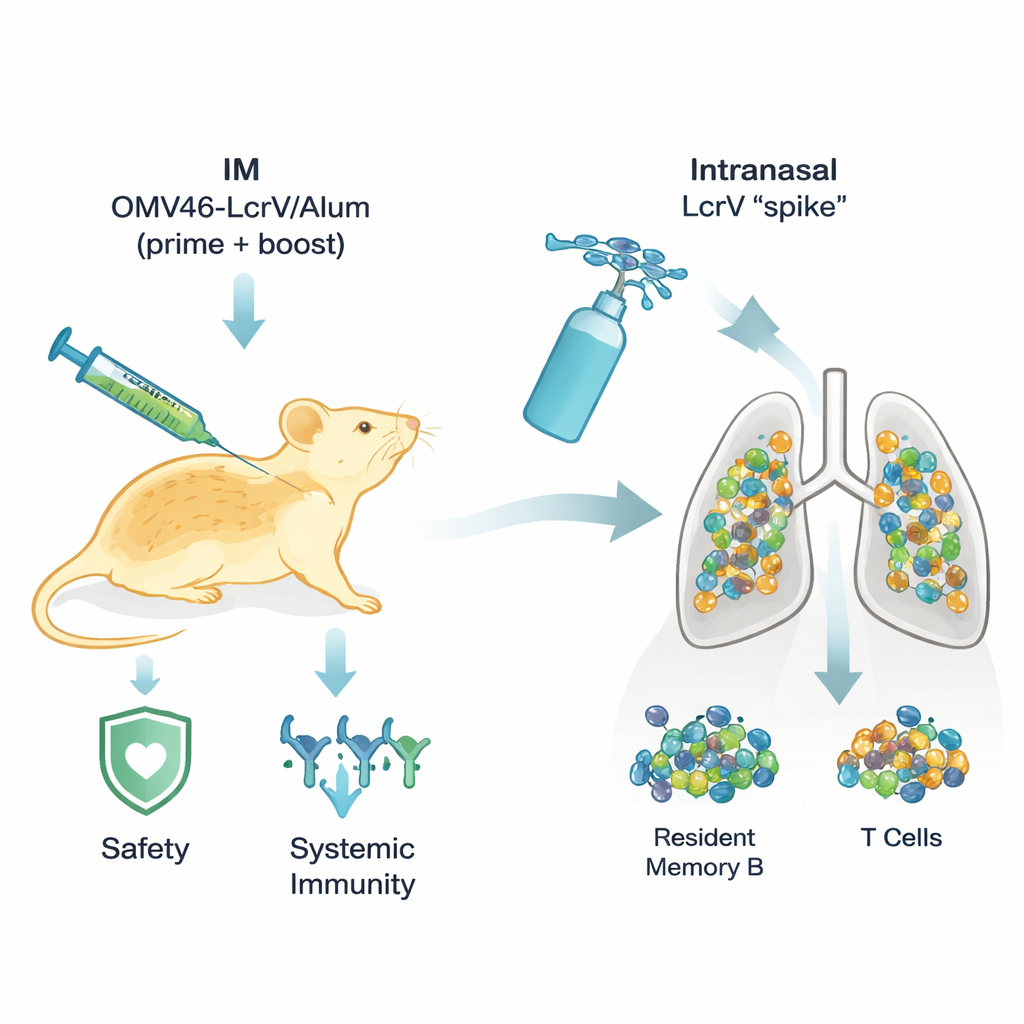
Why Lung-Focused Protection Matters
There are two main forms of plague: bubonic, which usually starts with swollen lymph nodes after a flea bite, and pneumonic, which attacks the lungs and can be nearly 100% fatal if treatment is delayed even a day. Existing experimental vaccines do a reasonable job against bubonic plague but have struggled against the lung form. One reason is that most shots are given into muscle and mainly train the immune system in the blood, not on the moist surfaces of the airways where pneumonic plague starts. To truly protect the lungs, scientists increasingly believe vaccines must seed “resident” immune cells that take up long‑term positions in lung tissue itself.
Building a Safer First-Line Vaccine
The team had previously created a promising plague vaccine based on outer membrane vesicles (OMVs) from a Yersinia relative, decorated with a key plague protein called LcrV. This OMV46‑LcrV shot strongly protected mouse lungs but caused some short‑term reactions. To tame these side effects, the researchers absorbed the OMVs onto aluminum salts ("alum"), a long‑used vaccine ingredient that generally makes vaccines safer and helps antibodies form. In mice, this alum‑bound version (OMV46‑LcrV/Alum) no longer produced weight loss or worrying inflammation signals in the blood, yet still protected most animals from very high‑dose plague challenges. However, its protection dipped slightly at the very highest infection dose, hinting that safety had been improved at a small cost to potency.
Adding a Nasal “Spike” to Supercharge the Lungs
To restore and even enhance protection without adding harsh ingredients to the lungs, the researchers borrowed a concept first tested for COVID‑19: “prime and spike.” Mice were first primed and boosted with the safer intramuscular OMV46‑LcrV/Alum shots, and later received a small dose of purified LcrV protein sprayed into the nose, with no added adjuvant. This intranasal “spike” did not disturb weight, blood counts, or lung structure. Yet it completely changed immunity in the airways. After the nasal booster, the liquid washing the lungs contained far more LcrV‑specific antibodies, including IgA, a form especially suited to coating and protecting mucosal surfaces. The lungs also filled with resident memory B cells (which can rapidly become antibody‑secreting factories), resident memory T cells, and specialized macrophages that patrol the air spaces. These cells produced a mix of helpful signaling proteins—such as interferon‑γ, IL‑17A, and IL‑4—that together supported bacterial clearance and balanced inflammation.
Testing Protection Under Tough Conditions
These immune changes translated into striking real‑world protection. Mice that received only the alum‑bound OMV shots survived many plague challenges, but a fraction died after the very largest bacterial doses. Mice that also received the intranasal LcrV spike survived even the most extreme lung infections, including from strains lacking a major capsule component and in older animals with an iron‑overload condition that normally makes Yersinia infections worse. In these “prime‑spike” mice, bacteria were almost completely absent from lungs, liver, and spleen within days. When the scientists neutralized key T‑cell signaling molecules, or blocked the movement of lymphocytes from lymph nodes to tissues, protection dropped sharply, showing that the resident lung cells generated by the regimen are essential. Transferring these resident B and T cells into other mice directly improved their survival, underscoring that the cells themselves, not just circulating antibodies, provide the added shield.
Long-Lasting Lung Memory and Future Promise
The team also asked whether a nasal booster given many months after the shots could still awaken lung immunity. When the intranasal LcrV dose was delayed for about six months, it again sharply boosted antibodies and resident memory cells in the lungs and restored full protection against a heavy pneumonic plague challenge, while animals that only had the intramuscular shots were partly unprotected. This suggests that people vaccinated earlier with a similar shot could, in principle, receive a simple nasal top‑up during an outbreak to quickly refresh their lung defenses. Although these experiments were done only in mice, and human trials will require more animal models first, the work points toward vaccine strategies that combine the practicality of standard injections with the powerful, localized protection of mucosal immunity.
What This Means for People
For a layperson, the central message is that where a vaccine sends your immune defenders can matter as much as how many it creates. By first training the immune system with a safe injection and then guiding a key plague protein directly to the nose, this two‑step approach teaches the lungs themselves to remember and repel plague bacteria. If similar strategies work in humans, they could offer a rapid, long‑lasting way to guard against pneumonic plague—and may inspire new vaccines that better protect our airways against a range of dangerous respiratory germs.
Citation: Majumder, S., Das, S., Saqib, M. et al. Intranasal unadjuvanted LcrV boosts parental Yersinia OMV primed lung immunity against pneumonic plague in mice. Nat Commun 17, 1624 (2026). https://doi.org/10.1038/s41467-026-68334-2
Keywords: pneumonic plague, mucosal vaccine, intranasal booster, lung immunity, Yersinia pestis