Clear Sky Science · en
Spatiotemporally ordered topological transformation in layered double hydroxides enables synergistic mineralization of AsIII/Cd2+
Cleaning up two stubborn toxins at once
Arsenic and cadmium are among the most worrying toxic metals in drinking water and soils worldwide. They damage organs, increase cancer risk and are notoriously hard to remove, especially when they appear together. This study describes a new mineral-like material that can pull both pollutants out of water and soil far more efficiently than existing methods, and even works better when the two toxins are present at the same time.
Why arsenic and cadmium are so difficult
Arsenic and cadmium behave very differently in water. The most mobile form of arsenic, called arsenite, is uncharged and slips easily through filters, while cadmium carries a positive charge and clings strongly to many mineral surfaces. In most cleanup materials, cadmium rushes in first and occupies the key reactive spots, blocking arsenite from attaching or being converted into a safer form. That means current technologies often remove one metal at the expense of the other, forcing engineers to accept trade-offs or use complex, multi-step treatments.
A shape-shifting mineral sponge
The researchers tackled this problem by redesigning a class of materials known as layered double hydroxides—minerals made of positively charged sheets with water and ions sandwiched in between. By heating these minerals, they created a related form called a layered double oxide that is full of atomic-scale defects and is eager to react with water. When the oxide is placed in water, it rapidly soaks up water molecules throughout its entire volume, creating plentiful hydroxyl groups (reactive –OH sites) instead of just decorating its outer surface. These bulk reactive sites act like a three-dimensional sponge for metal ions rather than a thin skin, dramatically boosting the capacity for capturing pollutants.
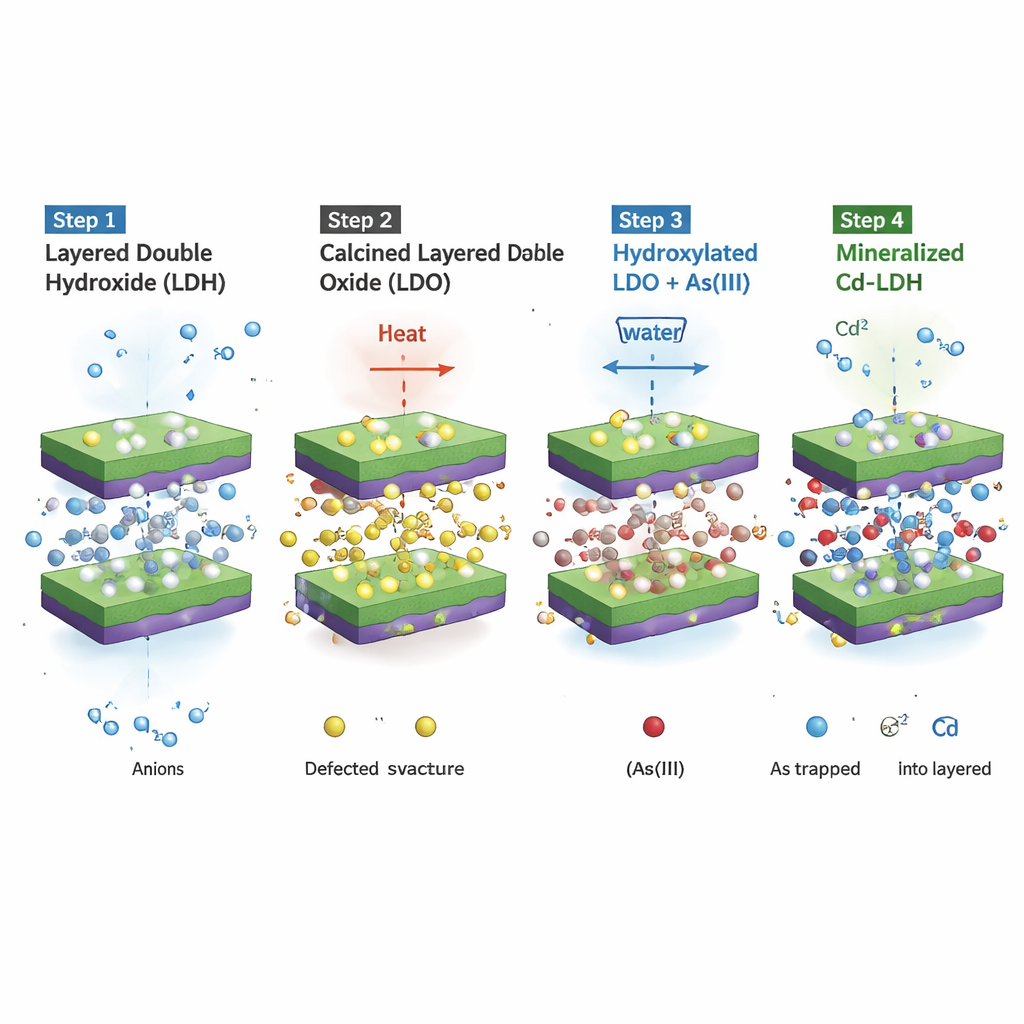
Turning arsenic and cadmium into helpers, not competitors
In tests where arsenite and cadmium were both present, the new material, made from magnesium and manganese (MgMn-LDO), captured up to about 822 milligrams of arsenic and 1,896 milligrams of cadmium per gram of material—several times better than the best previously reported sorbents. Surprisingly, the two pollutants stopped competing and began helping each other. The presence of cadmium sped up arsenite removal so much that the process reached equilibrium in minutes instead of hours, with reaction rates increasing by roughly 181-fold compared with arsenic alone. The material could clean contaminated water down to or below World Health Organization guidelines, even when starting from relatively high pollution levels, and it performed well in both laboratory solutions and real mining wastewater and soils.
A four-step internal rearrangement
The key lies in a carefully ordered series of internal transformations that unfold inside each particle. First, heating turns the original layered hydroxide into a defect-rich oxide. Second, contact with water drives “bulk hydroxylation,” filling the material with water-derived –OH groups and priming it for reaction. Third, arsenite arrives and is oxidized on manganese sites to a less toxic, negatively charged arsenate form; at the same time, electrons flow to manganese, and the structure “remembers” and rebuilds its original layered arrangement. In this rebuilt state, arsenic is tucked between the layers, strongly locked in place. Only after this does the fourth step occur: cadmium begins to swap in for magnesium atoms within the layers, in a process similar to natural substitutions in geological minerals, creating a more stable, mineralized final form that resists leaching.
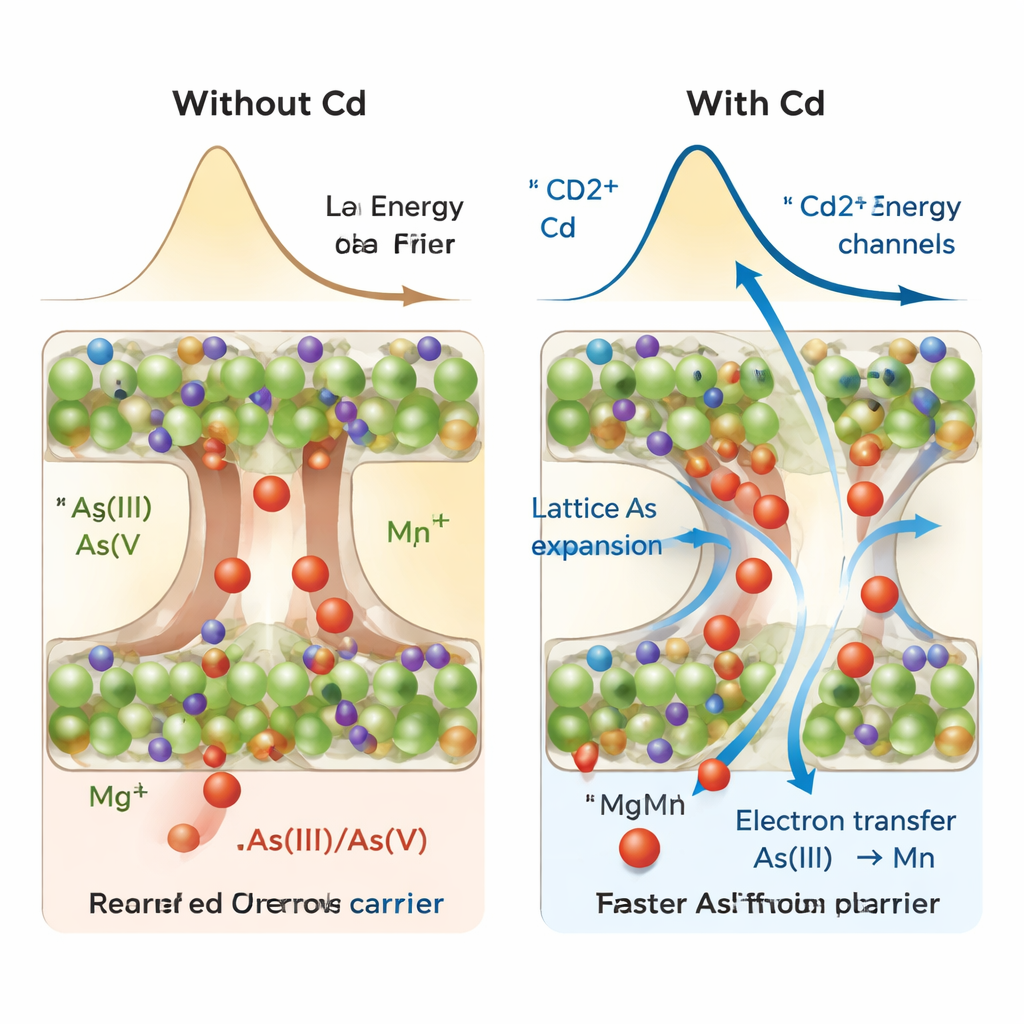
How cadmium accelerates arsenic trapping
This atom-swapping by cadmium does more than just secure cadmium itself. Because cadmium ions are slightly larger than magnesium, their substitution expands the crystal lattice and widens diffusion channels within the material. Computer simulations and spectroscopy experiments show that this expansion lowers the energy barrier for arsenic species to move deeper into the structure along certain pathways, while also slightly weakening specific metal–oxygen bonds. That makes it easier for electrons to transfer from arsenite to manganese and for arsenic to be converted and locked away between layers. In short, cadmium reshapes the internal architecture so arsenic can migrate and be immobilized more rapidly and thoroughly.
From lab discovery to real-world cleanup
Because the material is made from relatively common elements using a simple heating step, it can be produced on at least kilogram scales. Field tests on mining wastewater and highly contaminated industrial soils showed large drops—often around 90% or more—in both arsenic and cadmium that met irrigation or drinking-water-related standards. For non-specialists, the take-home message is that the authors have created a smart, adaptable mineral that rearranges itself in time and space so that arsenic is neutralized first and cadmium is then built into the structure. This clever ordering turns two dangerous metals into mutual allies in their own capture, pointing the way toward more effective and practical cleanup of complex metal pollution.
Citation: Zheng, M., Du, H., Cao, X. et al. Spatiotemporally ordered topological transformation in layered double hydroxides enables synergistic mineralization of AsIII/Cd2+. Nat Commun 17, 1619 (2026). https://doi.org/10.1038/s41467-026-68326-2
Keywords: arsenic removal, cadmium pollution, water purification, layered double hydroxide, heavy metal remediation