Clear Sky Science · en
Glycerol 3-phosphate acyltransferase exacerbates α-synuclein-induced toxicity by increasing lipid peroxidation
Why fats in the brain matter for Parkinson’s
Parkinson’s disease is usually described as a problem with a protein called alpha‑synuclein that clumps up and harms brain cells controlling movement. This study shows that brain fats—especially the way they are made and damaged—play a surprisingly powerful role in how toxic alpha‑synuclein becomes. By uncovering a fat‑making enzyme that worsens nerve cell injury, the work points to a new, druggable pathway that could complement existing efforts to tackle Parkinson’s at its roots.
A protein that misbehaves in Parkinson’s
People with Parkinson’s gradually lose dopamine‑producing nerve cells in a deep brain region that coordinates movement. Inside these dying cells, scientists often find dense deposits called Lewy bodies, packed with the protein alpha‑synuclein. In rare families, mutations or extra copies of the alpha‑synuclein gene directly cause Parkinson’s, but common genetic variants in that gene only modestly raise risk. This suggests that other genes and pathways modify how harmful alpha‑synuclein becomes. Increasingly, evidence points to lipids—fats and fat‑like molecules that form cell membranes and energy stores—as key partners in both alpha‑synuclein clumping and nerve cell death.
Finding a powerful lipid enzyme in fruit fly models
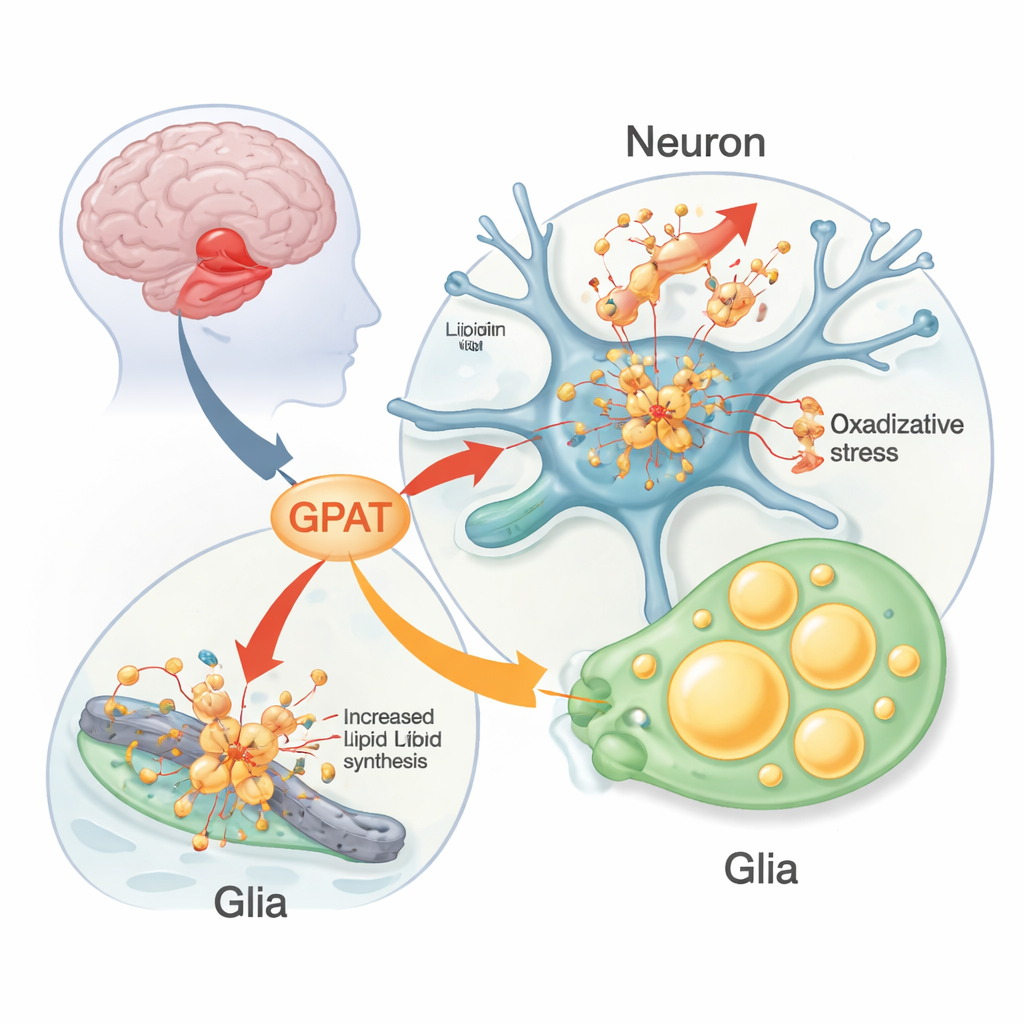
The researchers used fruit flies engineered to make human alpha‑synuclein in their nervous system as a living test bed. These flies develop Parkinson‑like problems: loss of dopamine‑producing neurons, difficulty climbing, and disrupted daily activity rhythms. The team systematically altered fly versions of human Parkinson’s risk genes to see which ones changed alpha‑synuclein’s impact. One stood out: a gene called mino, which makes a mitochondrial form of the enzyme glycerol 3‑phosphate acyltransferase (GPAT). GPAT sits at the entry point of building phospholipids and triglycerides—the lipids that form membranes and fat droplets. When the team reduced mino activity in neurons, alpha‑synuclein flies kept more of their dopamine neurons and moved better for longer; boosting mino had the opposite, harmful effect.
Damaged fats, stressed mitochondria, and glial fat droplets
Diving deeper, the scientists found that GPAT influenced how much oxidative damage accumulated in brain lipids. In alpha‑synuclein flies kept at higher temperature (which worsens disease features), lipid peroxidation—chemical “rusting” of fats—rose in brain membranes. Lowering mino cut this damage, while overexpressing it increased it; without alpha‑synuclein, changing mino had little effect. Markers of cell death in the visual brain region mirrored this pattern. The team also observed a striking build‑up of lipid droplets—tiny fat storage spheres—not in neurons themselves but in neighboring glial cells. These droplets expanded with age in alpha‑synuclein flies and were modulated by enzymes that build or break down triglycerides, highlighting an active metabolic partnership between neurons and glia under stress.
Metabolic rewiring and alpha‑synuclein clumping
Metabolite measurements from fly brains revealed that alpha‑synuclein expression was linked to a bottleneck in the cell’s energy‑producing cycle: citrate and isocitrate, two tricarboxylic acid (TCA) intermediates, accumulated strongly, while downstream steps shifted more modestly. Lactate levels also rose, consistent with ramped‑up glycolysis. At the same time, detailed lipid profiling showed changes in the balance of membrane phospholipids and their fatty acid makeup, favoring species more prone to oxidative damage. When the team reduced several GPAT enzymes—mino in mitochondria and related enzymes on the endoplasmic reticulum—alpha‑synuclein still accumulated, but its tendency to form higher‑order oligomers (multi‑protein bundles) dropped, and mitochondria showed fewer signs of reactive oxygen stress and “aging.”
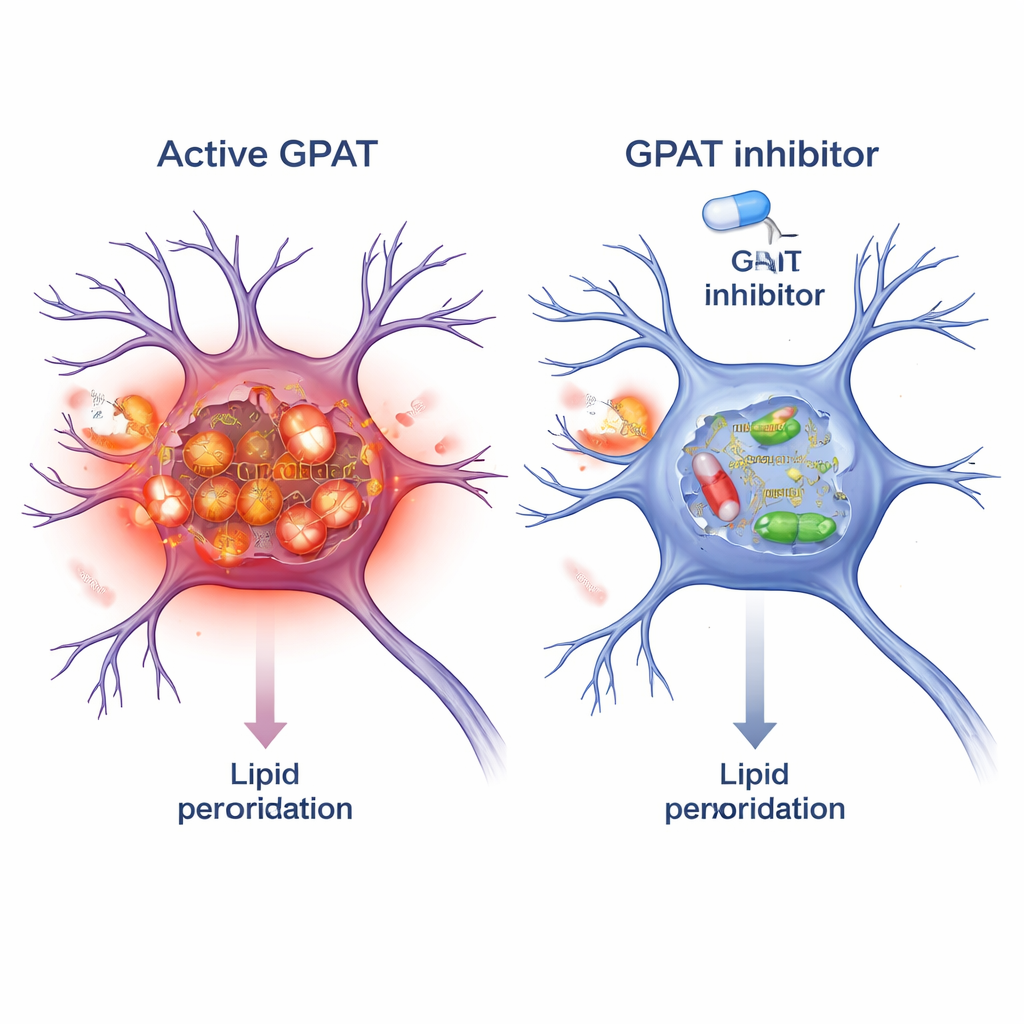
Blocking GPAT as a protective strategy
Because GPAT is an enzyme, it can be targeted by small‑molecule drugs. The researchers tested FSG67, an existing GPAT inhibitor originally developed for obesity and diabetes. In alpha‑synuclein flies, adding FSG67 to the food reproduced the benefits of genetic GPAT knockdown: improved movement, better survival of dopamine neurons, fewer damaging alpha‑synuclein oligomers, and reduced mitochondrial oxidative stress. To see if this concept carried over to mammals, they treated mouse brain neurons in culture with pre‑formed alpha‑synuclein fibrils, which seed toxic aggregates. Co‑treatment with FSG67 reduced the accumulation of phosphorylated alpha‑synuclein, and lowered multiple independent markers of lipid peroxidation within these neurons.
What this means for people with Parkinson’s
In plain terms, this work shows that how the brain handles fats can dial alpha‑synuclein toxicity up or down. When GPAT is highly active, more vulnerable lipids are built into membranes and storage droplets, making them easier to oxidize; this damaged fat environment appears to favor harmful forms of alpha‑synuclein and strain mitochondria, the cell’s power plants. Dialing GPAT back—either genetically or with a drug—shifts the balance toward less lipid “rust,” fewer toxic protein assemblies, and healthier neurons. While these findings are early and come from flies and cultured mouse cells, they spotlight lipid metabolism, and GPAT in particular, as a promising new angle for Parkinson’s therapies that could complement strategies directly targeting alpha‑synuclein.
Citation: Ren, M., Lim, G.G.Y., Tang, W. et al. Glycerol 3-phosphate acyltransferase exacerbates α-synuclein-induced toxicity by increasing lipid peroxidation. Nat Commun 17, 1618 (2026). https://doi.org/10.1038/s41467-026-68325-3
Keywords: Parkinson’s disease, alpha-synuclein, lipid peroxidation, GPAT inhibitor, neurodegeneration