Clear Sky Science · en
Growth of non-layered 2D transition metal nitrides enabled by transient chloride templates
Why ultrathin metals matter
Electronics, batteries, and future quantum devices all rely on materials only a few atoms thick. Most of today’s “2D materials,” like graphene, are naturally layered and therefore relatively easy to peel into sheets. But some of the most promising compounds for catalysis, data storage, and high‑power electronics—transition metal nitrides—do not come in layers. This paper reports a way to reliably grow these stubborn materials as ultra‑thin, sheet‑like crystals, opening a door to new technologies that need strong, flexible, and magnetic films only atoms thick.
Turning a weakness into a strength
Transition metal nitrides are famous for being hard, heat‑resistant, and sometimes superconducting, but those same strong metal–nitrogen bonds lock atoms together in all directions. That three‑dimensional bonding makes it extremely difficult to make them as flat, two‑dimensional flakes. Earlier methods either etched away complex precursors or relied on salts whose atomic lattices happened to match the desired nitride. These routes worked only for a few compositions and often left unwanted chemical groups on the surface, masking the nitrides’ true behavior.
A clever role for fragile chlorides
The authors realized that transition metal chlorides—salts such as iron chloride or cobalt chloride—could act as temporary, or “transient,” scaffolds. On paper, these chlorides should convert into metal nitrides with relatively little energy compared with oxides or sulfides, and many of them naturally stack in layers, just like graphite. The catch is that they are volatile and unstable at the high temperatures needed to form nitrides, so in a conventional furnace they simply evaporate before they can transform. The team’s key insight was to briefly stabilize these chlorides just long enough to grow them as thin layers on a cool surface, and then very rapidly expose them to the hot, nitrogen‑rich environment needed for conversion.
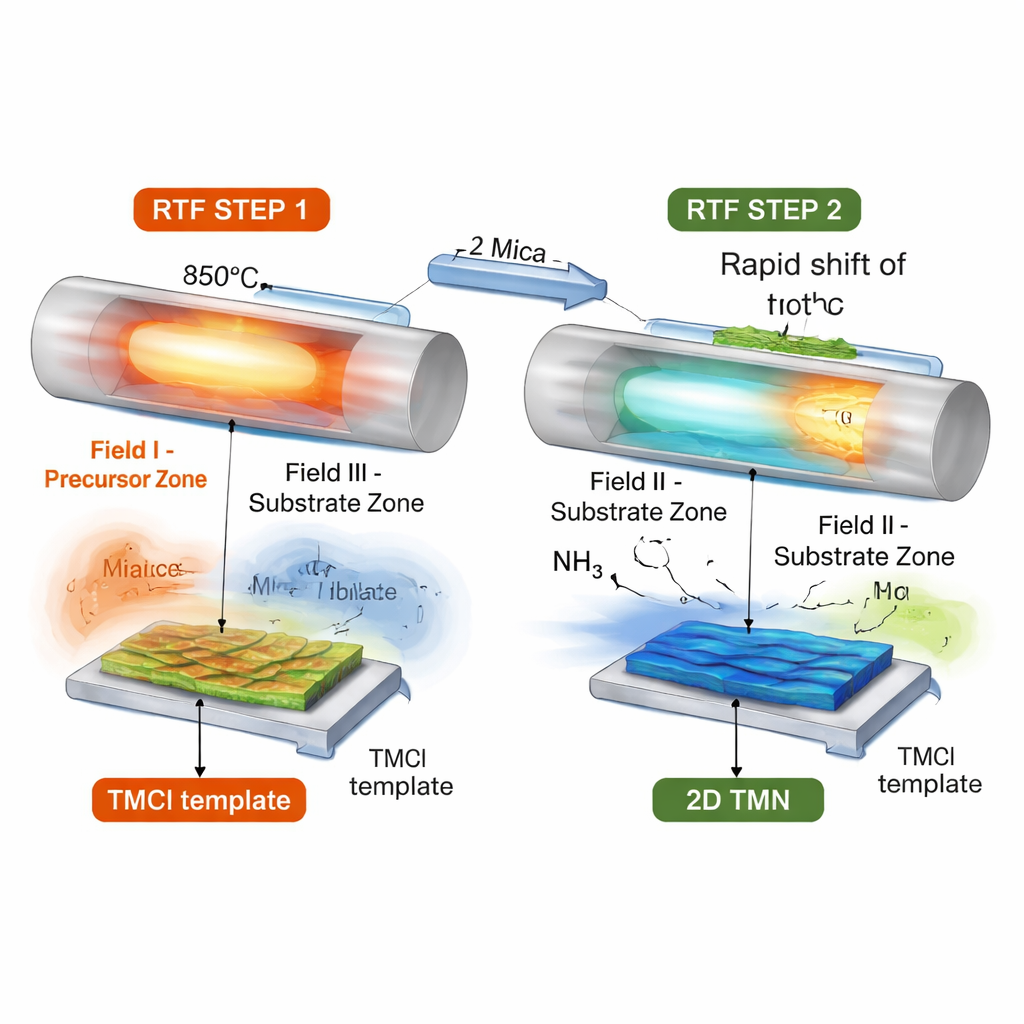
Reversing the heat to make sheets
To pull this off, the researchers designed a “reverse‑thermal‑field” chemical vapor deposition process. In the first step, a movable furnace heats the metal chloride source while keeping the receiving mica substrate relatively cool. This encourages flat, layered chloride crystals to grow on the substrate. In the second step, the hot region of the furnace is quickly shifted so that the substrate, not the source, is suddenly at high temperature, and a flow of ammonia gas is introduced. Within seconds, the fragile chloride templates are converted in place into ultra‑thin transition metal nitride sheets, while the source region cools down to limit further evaporation and contamination. Because many different metal chlorides share similar behavior, the same basic recipe works across a wide range of elements.
Building a library of atom‑thin nitrides
Using this strategy, the team produced fifteen distinct two‑dimensional materials: seven made from a single metal and eight alloys containing two to four different metals. Examples include VN, CrN, MnN, Fe2N, CoN, and several forms of NiN, as well as mixed compounds such as Co–Ni–N and Cr–Fe–Co–Mn–N. Microscopy and electron diffraction measurements show that these flakes are single crystals with well‑ordered atoms and clean compositions, often just over a nanometer thick and tens of micrometers across. Their shapes—hexagons or rectangles—can be tuned by growth temperature, which changes the structure of the original chloride template. Chemical mapping confirms that in the alloy flakes, different metal and nitrogen atoms are evenly mixed, rather than separating into patches.
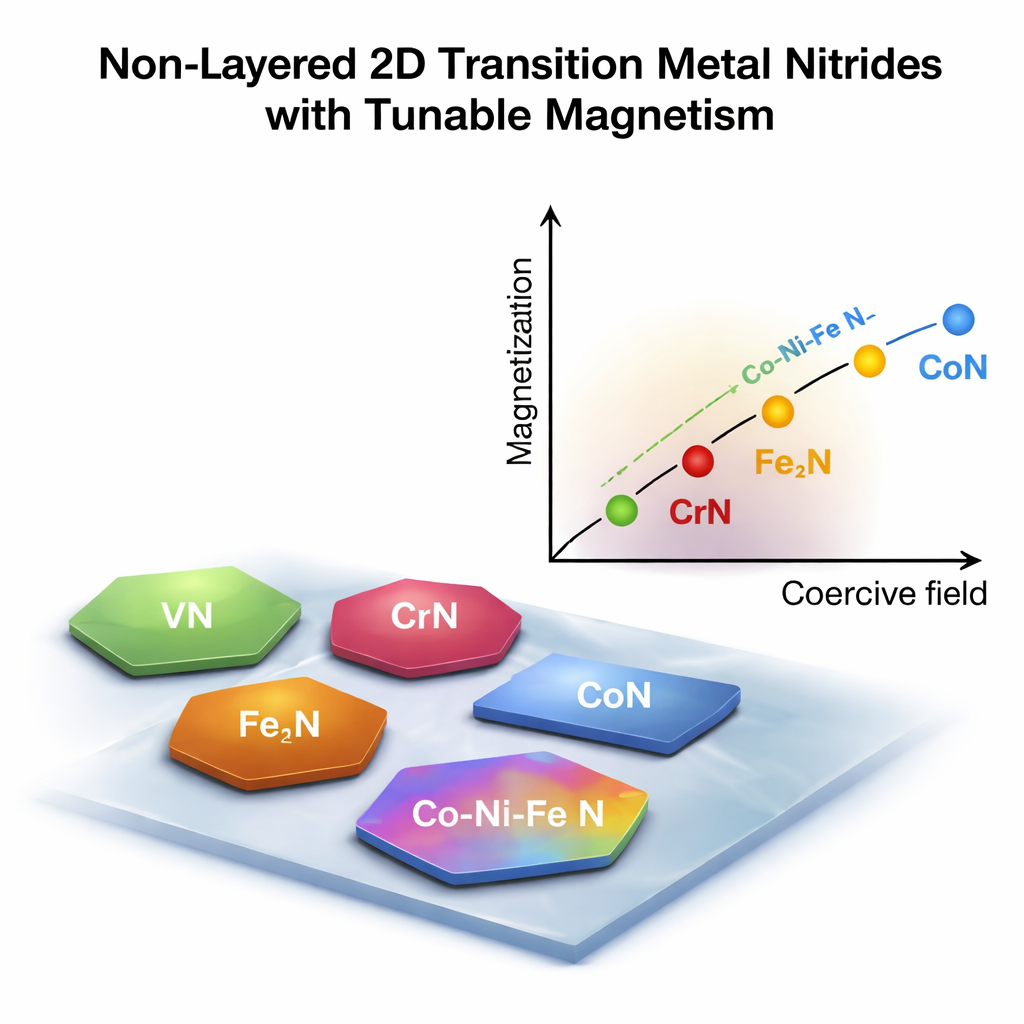
Dialing in magnetic behavior
Because many transition metal nitrides are magnetic, the authors next explored how magnetism changes when they are thinned down and alloyed. Using magnetic force microscopy and ultra‑sensitive magnetization measurements, they found that two‑dimensional nitrides can behave very differently from their bulk counterparts. Some, like certain cobalt‑rich compounds, act as hard magnets with large coercive fields; others are softer or even antiferromagnetic, where neighboring atomic spins oppose each other. By adjusting which metals are combined in an alloy, the team could strengthen or weaken the overall magnetic response and shift materials along a spectrum from soft to hard magnets. This tunability is crucial for applications ranging from spin‑based electronics to tiny magnetic sensors.
What this means going forward
In simple terms, the researchers have invented a general recipe for turning a wide variety of tough, three‑dimensional nitride compounds into atomically thin, high‑quality sheets. By briefly using fragile chlorides as templates and rapidly reversing the heat in the furnace, they avoid the usual roadblocks that made these materials hard to access in 2D form. The resulting films are not only structurally clean but also show a rich range of magnetic behaviors that can be adjusted through composition. This work significantly expands the family of available two‑dimensional materials and lays the groundwork for future devices that exploit the strength, stability, and controllable magnetism of ultrathin transition metal nitrides.
Citation: He, L., Wang, J., Cai, Z. et al. Growth of non-layered 2D transition metal nitrides enabled by transient chloride templates. Nat Commun 17, 1615 (2026). https://doi.org/10.1038/s41467-026-68321-7
Keywords: two-dimensional materials, transition metal nitrides, chemical vapor deposition, magnetism, materials synthesis