Clear Sky Science · en
Structure of SHOC2-KRAS-PP1C complex reveals RAS isoform-specific determinants and insights into targeting complex assembly by RAS inhibitors
How Cells Decide When to Grow
Our cells rely on an internal wiring system to decide when to grow, divide, or stay quiet. At the heart of this system are RAS proteins, tiny molecular switches that often go wrong in cancer. This study looks deep into how a particular group of proteins—SHOC2, RAS, and PP1C—join together to flip a key growth switch, and how modern cancer drugs might be tuned to block that process more effectively and prevent tumors from outsmarting treatment.
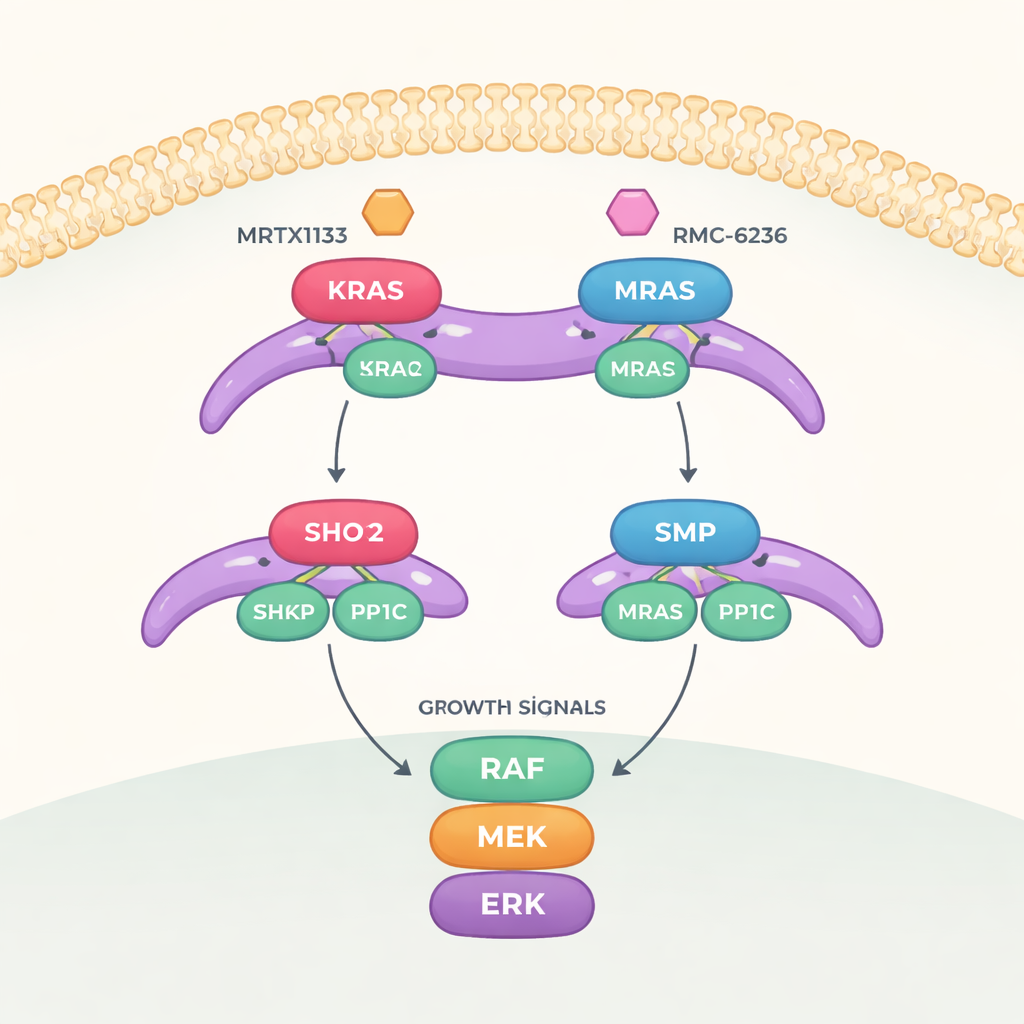
A Growth Switch with Three Critical Parts
The work focuses on a signaling route called the MAPK pathway, which carries growth messages from the cell surface down to the nucleus. To turn this pathway fully on, another protein called RAF must be switched from an “off” state to an “on” state. That switch is controlled by a three-part complex made of SHOC2 (a scaffold that holds things together), PP1C (an enzyme that removes a phosphate tag), and an active RAS protein. When assembled, this trio removes a specific phosphate mark from RAF, freeing it to activate the downstream growth cascade.
Why a Lower-Affinity Partner Still Matters in Cancer
There are several kinds of RAS proteins. MRAS forms a very tight complex with SHOC2 and PP1C, while the better-known cancer drivers KRAS, HRAS, and NRAS form weaker versions of the same complex. Surprisingly, cancer cells driven by mutant KRAS, HRAS, or NRAS turn out to be strongly dependent on SHOC2, even though their complexes are less stable. Large genetic screening datasets show that many tumors with certain RAS mutations—especially those affecting key positions like Q61 and G13—are highly sensitive to losing SHOC2, meaning this assembly is essential for their survival.
Revealing the Shape of the KRAS Complex
Until now, scientists had only seen a high-resolution structure of the tight MRAS-based complex. The weaker KRAS version fell apart too easily to capture. In this study, the authors used disease-linked mutations that subtly strengthen contacts between the components to stabilize the SHOC2–KRAS–PP1C complex long enough to image it with cryo–electron microscopy. They found that the overall architecture closely matches the MRAS complex, but KRAS lacks several small structural features that MRAS uses to grip SHOC2 and PP1C more firmly. As a result, the KRAS assembly buries less contact area and forms fewer bonds, explaining why it is intrinsically less stable, even though it remains highly important in tumors where KRAS is overactive.
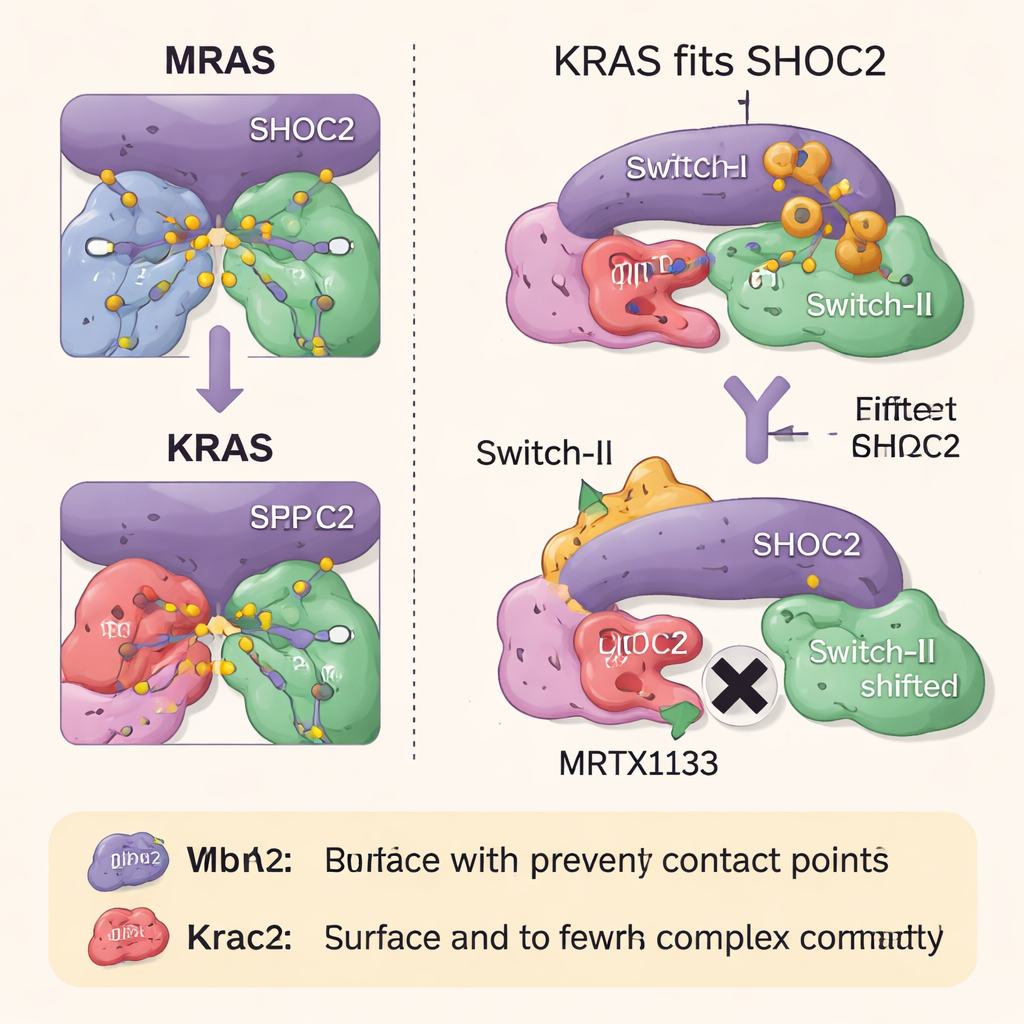
How RAS Drugs Block Complex Assembly
The team then explored how two classes of RAS-targeting drugs influence this three-part complex. MRTX1133, designed to bind a pocket near a flexible region of KRAS known as Switch-II, locks this region into a shape that clashes with the SHOC2 surface. This strongly prevents new SHOC2–KRAS–PP1C complexes from forming, though it is less effective at breaking apart ones that already exist. A second drug, RMC-6236, operates together with a helper protein (cyclophilin A) and also blocks the same KRAS surfaces needed for SHOC2 binding. Neither drug, however, naturally binds MRAS, so the high-affinity MRAS complex stays intact and can help reawaken the growth pathway when KRAS is inhibited.
Toward Dual Targeting to Prevent Resistance
To test whether dual targeting is possible, the researchers engineered a version of MRAS whose Switch-II pocket was altered so that MRTX1133 could now bind. This mutant still formed a strong complex with SHOC2 and PP1C, but the drug now blocked its assembly and reduced its ability to switch on RAF in test-tube assays. Together with data on SHOC2-binding compounds, these results show that it is realistic to design drugs that shut down both the KRAS-based and MRAS-based complexes. To a layperson, the key message is that cancer cells use multiple, closely related switches to keep growth signals flowing, especially when one switch is drugged. By understanding the precise shapes and contacts within these protein assemblies, researchers can now plan therapies that hit both backup routes at once, making it harder for RAS-driven cancers to adapt and resist treatment.
Citation: Bonsor, D.A., Finci, L.I., Potter, J.R. et al. Structure of SHOC2-KRAS-PP1C complex reveals RAS isoform-specific determinants and insights into targeting complex assembly by RAS inhibitors. Nat Commun 17, 1614 (2026). https://doi.org/10.1038/s41467-026-68319-1
Keywords: RAS signaling, KRAS inhibitors, MAPK pathway, protein complexes, drug resistance