Clear Sky Science · en
Injectable hydrogels for osteomyelitis treatment induce metabolic reprogramming for protection against reinfection
Turning the Body Into Its Own Infection Fighter
Bone infections, especially those caused by antibiotic‑resistant bacteria, are notoriously hard to cure and often come back even after long courses of drugs and surgery. This study describes an injectable gel that is placed directly inside infected bone, where it not only kills germs but also trains the body’s innate defenses and helps the bone rebuild itself. The work points toward a future in which materials do more than deliver antibiotics—they also coach our immune system to prevent relapse.
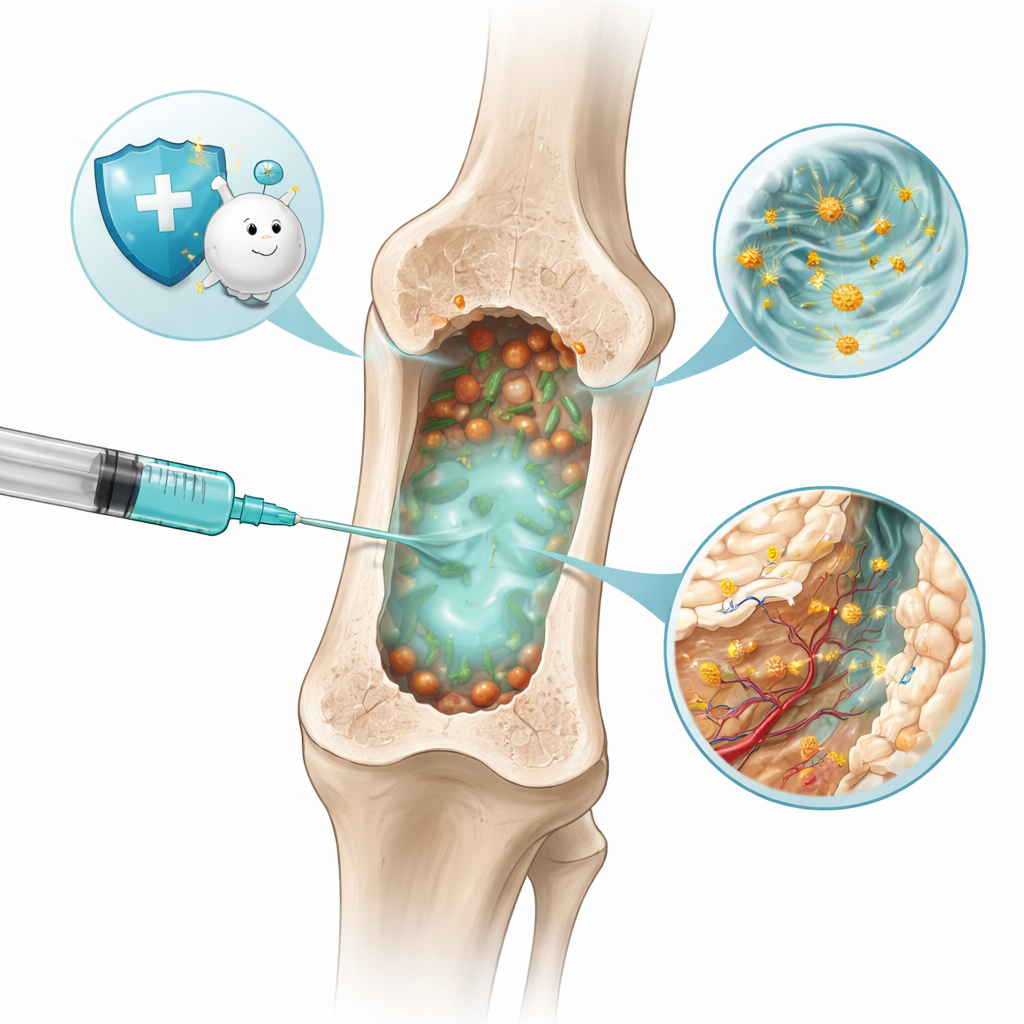
A Sticky Gel That Forms Inside the Bone
The researchers created a soft material called a hydrogel that can be injected as a liquid and then quickly firms up inside the bone marrow cavity, the spongy center of bones where many immune cells are born. Their gel, named GaCuVan&HACHO‑BSA, is built from two interlocking networks. One network combines a natural plant molecule from licorice (glycyrrhizic acid), copper ions, and the antibiotic vancomycin. The other uses a modified form of hyaluronic acid—a sugar found in our joints—linked to a common blood protein. Together these components form a porous, flexible scaffold that flows through a needle, fills irregular spaces, and slowly breaks down over about a month.
Trapping Bacteria and Toxins at the Source
Once inside the infected bone, the hydrogel acts like a smart sponge. Its tiny pores and chemical groups allow it to catch bacteria such as methicillin‑resistant Staphylococcus aureus (MRSA), along with pieces of bacterial cell walls and inflammatory molecules drifting in the infected tissue. Vancomycin on the gel surface helps it grab and then kill the bacteria it captures, while other chemical bonds latch onto bacterial toxins and signaling proteins. Lab tests showed that this gel could remove most bacteria and harmful factors from its surroundings much better than simpler gels, and it remained strong enough to support and fill bone defects as it did so.
Reprogramming Innate Immunity
Beyond acting as a local drug depot, the hydrogel turns the infected marrow into a training ground for front‑line immune cells. Because the gel holds on to bacterial fragments and inflammatory signals, these cues are presented continuously to nearby macrophages and other innate cells instead of being rapidly washed away. In rat experiments, this led to a wave of short‑term inflammation: more white blood cells arrived, and bone‑resident immune cells switched into an activated, bacteria‑fighting state. Deep genetic and metabolic analyses of bone marrow showed that key danger‑sensing pathways were switched on and that cells rewired their energy use toward fast, sugar‑burning metabolism—hallmarks of so‑called “trained immunity,” a form of innate memory that prepares cells to respond more strongly the next time they encounter invaders.
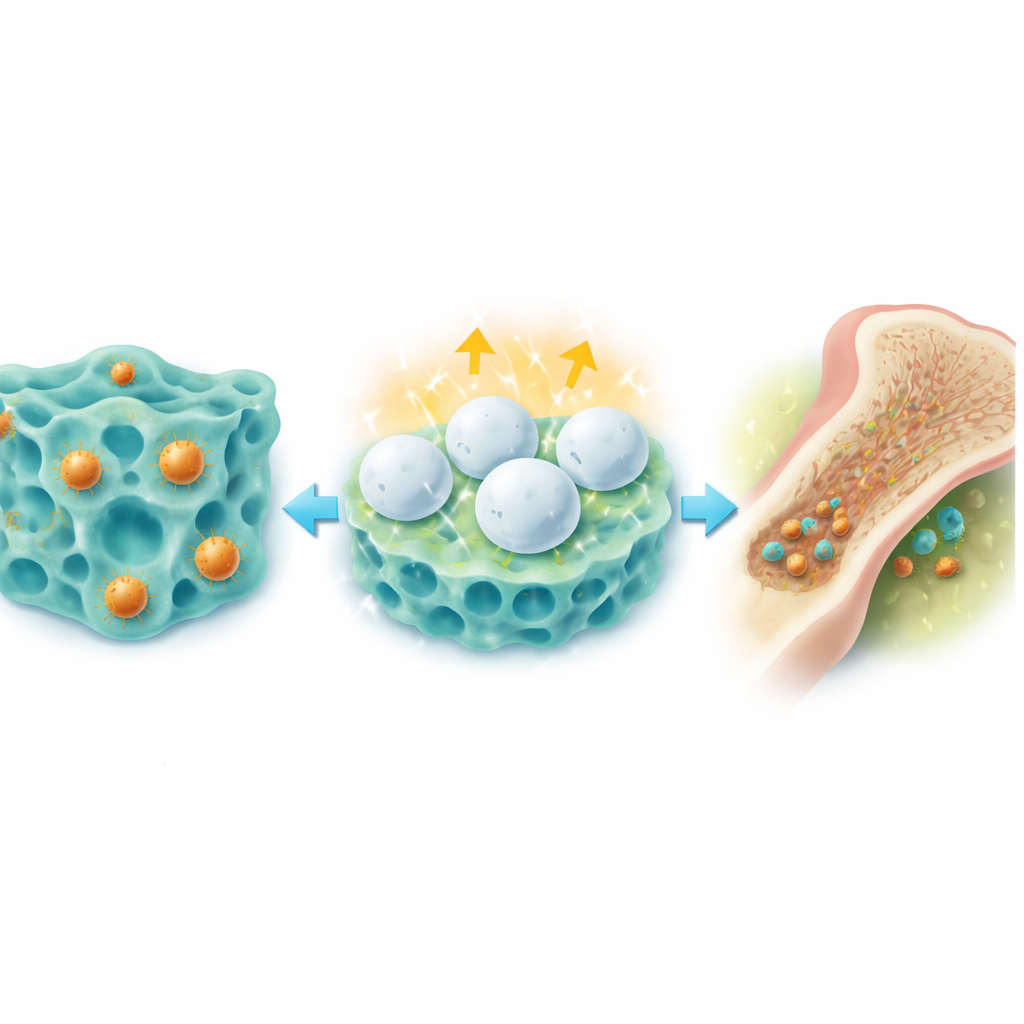
Clearing Infection and Helping Bone Heal
In rats with severe MRSA bone infections, a single in‑bone injection of the hydrogel rapidly reduced bacterial counts in the marrow to nearly zero, while animals treated with standard vancomycin or simpler gels often remained heavily infected. The gel also encouraged the growth of new bone. Copper and the licorice‑derived component both nudged stem cells toward becoming bone‑forming cells, boosting markers of bone growth and increasing bone density and strength in damaged tibias. Imaging and tissue staining over several weeks showed that defects in infected bones of treated animals nearly closed, with organized, healthy new bone replacing the damaged regions.
Protection Against Future Infections
A striking finding was that bones pretreated with the hydrogel resisted later attacks. When rats that had their initial infection cleared were challenged again—either with the same MRSA strain or with different strains and related bacteria—their bones quickly marshaled strong inflammatory responses, limited bacterial growth, and often avoided reinfection altogether. Blocking the cells’ altered sugar metabolism erased this protection, confirming that metabolic “reprogramming” of innate immune cells was essential. The effect worked in normal and aged rats but was weaker in diabetic animals, whose immune systems are already metabolically disturbed.
What This Could Mean for Patients
For someone facing chronic or drug‑resistant bone infection, this kind of injectable gel could one day offer more than a temporary antibiotic boost. By seeding the marrow cavity with a material that captures germs, releases antibiotics and bone‑building ions, and trains innate immune cells to react faster and harder to future threats, doctors might both cure current disease and lower the risk of relapse. While more work is needed—especially in larger animals and people with complex illnesses—the study introduces a new way of thinking about bone infection treatment: using smart materials not just to deliver drugs, but to reshape the local immune and repair environment for long‑term protection.
Citation: Chen, H., Wei, L., Yu, Q. et al. Injectable hydrogels for osteomyelitis treatment induce metabolic reprogramming for protection against reinfection. Nat Commun 17, 1613 (2026). https://doi.org/10.1038/s41467-026-68318-2
Keywords: osteomyelitis, hydrogel therapy, trained immunity, bone regeneration, antibiotic resistance