Clear Sky Science · en
Valence-free open nanoparticle superlattices
Building Crystals from Tiny Building Blocks
Imagine being able to stack particles a thousand times smaller than a grain of sand into perfect, repeating structures—like LEGO bricks at the nanoscale. Such ordered “superlattices” could one day steer light for advanced photonics, improve catalysts, or pack information into ultra‑compact devices. This article reports a simple, broadly applicable recipe for snapping together nanoparticles into open, airy crystal structures that were previously very hard to make.
Why Open Lattices Matter
Crystals do not just exist in nature; they can be engineered. Arranging nanoparticles into specific patterns changes how they interact with light, electricity, and chemicals. In particular, “diamond‑like” and other open cubic lattices are coveted because their repeating voids can create photonic band gaps—ranges of colors that cannot pass through the material—useful for light‑based circuits and sensors. Until now, making these lattices required custom “patchy” particles with carefully placed sticky spots, mimicking the directional bonds of atoms in a diamond crystal. That complexity has limited how easily such materials can be designed and scaled.
A Simple Recipe: Charge and Soft Coats
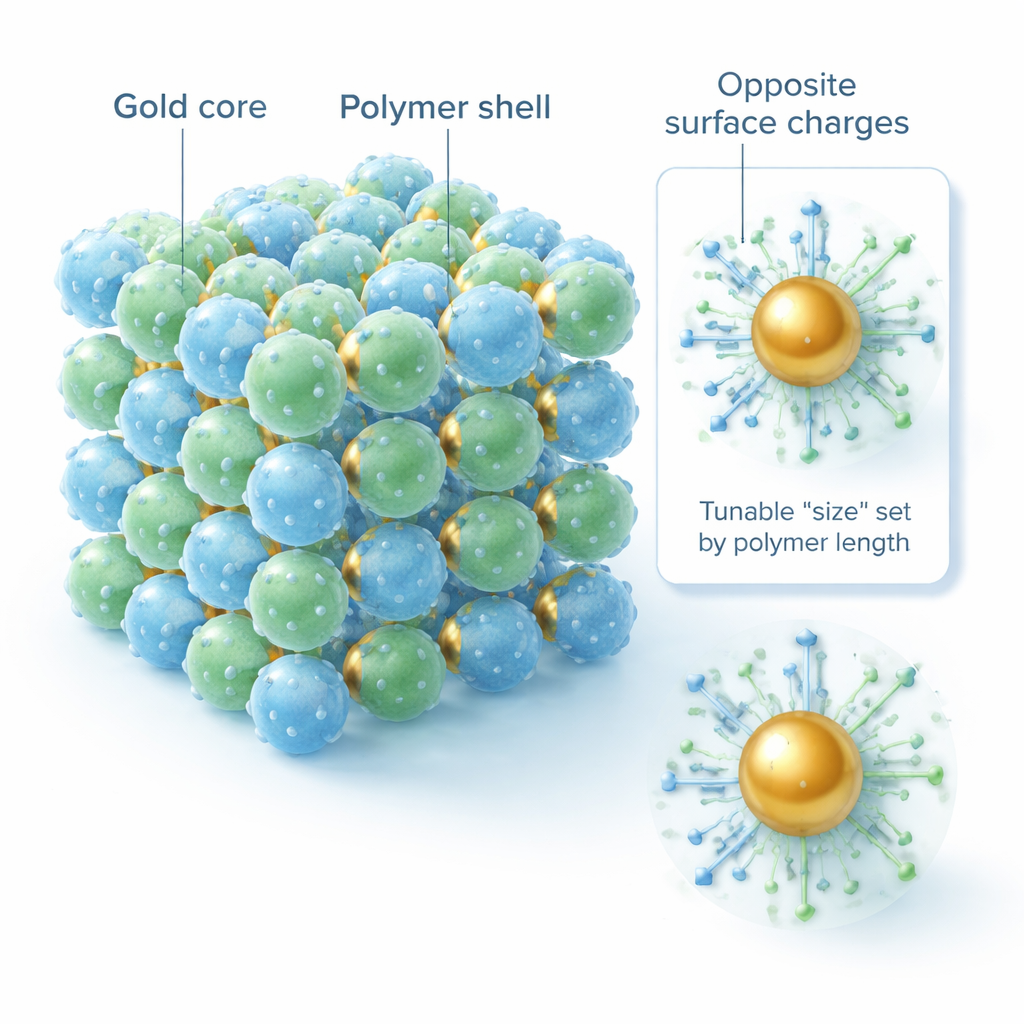
The researchers show that intricate directional bonding is not necessary. Instead, they start with spherical gold nanoparticles and give them soft, water‑loving coats made of polymer chains known as PEG. Some chains end in positively charged groups, others in negatively charged groups. When mixed in water at the right acidity (pH), oppositely charged particles attract each other while like‑charged particles repel one another. By choosing how long each polymer shell is and how strongly it is charged, the team can effectively control both the “size” of the coated particles and how strongly they pull together or push apart.
From Salt Crystals to Diamond‑Like Structures
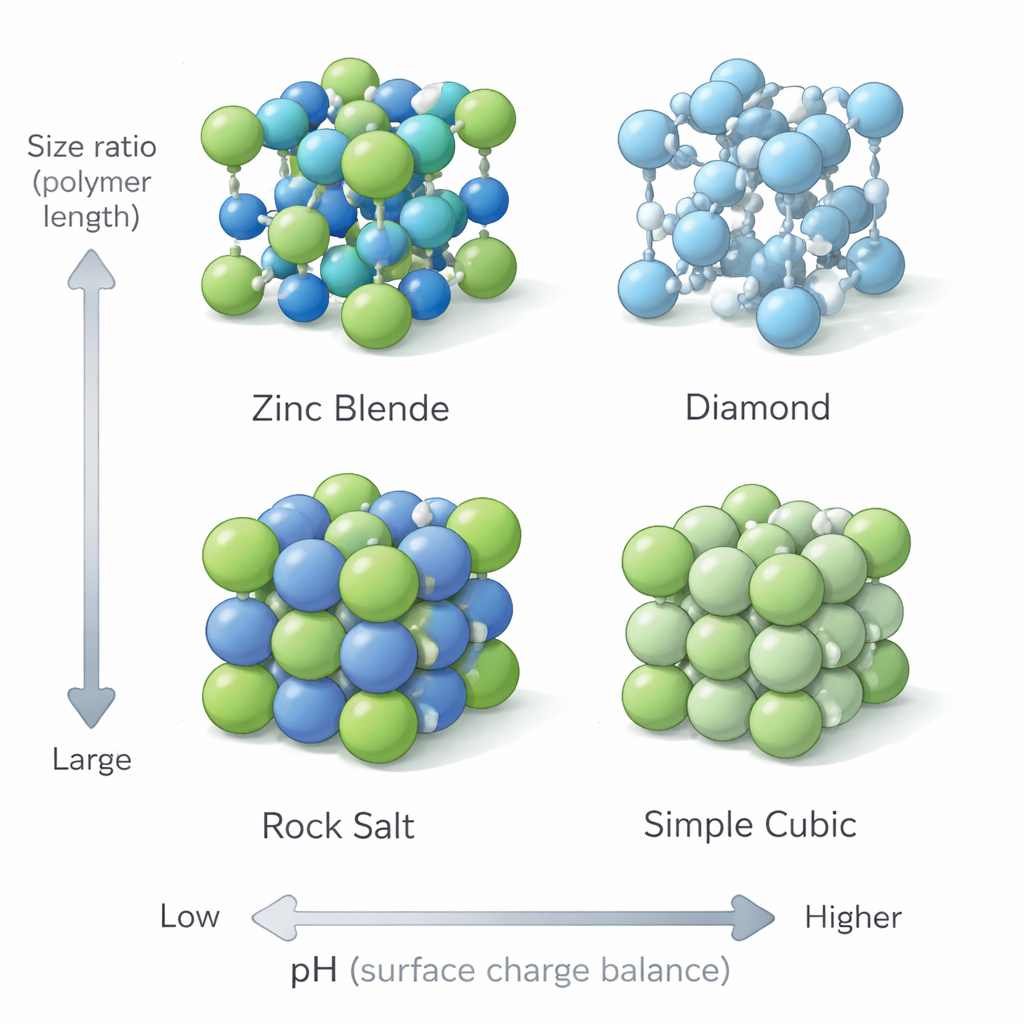
Borrowing ideas from ordinary table salt and other ionic solids, the authors treat the nanoparticles like big, softly coated ions. In such crystals, the pattern of positive and negative ions is largely dictated by their relative sizes. Likewise, the group defines two simple knobs: the ratio of the effective particle sizes, and the ratio of polymer chain lengths with different end groups. By tuning these, they coax mixtures of two nanoparticle types into a wide menu of superlattices: analogs of rock salt, cesium chloride, zinc blende, and even a rare simple cubic structure. When the two nanoparticle cores are the same size but their soft shells are adjusted appropriately, the zinc‑blende arrangement smoothly turns into a diamond‑like lattice—exactly the open, low‑coordination structure that is so valuable for photonic applications.
Watching Particles Assemble and Testing the Rules
To see which structures form, the team shines powerful X‑ray beams through the nanoparticle suspensions and analyzes the resulting diffraction patterns. The positions of the peaks reveal how the particles are arranged, and the sharpness of the peaks shows how well ordered the crystals are. By systematically varying particle size, polymer length, and pH, they map out a phase diagram that links simple, measurable parameters to the final lattice type. Computer models and detailed molecular simulations back up these observations. The simulations show how opposite charges at the ends of the polymer chains can form strong, hydrogen‑bond‑assisted links, while like charges remain separated, reinforcing the formation of orderly, open frameworks.
A General Toolkit for Designer Nanomaterials
In everyday terms, this work provides a straightforward set of “kitchen rules” for cooking up complex nanoparticle crystals: pick two particle types, dress them in oppositely charged polymer coats of chosen lengths, set the acidity, and let them self‑assemble. With just these knobs, the same strategy can be applied to many kinds of nanoparticles beyond gold, and the spacing between particles—and thus their optical behavior—can be adjusted simply by changing core size or polymer weight. This valence‑free approach opens a practical route to building tailor‑made nanostructured materials for technologies ranging from light‑manipulating devices to advanced catalysts.
Citation: Nayak, B.P., Wang, W., Kakkar, P. et al. Valence-free open nanoparticle superlattices. Nat Commun 17, 1611 (2026). https://doi.org/10.1038/s41467-026-68316-4
Keywords: nanoparticle superlattices, self-assembly, photonic materials, polymer-coated nanoparticles, colloidal crystals