Clear Sky Science · en
Reversible lipid-mediated pH-gating of connexin-46/50 by cryo-EM
Why tiny cell channels matter for health and disease
Every second, our cells exchange electrical signals and small molecules through microscopic tunnels called gap junctions. These channels help the heart beat in sync, the brain clear toxic by-products, and the eye lens stay transparent. When conditions inside cells turn acidic, as happens during stroke, heart attack, or cataract formation, many of these channels shut down—but how they sense and respond to acidity has been a long-standing mystery. This study uses high‑resolution cryo‑electron microscopy to reveal, almost atom by atom, how specific channels in the eye lens close in response to acid, and how this process can fully reverse.
Cellular tunnels that keep tissues in sync
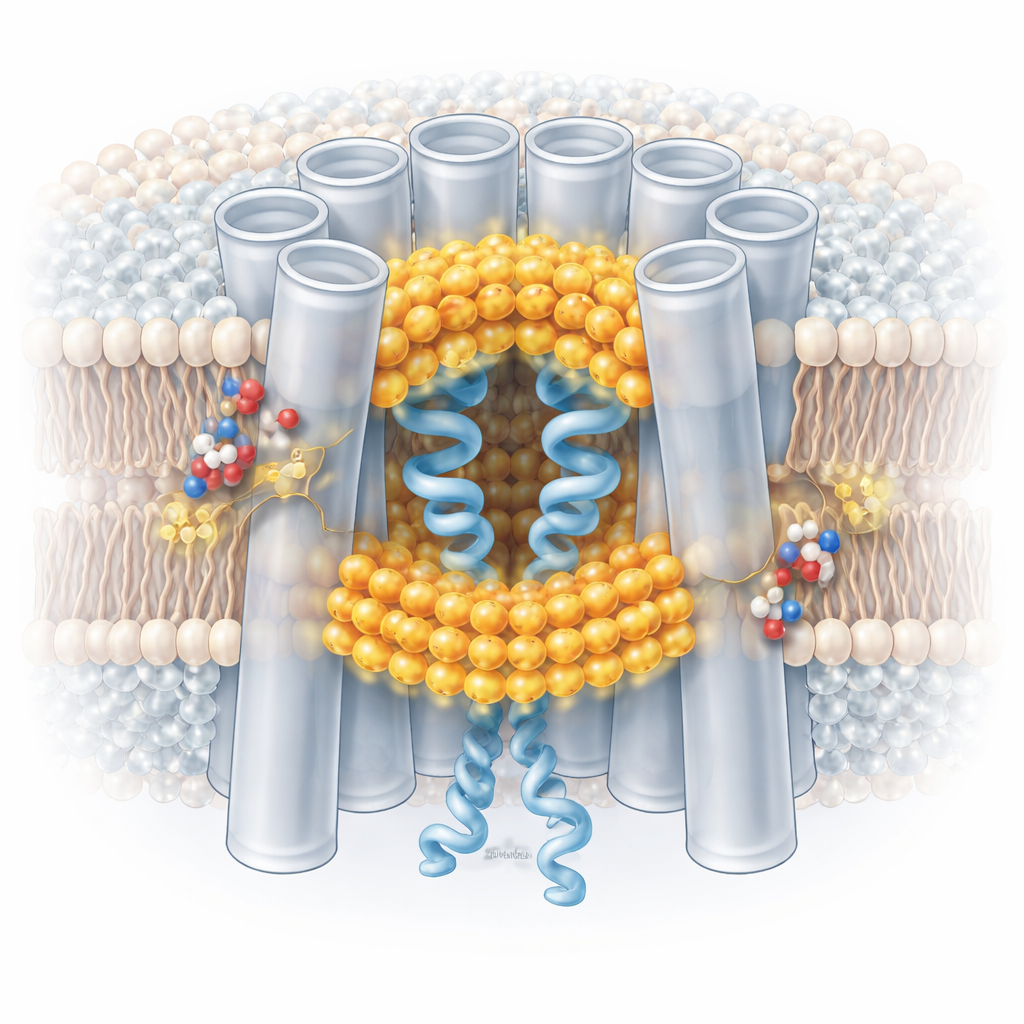
Gap junctions are built from proteins called connexins, which assemble into ring‑shaped channels spanning the membranes of two neighboring cells. This work focuses on a pair of closely related connexins, connexin‑46 and connexin‑50, that form channels in the lens of the eye. These channels allow ions and small molecules to flow directly between cells, helping maintain lens clarity and overall tissue health. When cells are stressed, however, the same channels can become pathways for damaging signals. To protect tissues, gap junctions close when the cell interior becomes more acidic. Understanding the fine‑scale mechanism of this “pH‑gating” is important for deciphering how tissues survive stress and why certain mutations lead to diseases such as cataracts.
Seeing channels move with cryo‑EM
The researchers purified native connexin‑46/50 channels from aged sheep lenses and embedded them in a model membrane. Using cryo‑electron microscopy, they imaged the channels at near‑atomic resolution under neutral conditions, which favor an open pore, and under mildly acidic conditions, which trigger gating. At neutral pH, the channels largely adopt a stable open state, with a central tunnel wide enough for ions and small molecules and with a flexible segment, the N‑terminal domain, lining the pore entrance. Under these conditions the pore is clean: no extra molecules block the way, and the surrounding membrane lipids remain organized in regular layers.
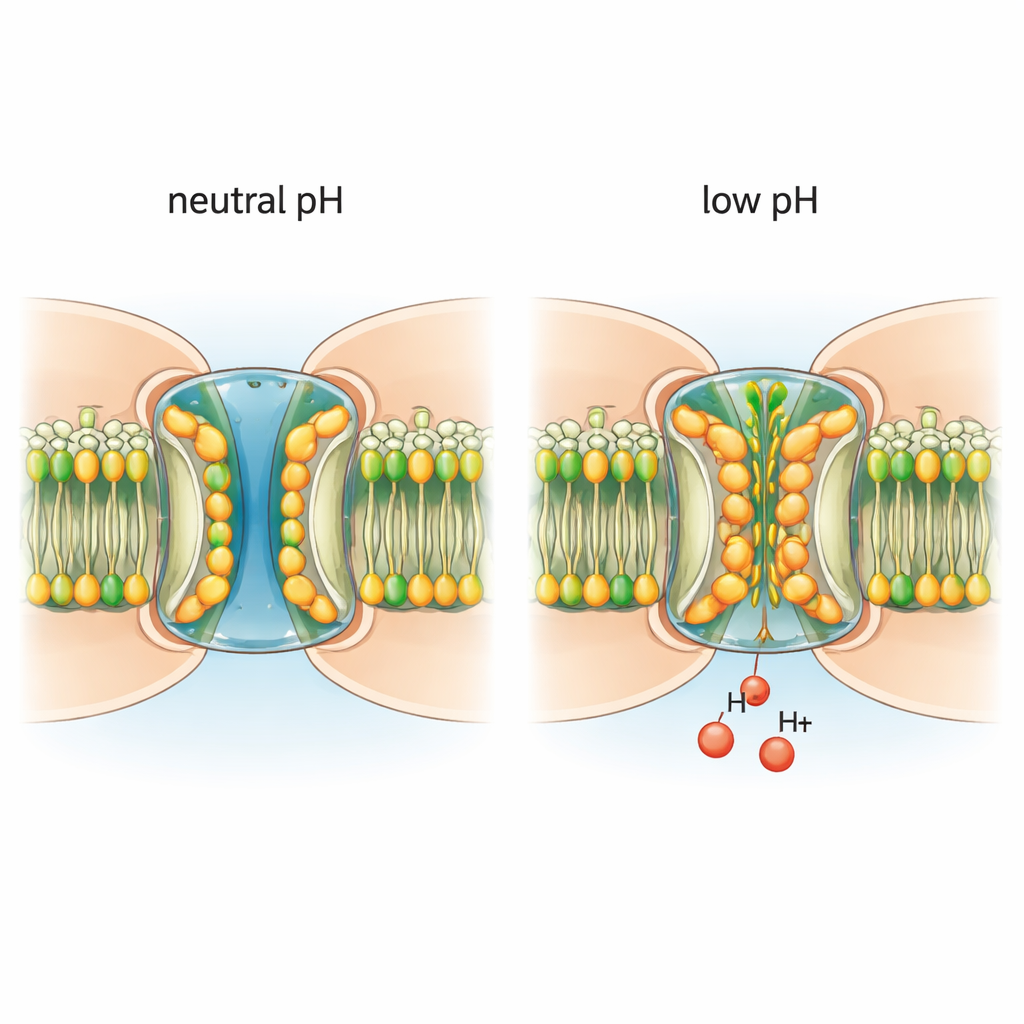
Acid draws lipids into the pore
When the environment was made mildly acidic, the channel’s architecture changed in a striking way. Lipid molecules from the surrounding membrane were drawn into the pore, slipping between and beneath the N‑terminal segments. These invading lipids form a two‑layered hydrophobic “gasket” that pushes the N‑terminal domains inward, narrowing the channel and helping to block traffic. Additional lipids wedge between neighboring protein subunits at their interfaces, apparently serving as entry paths from the membrane into the pore. Importantly, when the channels were placed in a detergent environment that lacks a true lipid membrane, low pH no longer induced these structural changes. This demonstrates that real lipids are not just bystanders: they are required partners in the pH‑gating process.
A reversible and finely tuned safety switch
By tracking millions of individual channel particles, the authors found that low pH does not simply flip channels from fully open to fully closed. Instead, it shifts the population toward a mixture of states: some subunits within a channel show lipid‑stabilized, gated conformations while others remain more open. The gating behavior is largely non‑cooperative—each building block of the channel can respond somewhat independently. Acidic conditions favor these gated conformations, but when the pH is returned to neutral, the pore lipids retreat back into the membrane and the channels revert to the open state. Conserved amino acids called histidines, which gain positive charge at low pH, appear to help recruit and stabilize the interfacial lipids that initiate this process, offering a plausible molecular pH sensor.
Implications for cataracts and tissue protection
These results support a model in which lipids act as movable plugs that reversibly control the flow through gap junctions in response to acidity. In the eye lens, such a mechanism may help isolate damaged cells and prevent toxic signals from spreading, but chronic or misregulated gating could also contribute to age‑related cataract formation. Because similar lipid intrusions have been seen in related channel families, the work suggests a more general principle: under stress, cell membranes themselves can supply dynamic components that help switch vital channels on or off, offering new ideas for drugs that stabilize either the open or closed states in disease.
Citation: Jarodsky, J.M., Myers, J.B. & Reichow, S.L. Reversible lipid-mediated pH-gating of connexin-46/50 by cryo-EM. Nat Commun 17, 1606 (2026). https://doi.org/10.1038/s41467-026-68311-9
Keywords: gap junctions, connexin 46/50, pH gating, cataract, cryo-EM