Clear Sky Science · en
Dynamic single-cell transcriptomics reveals lsamp-guided neural network formation in male S. japonicum driving female reproduction
Why a parasite’s love life matters
Schistosomiasis is a parasitic disease that sickens over 250 million people, mainly in poorer regions with limited medical care. The illness is driven not by the worms’ bites or toxins, but by the enormous numbers of eggs laid by female parasites inside the body. Surprisingly, female worms can only mature and start laying eggs after coupling with a male. This study asks a simple but crucial question: what, exactly, does the male do to switch on the female’s fertility—and could that switch be turned off to stop the disease?
Mapping a hidden world cell by cell
To answer this, the researchers built a detailed “cell atlas” of the blood fluke Schistosoma japonicum during the key window when males and females pair and grow sexually mature. Using single-cell RNA sequencing, a technique that reads which genes are active in individual cells, they analyzed more than 100,000 cells from worms collected at four time points after infection in mice. They identified 76 distinct cell types, including many kinds of nerve cells, stem cells, and reproductive cells. This allowed them to track how specific cell populations expand, shrink, or change identity as the worms grow and begin reproducing.
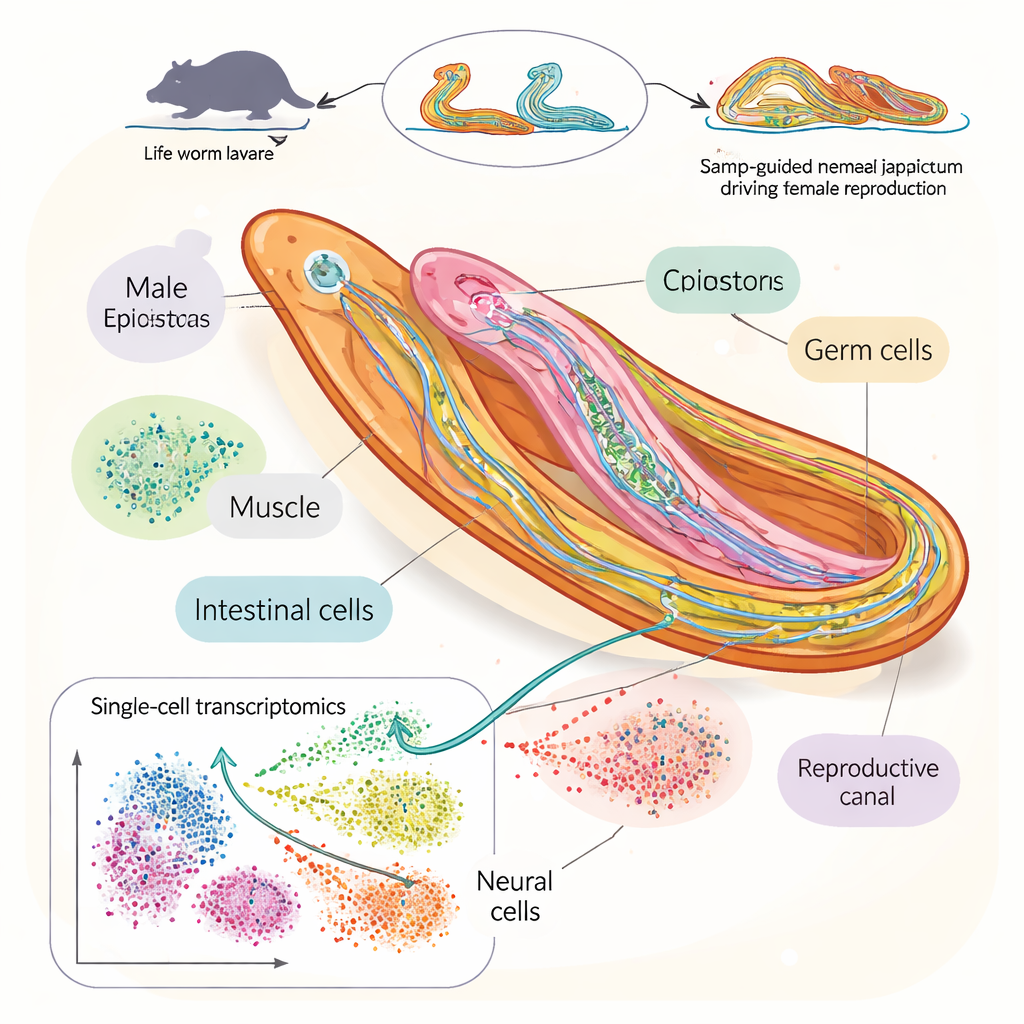
Following the rise of eggs and sperm
By zooming in on cells from the testes, ovaries, and related tissues, the team reconstructed the stepwise development of sperm and eggs. They showed how germline stem cells give rise to late-stage sperm and egg cells through distinct gene activity programs in males and females. In females, the genes that support egg growth emphasize protein production and packaging, while in males they focus more on structures like cilia and microtubules that help sperm move. The study also mapped how the massive egg-supporting organ in females, the vitellaria, develops through intermediate stages, and identified new genetic markers that reveal how mature each reproductive tissue is.
A male-only nerve circuit in a clasping canal
One of the most striking discoveries came from the nervous system. Rather than a loose collection of nerve cells, the atlas revealed five clear neuronal lineages, including several that differed between males and females. Three neuron types—named N2.2, N3.2, and N4.3—were heavily enriched in males and clustered inside the gynecophoric canal, a specialized groove the male uses to cradle the female. Among these, the N4.3 neurons stood out because they expressed a gene called nrps, which makes the enzyme that produces BATT, a small peptide pheromone previously shown to stimulate female sexual development. In other words, these N4.3 cells are the male’s “signal stations” for turning on female fertility.
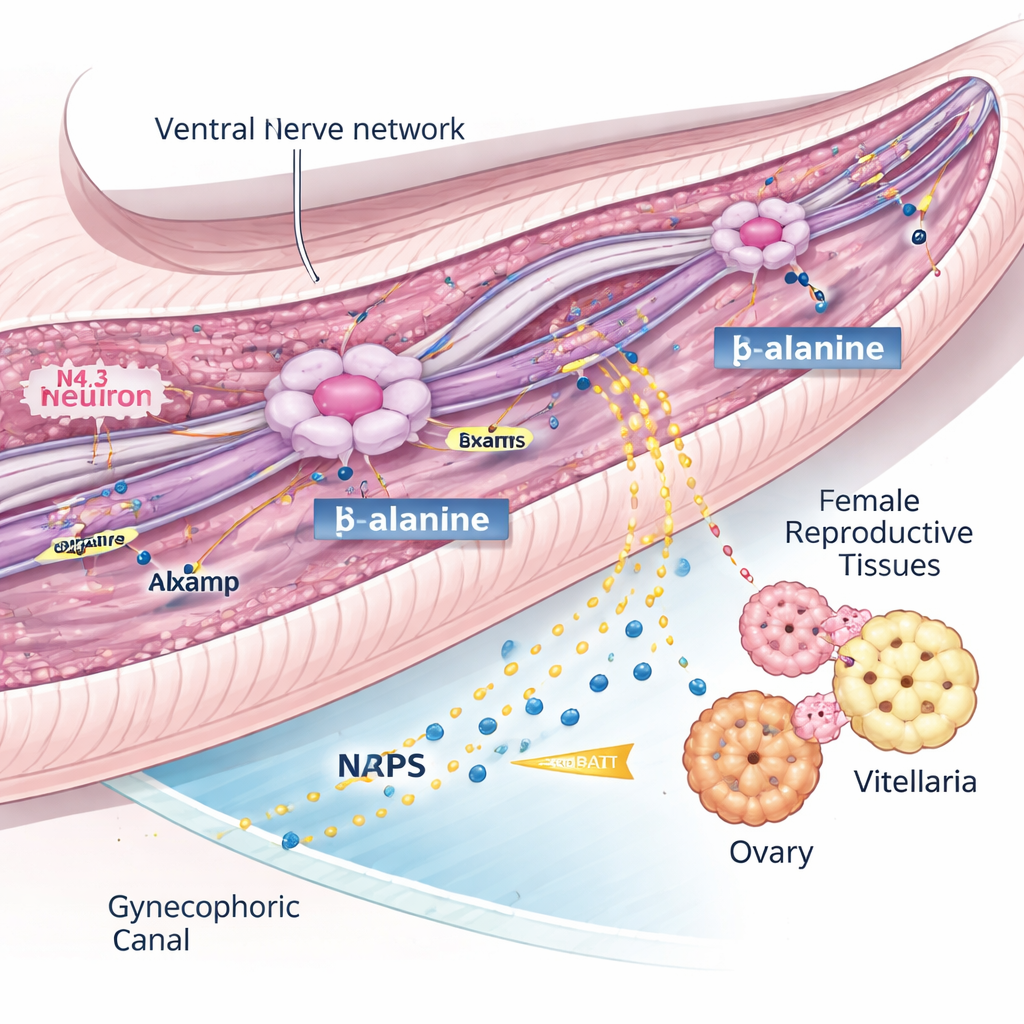
A guiding molecule that keeps the signal flowing
To understand how these N4.3 neurons work, the researchers searched for genes that seemed to steer their development and function. They homed in on lsamp, a gene known in other animals to help nerve cells connect and maintain their axons. In male flukes, lsamp was switched on in the same N4.3 neurons that make the BATT enzyme, and its activity rose sharply as the worms matured. Using RNA interference to reduce lsamp in males, the team found that paired females failed to fully develop their ovaries and vitellaria and laid almost no eggs. Yet the N4.3 neurons were still present, the nrps gene was still active, and the raw chemical ingredients for BATT were unchanged. Instead, lsamp-deficient males showed damaged nerve fibers and disrupted transport of vesicles along stabilized microtubules, and they produced far less BATT inside their bodies and into the surrounding medium. This suggests lsamp is vital for maintaining a ventral nerve network that delivers a key precursor, likely β-alanine, to N4.3 neurons so they can synthesize enough pheromone to activate the female.
Turning down the reproductive switch
For non-specialists, the main message is that this parasite’s ability to cause disease depends on an intimate conversation between male and female worms, carried out through a specialized nerve network and a tiny pheromone. The study shows that a single guidance molecule, lsamp, helps build and maintain the male’s ventral nerve wiring; when that wiring fails, the male can no longer send a strong enough chemical signal to fully mature the female’s reproductive organs, and egg production drops sharply. By charting the parasite’s cells at high resolution and uncovering this male-only neural circuit, the work points to new ways to block egg production—offering a potential strategy to curb schistosomiasis where current treatment relies on just one aging drug.
Citation: You, Y., Cheng, S., Chen, X. et al. Dynamic single-cell transcriptomics reveals lsamp-guided neural network formation in male S. japonicum driving female reproduction. Nat Commun 17, 1602 (2026). https://doi.org/10.1038/s41467-026-68305-7
Keywords: schistosomiasis, single-cell transcriptomics, parasite reproduction, neural circuits, pheromone signaling