Clear Sky Science · en
Lewis acid-triggered hydroxyl spillover enables selective urea electrooxidation to nitrite with concurrent energy-saving hydrogen production
Turning Waste into Useful Chemicals and Clean Fuel
Urea is best known as a component of urine and fertilizer, but in wastewater it becomes a stubborn pollutant. This study shows how urea can be transformed from a liability into a resource: using a smartly designed catalyst, the authors turn urea into nitrite, a valuable chemical for fertilizers and medicines, while simultaneously producing hydrogen fuel using less electricity than conventional water splitting. The work offers a glimpse of future treatment plants that clean water, make useful products, and generate clean energy all at once.
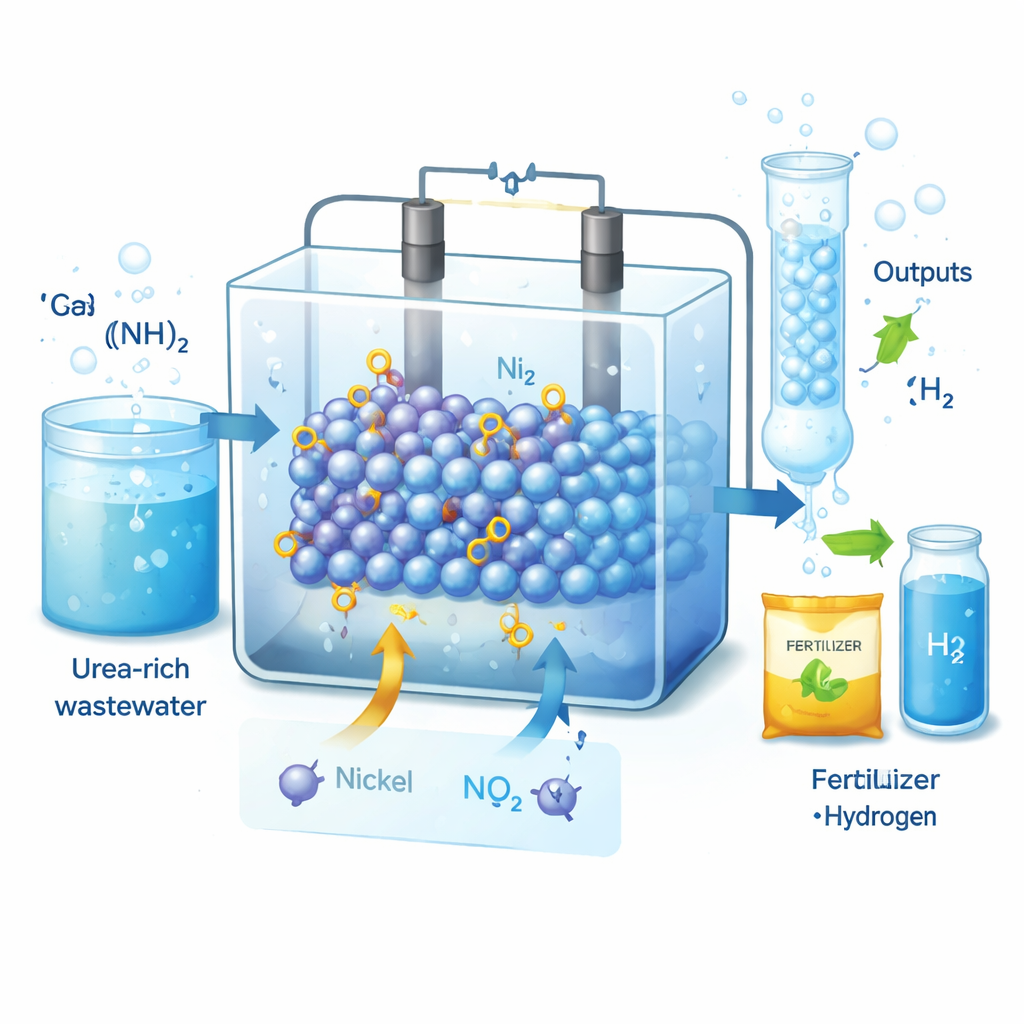
Why Nitrite and Hydrogen Matter
Nitrite is a key ingredient in agriculture, food preservation, and pharmaceuticals, and global demand is measured in millions of tons per year. Today most nitrite is made by the Ostwald process, which runs at high temperature, consumes large amounts of energy, and releases polluting nitrogen oxides. At the same time, wastewater from households and industry carries a heavy load of urea, representing 70–80% of nitrogen pollutants. If that urea could be electrochemically upgraded into nitrite while also yielding hydrogen gas, we could both clean water and supply two high-value products—provided the process is efficient and selective enough.
Guiding Reactions Along the Right Path
When urea is oxidized in alkaline solution, it can follow two main pathways. One leads to harmless nitrogen gas and carbon dioxide; the other, more desirable route leads to nitrite and nitrate, which are marketable chemicals. The problem is that most nickel-based catalysts, the workhorses of this reaction, are not very choosy—they tend to produce a mix of products and also waste energy by driving the oxygen evolution reaction, which makes oxygen gas but no revenue. The authors set out to redesign the catalyst surface so that hydroxide ions, the reactive OH⁻ species in alkaline media, are concentrated and steered in a way that favors cutting carbon–nitrogen bonds and forming nitrite rather than letting nitrogen atoms couple into N₂.
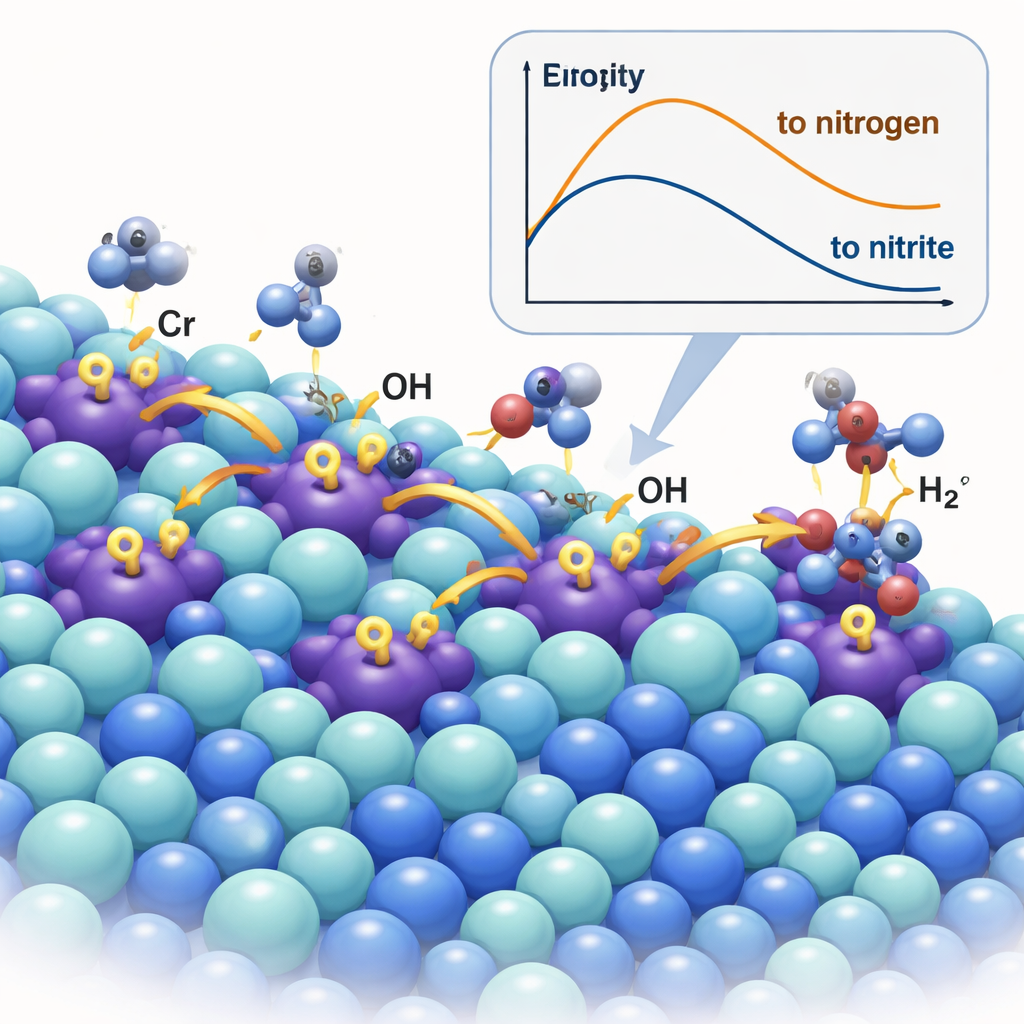
A Catalyst that Pumps Reactive Species
The team created a new material by doping nickel sulfide (Ni₃S₂) with a small amount of chromium, forming Cr–Ni₃S₂. Chromium ions act as so-called Lewis acid sites—electron-poor centers that strongly attract hydroxide ions. Using advanced microscopy, X-ray techniques, and spectroscopy, the researchers confirmed that Cr atoms sit within the nickel sulfide lattice and subtly shrink and distort it, changing how electrons are distributed. Under reaction conditions, the chromium sites act like tiny pumps: they capture OH⁻ and then "spill" it over to neighboring nickel sites, where the actual urea oxidation occurs. In situ Raman and infrared measurements, as well as isotope-labeling experiments, directly traced this spillover of hydroxide from Cr to Ni and showed that it accelerates the formation of active nickel oxyhydroxide (NiOOH) sites that drive the desired chemistry.
High Yields, Lower Energy, and Solid Stability
Because OH⁻ is delivered efficiently to the right places, the Cr–Ni₃S₂ catalyst converts urea to nitrite with impressive selectivity. At industrially relevant current densities, it achieves a nitrite yield of about 121 milligrams per hour per square centimeter with faradaic efficiencies for nitrite above 80%, while keeping competing oxygen evolution below 1.5%. The catalyst remains stable over hundreds of hours of continuous operation with negligible chromium leaching. The same material also dramatically lowers the voltage needed when paired with a hydrogen-evolving cathode in a urea-assisted water-splitting device, cutting the electricity cost of hydrogen production to roughly 3.7 kilowatt-hours per cubic meter of H₂—less than conventional alkaline electrolysis. Techno-economic analysis suggests that, at 400 milliamps per square centimeter, processing a ton of urea in this system could yield on the order of $1,200 in net value when both nitrite and hydrogen are counted.
From Lab Cell to Practical Energy Devices
To demonstrate real-world potential, the authors built a flow cell for continuous urea-assisted water splitting and a Zn–urea–air battery. In the battery, replacing the usual oxygen evolution reaction during charging with the urea oxidation reaction cut the charging voltage by nearly 0.3 volts while maintaining stable performance for more than 100 hours. This means the device can both clean up urea-containing streams and supply electricity storage with higher energy efficiency. The same Lewis acid design strategy also worked when other metals such as tin and titanium, or a different host like copper sulfide, were used, suggesting that the approach is broadly applicable.
A Simple Idea Behind a Complex Reaction
For non-specialists, the key idea is that the researchers have learned how to guide where and how a common reactive ingredient—hydroxide—lands and moves on a catalyst surface. By adding chromium sites that act as strong attractors and relays for OH⁻, they make it easier for urea molecules to be sliced into nitrite instead of being fully burned off to nitrogen gas. At the same time, this route requires less electrical energy and naturally produces hydrogen fuel. In essence, the work shows that careful atomic-scale design of catalyst "traffic patterns" can turn wastewater into a source of both chemicals and clean energy.
Citation: Fan, C., Zhang, M., Li, Y. et al. Lewis acid-triggered hydroxyl spillover enables selective urea electrooxidation to nitrite with concurrent energy-saving hydrogen production. Nat Commun 17, 1585 (2026). https://doi.org/10.1038/s41467-026-68302-w
Keywords: urea oxidation, nitrite production, hydrogen generation, electrocatalysis, wastewater valorization