Clear Sky Science · en
Mixing protocols determine liquid–liquid phase separation dynamics in polyelectrolyte complex coacervation
Why how we mix matters
Many of nature’s most important droplets are not made of oil and water but of charged polymers dissolved in water. These liquid “coacervates” help cells organize their contents and allow marine animals like mussels and sandcastle worms to glue themselves to wet rocks. This study asks a deceptively simple question with big consequences: if you start from the same ingredients, but mix them in different ways, how much does that change how fast and how smoothly these droplets form?
Droplets born from charged polymers
The work focuses on coacervates formed when positively and negatively charged polymer chains meet in water. Like magnets snapping together, opposite charges attract and pull the chains into a dense liquid phase, leaving a more dilute surrounding solution. This liquid–liquid separation resembles the way oil beads up in water, but here everything is water-based and highly charged. Such polymer-rich droplets are thought to underlie “membraneless organelles” inside cells and the fast-setting underwater glues used by marine organisms. Yet while scientists have studied the end state of these droplets in detail, the step‑by‑step journey—the dynamics of how they emerge and grow—has remained much less clear.
Three ways to start, three very different journeys
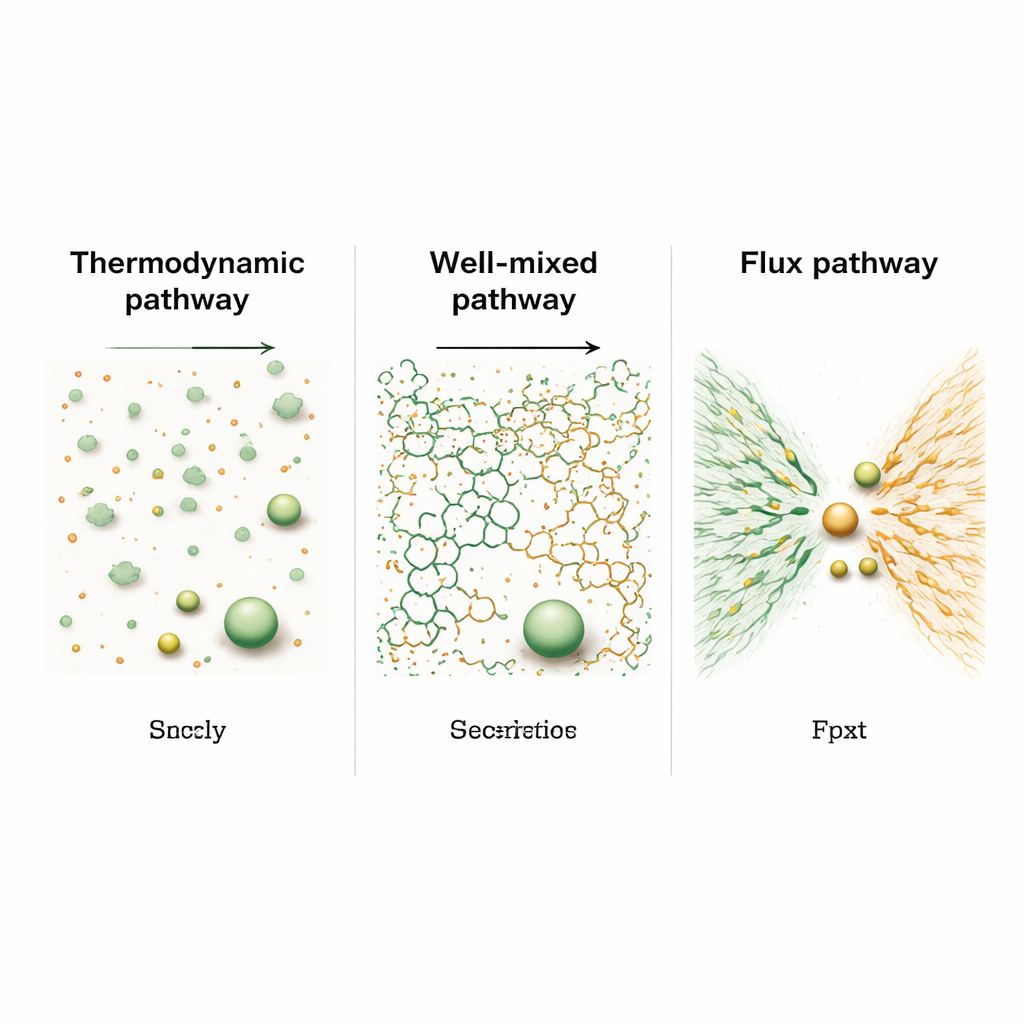
Using large-scale molecular simulations that include both electrical forces and fluid flow, the authors compared three idealized ways of starting the system. In the “thermodynamic” pathway, the polymers begin as many small, already paired clusters scattered throughout the liquid. These clusters slowly merge like coalescing raindrops, and the average droplet size grows with time following a classic, relatively slow law (proportional to time to the one‑third power). In contrast, if the polymers begin thoroughly stirred together at high concentration—the “well‑mixed” pathway—they first form a sponge-like network that spans the system before collapsing into bigger droplets. A third, “flux” pathway mimics mussels and sandcastle worms: positively and negatively charged polymers start in separate regions and are then driven to flow into a shared zone where droplets appear almost explosively.
Networks, flows, and ultra‑fast growth
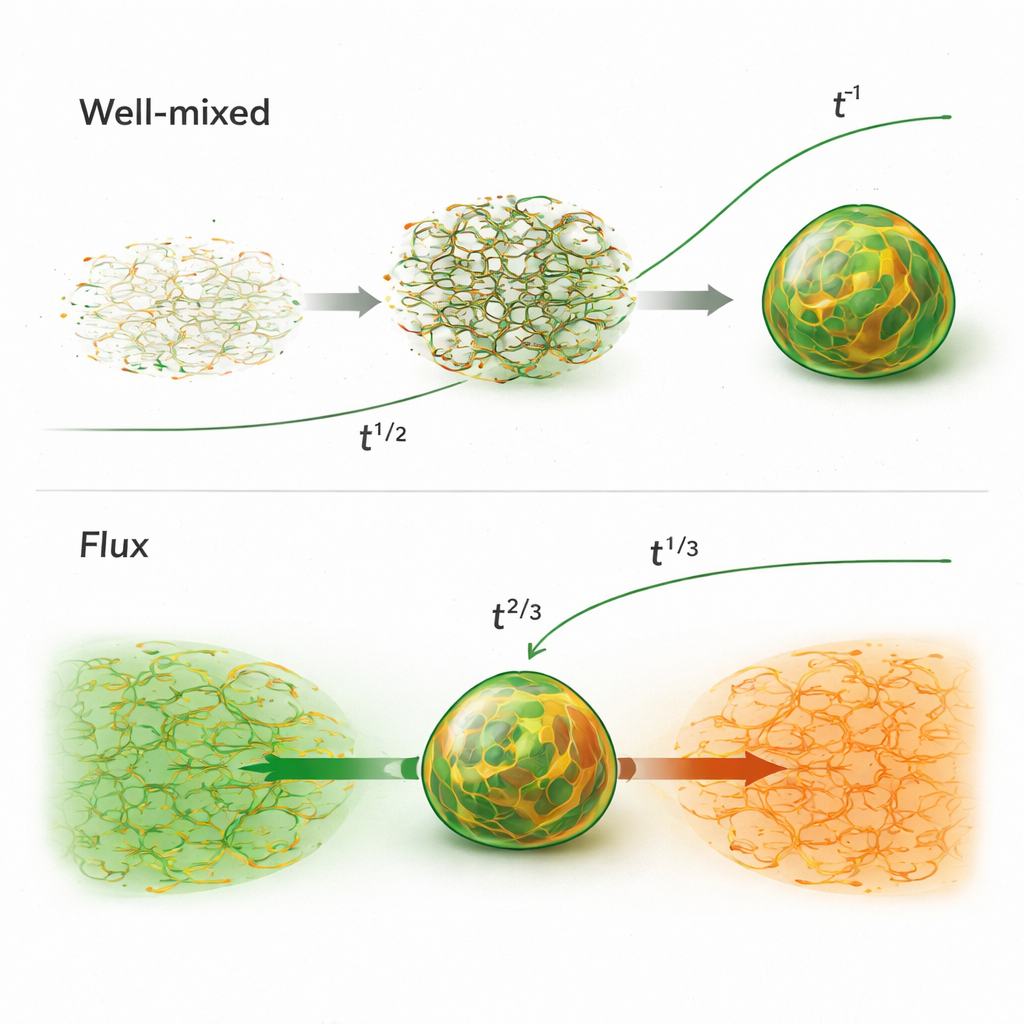
These starting conditions lead to strikingly different growth speeds. In the well‑mixed case, the early sponge-like network allows material to move efficiently through connected paths, making droplets grow roughly as fast as the square root of time—noticeably quicker than the classical droplet‑merging route. Depending on how evenly the charges are mixed at the beginning, this network later either crumbles into many droplets that then coarsen in the usual slow way, or remains connected and pumps fluid so effectively that the droplet size grows almost linearly with time. In the flux pathway, where two charged polymer domains rush together, the early growth is even faster, following a two‑thirds power of time. This burst of growth is driven by a strong electrical and concentration imbalance that pulls material toward the interface, much like water racing downhill under gravity.
What sets the speed limit
The simulations reveal that both the overall concentration and the local balance of charges act like knobs that tune the path of separation. At high polymer concentrations, a transient network forms and speeds up early growth; at lower concentrations the polymers instead form scattered droplets, and growth slows down. When positive and negative charges are well balanced in each local region, the connected structures remain intact and can channel fluid flows that dramatically accelerate coarsening. When the balance is poor, the network fragments, and the system reverts to slower, droplet‑by‑droplet growth. In all cases, given enough time, the system ends up in a similar final state: one large, soft droplet of coacervate surrounded by a dilute phase.
Implications from cells to underwater glue
For a non‑specialist, the central message is that “how you start” can change “how fast you get there” by orders of magnitude—even when the final material looks the same. Under biologically inspired mixing conditions, droplets that would take decades to assemble via the slow, classical route could instead form in seconds. This helps explain how cells rapidly build and remodel internal droplets, and how marine organisms generate strong underwater adhesives on demand. It also suggests practical design rules for technologies: by choosing the right mixing protocol, engineers could build smart materials, drug delivery systems, or bio‑inspired glues that switch on quickly and reliably, simply by controlling how and where the charged polymers first meet.
Citation: Wu, Z., Wang, ZG. & Chen, S. Mixing protocols determine liquid–liquid phase separation dynamics in polyelectrolyte complex coacervation. Nat Commun 17, 1580 (2026). https://doi.org/10.1038/s41467-026-68296-5
Keywords: polyelectrolyte coacervates, liquid–liquid phase separation, biomolecular condensates, underwater adhesives, mixing dynamics