Clear Sky Science · en
Stress vesicles link epidermal mechanotransduction to stem cell differentiation
Why skin’s hidden stress response matters
Every time you pinch your arm, stretch during a workout, or even sleep on a wrinkled pillow, your skin quietly absorbs that mechanical stress. Most of the time it bounces back without a mark. This study uncovers a surprising hidden system that helps skin’s stem cells sense those physical forces and decide whether to stay as stem cells or turn into mature skin cells. Understanding this system could shed light on how skin stays tough yet flexible, and why certain diseases or fragile-skin conditions develop.
Tiny bubbles inside stressed skin cells
Using advanced live imaging in mice, the researchers pressed, pulled, or suctioned the skin while watching individual cells in real time. They found that when skin was subjected to short bursts of force, stem cells in the deepest part of the epidermis quickly formed large internal bubbles, which the authors call “stress vesicles.” These vesicles filled with fluid from outside the cell and grew over seconds to minutes, pushing against and deforming the cell’s nucleus without bursting the cell. When the force was removed, most vesicles shrank and disappeared within a couple of hours, and the nuclei returned to their rounded shape, showing that this was a reversible response rather than outright damage. 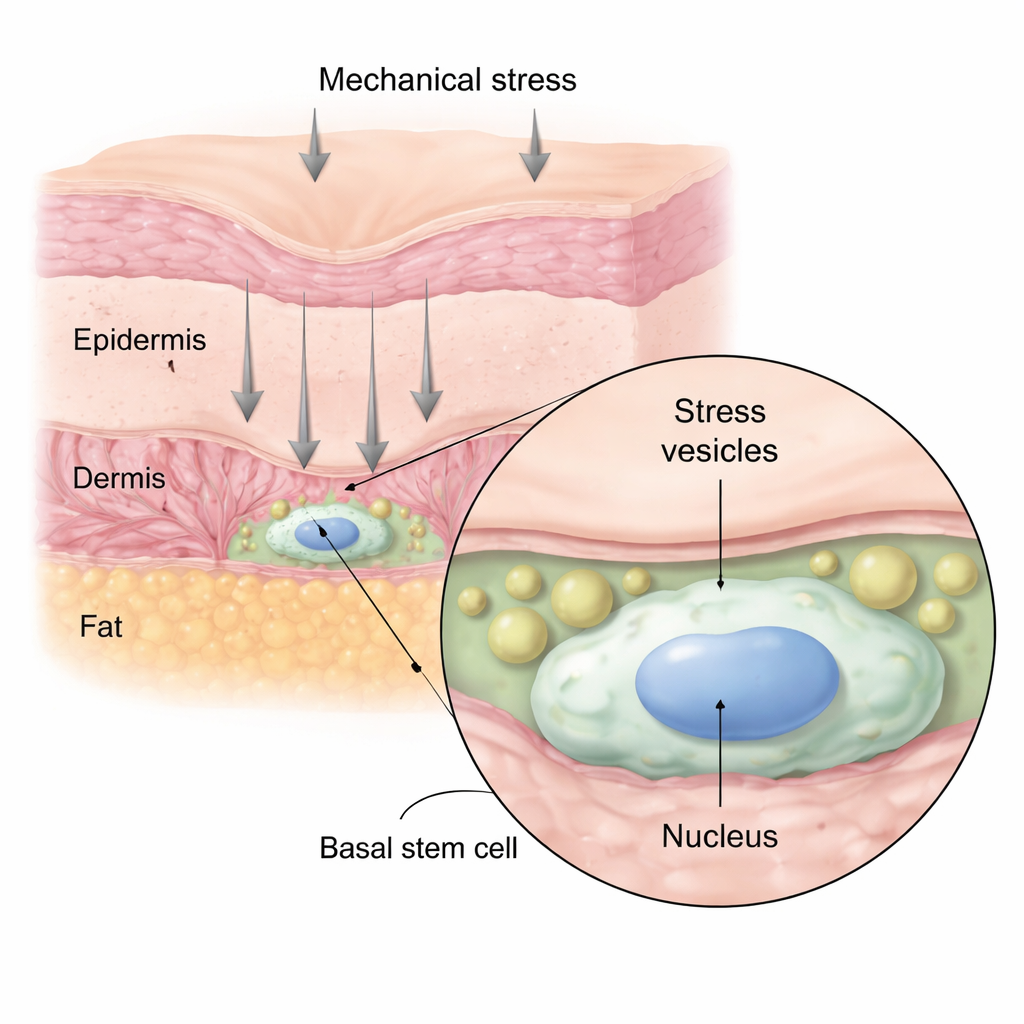
From mechanical push to cell fate choice
The team then asked whether these stress vesicles were just a structural oddity or actually influenced what skin stem cells do next. By tagging and tracking individual basal cells over time, they showed that cells whose nuclei were strongly deformed by stress vesicles were more likely to leave the stem cell layer and move upward to become mature, non-dividing skin cells. Cells that formed few or no vesicles, and whose nuclei stayed round, were more likely to stay put, remain stem-like, or divide. Interestingly, vesicles appeared most often in cells already beginning to shift toward differentiation, suggesting that mechanical stress can push “on-the-fence” cells further along the path to becoming fully mature epidermal cells.
Calcium signals and a force-sensing channel
Mechanical force did more than reshape cells—it also rewired their chemistry. Using a fluorescent calcium reporter, the researchers observed a wave of increased calcium inside epidermal cells immediately after compression. In many cells this spike was brief, but in others calcium stayed high for longer, and those were the same cells that maintained stress vesicles. This pointed to a tight link between persistent calcium elevation, vesicle stability, and a shift toward differentiation. The study then focused on Piezo1, a well-known force-sensitive channel in the cell membrane that opens in response to stretching. When Piezo1 was removed specifically from basal epidermal cells, mechanical stress caused more vesicles to form, calcium levels to remain abnormally high, and more cells to move upward and differentiate. Activating Piezo1 with a small molecule, by contrast, boosted calcium entry in a controlled way and actually reduced vesicle formation.
A conserved skin strategy across species
To test whether this phenomenon was unique to mouse skin, the team built human skin in the lab, grafted it onto mice, and used the same live-imaging and mechanical-stress setup. Human keratinocytes within these grafts formed stress vesicles that closely resembled those seen in mouse cells, including the associated nuclear deformation. This suggests that stress vesicles are a shared feature of mammalian skin, not a laboratory artifact. Additional genetic and microscopic analyses showed that when Piezo1 was missing, genes involved in skin barrier function and cell adhesion were altered, and the skin became more prone to blistering under stress, underscoring the protective role of this mechanosensing pathway. 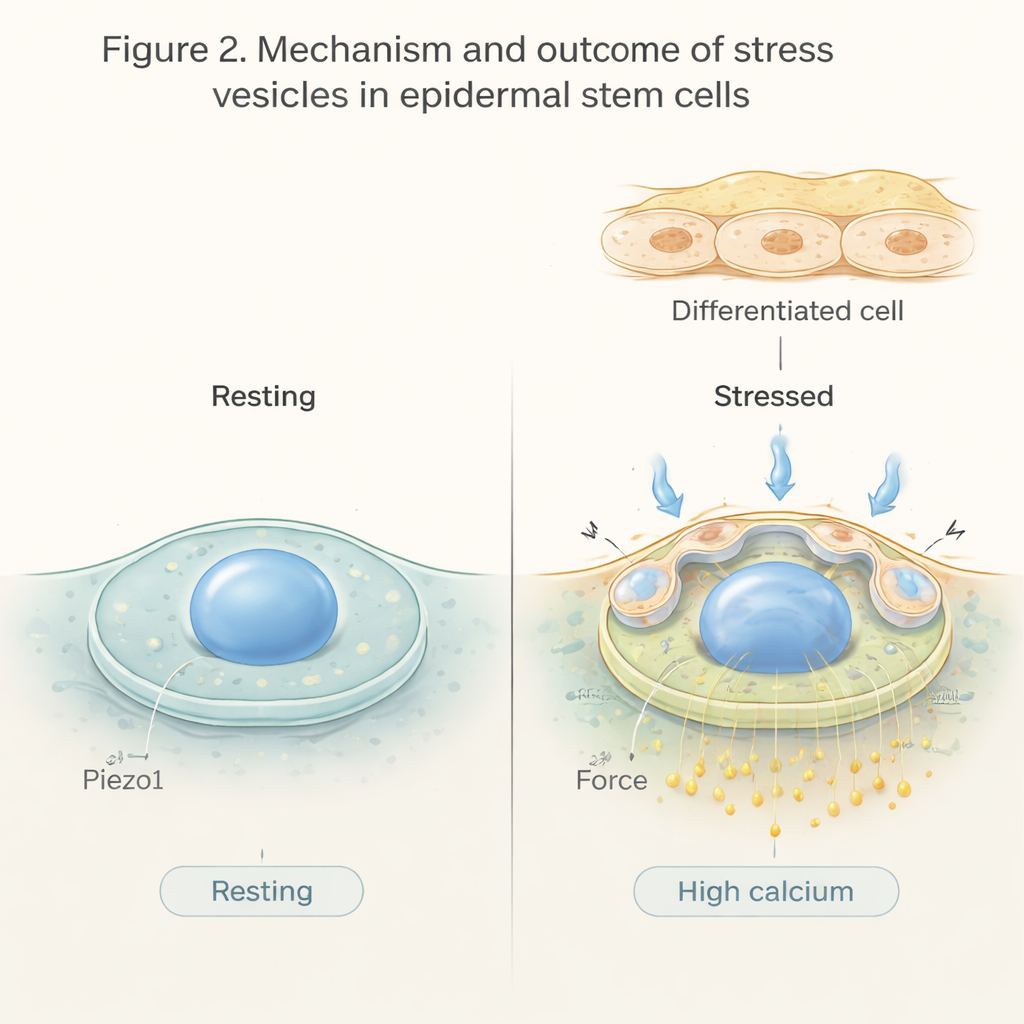
What this means for everyday skin health
In simple terms, this work reveals that skin stem cells don’t just passively endure stretching, pressure, and pulling—they read those forces and respond by taking up fluid into stress vesicles, bending their nuclei, changing calcium levels, and, in many cases, choosing to mature and move upward. The Piezo1 channel acts as a kind of safety valve, shaping how strong and how long these calcium signals last and limiting excessive vesicle formation. Together, these mechanisms help the epidermis renew itself in a controlled way while staying structurally sound under mechanical stress. In the future, targeting this system could open new approaches for treating fragile skin, improving wound repair, or engineering tougher, more resilient skin tissues.
Citation: Huang, S., Kuri, P., Zou, J. et al. Stress vesicles link epidermal mechanotransduction to stem cell differentiation. Nat Commun 17, 1578 (2026). https://doi.org/10.1038/s41467-026-68294-7
Keywords: skin mechanics, epidermal stem cells, calcium signaling, Piezo1 channel, cell differentiation