Clear Sky Science · en
Integrated multi-omic atlas reveals the hierarchy of spatiotemporal regulatory networks of mouse gastrulation
How an embryo builds its body plan
Every mammal, including humans, starts life as a tiny ball of cells that must rapidly organize into a complex body with a head, tail, back, belly, and internal organs. This dramatic reshaping happens during a short window called gastrulation. The paper summarized here builds a detailed, multilayered map of this process in mouse embryos, showing how genes, DNA switches, and chemical signals work together in space and time to guide cells toward their future fates.
Watching thousands of cells make their choices
To follow gastrulation in action, the researchers analyzed over 35,000 individual cells from mouse embryos across five closely spaced stages, from early to late gastrulation. For each cell, they measured not only which genes were turned on, but also which stretches of DNA were open and accessible, a sign that they can act as regulatory switches. Using these data, they identified 31 distinct cell types and traced how early, flexible cells progressively branch into the three main germ layers—ectoderm, mesoderm, and endoderm—that eventually form all tissues and organs. They also developed a new computational method, BioCRE, to more precisely connect genes with their controlling DNA elements, revealing that many important switches lie far from the genes they regulate.
Building a 3D molecular atlas in real embryo space
Most single-cell methods lose the original position of each cell in the embryo, but location is critical for understanding patterning. The team overcame this by aligning their single-cell data onto an existing three-dimensional map of gene activity in mouse embryos. The result is ST-MAGIC, a “digital embryo” in which each tiny location is annotated with the likely cell types present, the genes they express, and the accessibility of surrounding DNA. This atlas shows, for example, how different mesoderm subtypes—future heart, muscle, and supporting tissues—emerge in distinct regions and how widely expressed genes like Otx2 use different regulatory elements on the embryo’s outer (epiblast) and inner (visceral endoderm) surfaces.
When symmetry breaks and left becomes different from right
One striking feature of body plans is that left and right sides are not identical—think of the heart sitting slightly to the left. The authors used their spatial atlas to zoom in on the lateral mesoderm, where left–right differences first appear. They found subtle but consistent asymmetries in which cell types are enriched on each side and in which DNA regions are more open. On the right, regions linked to a growth signal pathway called BMP were more accessible; on the left, regions associated with genes needed for forming segments and heart structures were favored. Some of these DNA elements, including newly discovered ones controlling the gene Lefty2, become open before visible gene expression differences, suggesting that early “priming” of the chromatin landscape prepares each side of the embryo to interpret signals differently.
A relay of factors guides the body’s midline
The study then focuses on the axial mesendoderm, a population of cells that will form the notochord—a rod-like structure running along the midline that helps organize the spine and nervous system. Using their combined tools (ST-MAGIC and an extended version called ST-MAGIC (+)), the authors followed how these cells arise from the anterior primitive streak and split into two branches: node cells, which build a ciliated structure important for left–right sensing, and anterior mesendoderm, which contributes to midline tissues. They uncovered a hierarchical relay of transcription factors—proteins that control gene activity. Early factors, such as EOMES, and intermediate factors, including FOXA2 and LHX1, first open key regions of DNA and establish responsiveness to major signals like WNT and NODAL. Later, “terminal” factors such as NOTO, SOX9, and a newly implicated factor POU6F1 switch on specialized gene programs, for instance those needed for cilia or extracellular matrix.
Signals, chromatin, and late-acting specialists
By integrating public datasets on where WNT and NODAL signal effectors bind DNA, the authors showed that the embryo’s responsiveness to these signals shifts in space before the signal sources themselves move. In the region primed to become axial mesendoderm, DNA sites responsive to NODAL and WNT open early, and many carry motifs for FOXA2, Zfp281, and other regulators, hinting at cooperative control. Experimental deletion of late-acting factors NOTO and POU6F1 in mice disrupted the expression of node-specific and cilia-related genes and shortened node cilia, yet the underlying open DNA landscape remained largely intact. This indicates that earlier factors establish the epigenetic groundwork, while late factors mainly refine gene expression without remodeling chromatin.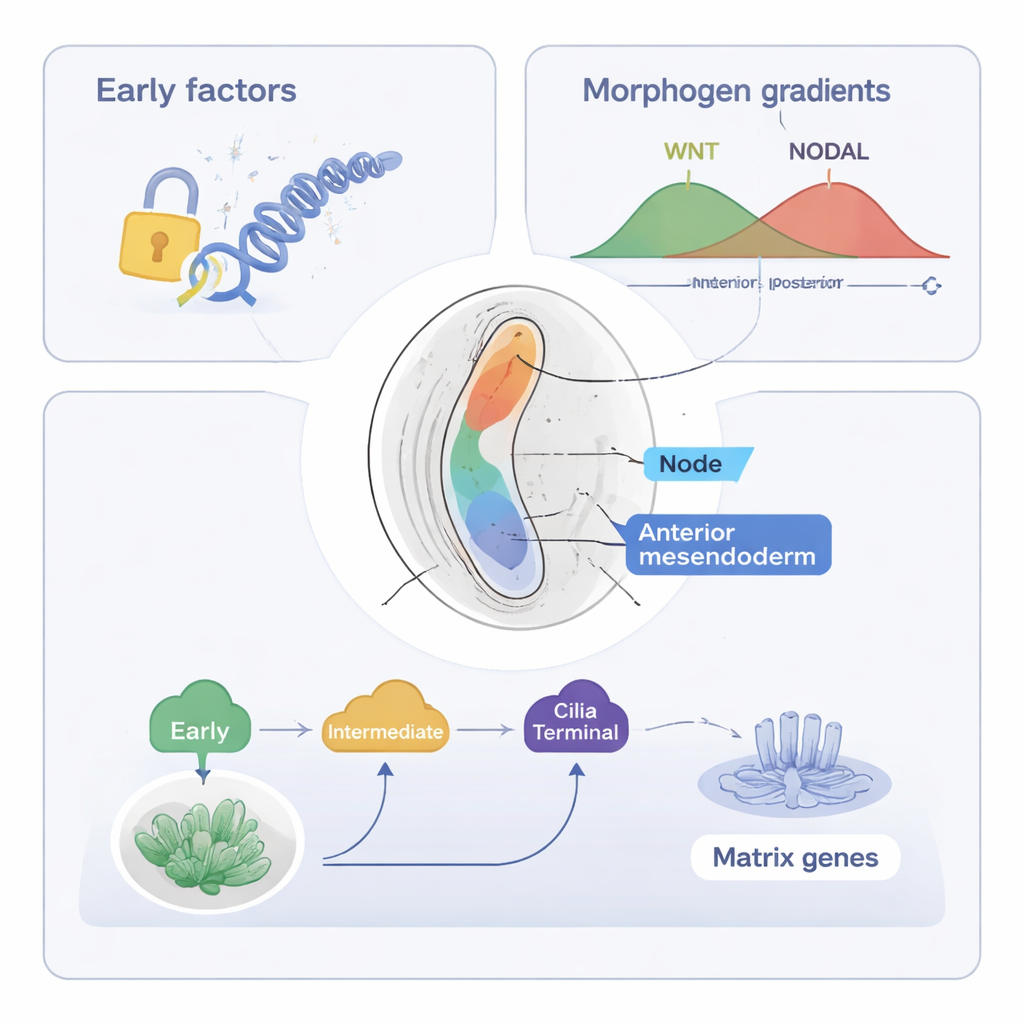
Why this matters for understanding development
To a non-specialist, this work can be viewed as building a high-resolution “wiring diagram” of how an embryo lays out its body plan. The authors show that cell fate decisions during gastrulation are governed not just by which signals are present, but also by when and where DNA switches are opened and which transcription factors act in sequence. Their ST-MAGIC and ST-MAGIC (+) atlases provide a resource for exploring these relationships across space and time, offering a framework that could inform studies of birth defects, stem-cell based embryo models, and, eventually, aspects of human development.
Citation: Yang, X., Xie, B., Shen, P. et al. Integrated multi-omic atlas reveals the hierarchy of spatiotemporal regulatory networks of mouse gastrulation. Nat Commun 17, 1572 (2026). https://doi.org/10.1038/s41467-026-68291-w
Keywords: gastrulation, gene regulatory networks, single-cell multi-omics, embryo patterning, mouse development