Clear Sky Science · en
A centrally positioned cluster of multiple centrioles in antigen-presenting cells fosters T cell activation
How Immune Cells Aim Their Inner Compass
Our immune system depends on close conversations between two key players: dendritic cells, which show fragments of microbes, and T cells, which decide whether to launch an attack. This study reveals that the tiny internal "compass" of dendritic cells—a structure built from centrioles at the cell’s center—is not just a bystander. When this compass is amplified and precisely positioned, it helps dendritic cells switch on T cells more efficiently, with implications for vaccines, immunotherapy, and even cancer treatment.
The Cell’s Tiny Organizing Hub
Every animal cell contains a microtubule-organizing center, anchored by centrioles—short cylinders that act like pegs from which structural fibers (microtubules) grow. These fibers shape the cell’s interior and move cargos such as signaling molecules and vesicles. T cells typically have just one such centrosome with two centrioles. By contrast, dendritic cells can build extra centrioles when they mature and encounter foreign material. The authors wanted to know whether these extra centrioles matter for how well dendritic cells talk to T cells across the immune synapse, the contact zone where information and molecules are exchanged.
Breaking and Weakening the Inner Scaffold
To probe the role of this internal hub, the team first disrupted centrioles or microtubules in mouse dendritic cells. Using a drug that blocks formation of new centrioles, they created cells with few or no centrioles, yet with some residual surrounding material still able to nucleate microtubules. These altered cells could still form microtubule fibers, but when mixed with naive T cells, they triggered far less T cell proliferation and activation. 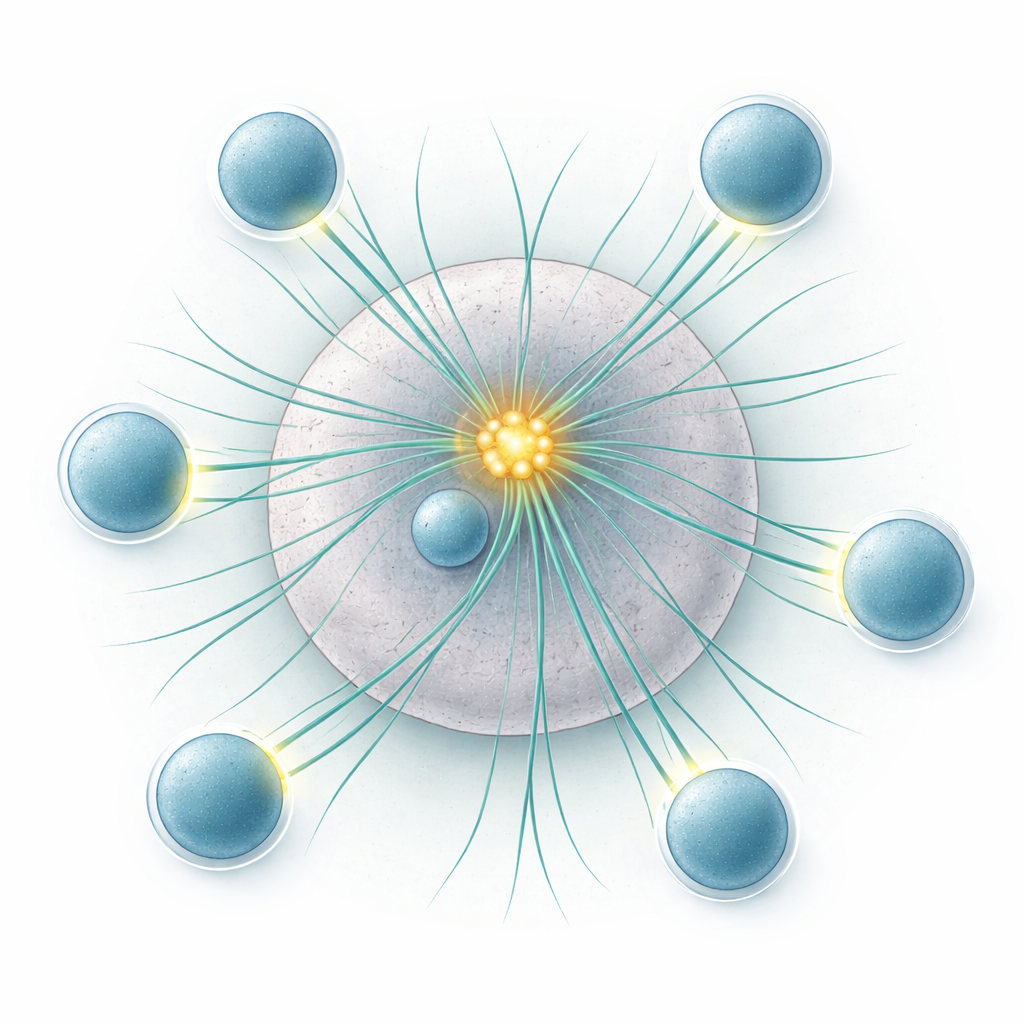
Extra Centrioles as Power Boosters
The authors then turned to dendritic cells that naturally carry multiple centrioles. They found that additional centrioles recruit more of the proteins that seed microtubules, turning the cell’s single organizing center into a more powerful source of fibers, without increasing the number of separate centers. During encounters with T cells, dendritic cells with extra centrioles generated denser microtubule arrays, yet their many centrioles stayed tightly clustered near the cell’s geometric center. Using specially engineered T cells whose fluorescence reports receptor signaling strength, the team showed that dendritic cells enriched for multiple centrioles activated more T cells, and did so faster, than cells with only two centrioles. Importantly, these dendritic cells could engage several T cells at once, and this multi-contact capacity did not depend on how many centrioles they had.
Why Clustering Beats Scattering
Surprisingly, the many centrioles in dendritic cells did not fan out toward each T cell contact. Instead, in culture and in lymph nodes of live mice, they remained as a tight cluster close to the nucleus and cell center. To test whether this clustering matters, the researchers used a drug that loosens centriole cohesion without altering key surface markers or cytokines. Under these conditions, centrioles spread apart, forming multiple weaker organizing centers and more microtubule origins scattered through the cell. T cells facing such "declustered" dendritic cells were activated less efficiently. This suggested that what matters is not just having more centrioles, but keeping them together at the right spot.
Physics Explains the Optimal Sweet Spot
To understand why a centralized centriole cluster is so effective, the team built mathematical and computer models of dendritic cells. They asked: where inside a roughly spherical cell should a microtubule-organizing center sit to minimize the average distance to any potential synapse on the surface? Their calculations showed that, given the presence of a nucleus, the best location is just above the nuclear surface or near the cell’s geometric center, depending on nuclear offset. Dynamic microtubules that grow and shrink from this central hub can then reach any surface point more quickly—a "search and capture" advantage. When they modeled multiple independent organizing centers scattered through the cell, the time needed for microtubules to find a target contact increased. In contrast, clustering several centrioles into one strong center, and increasing microtubule number, consistently sped up target capture.
Implications for Immunity and Therapy
In simple terms, this work shows that dendritic cells act more like efficient air-traffic controllers when their multiple centrioles are grouped into a bright, central hub. This hub launches many microtubule “tracks” that rapidly deliver stimulatory signals to T cells around the cell surface. Disrupting the hub’s integrity or scattering its components slows this delivery and blunts T cell activation. Beyond explaining a subtle layer of immune cell organization, these findings intersect with cancer research, where drugs that disrupt centriole clustering are explored to kill tumor cells. The study warns that such strategies might also dampen immune responses if they inadvertently affect antigen-presenting cells, and it points to centriole amplification and positioning as potential levers to fine-tune vaccines and immunotherapies. 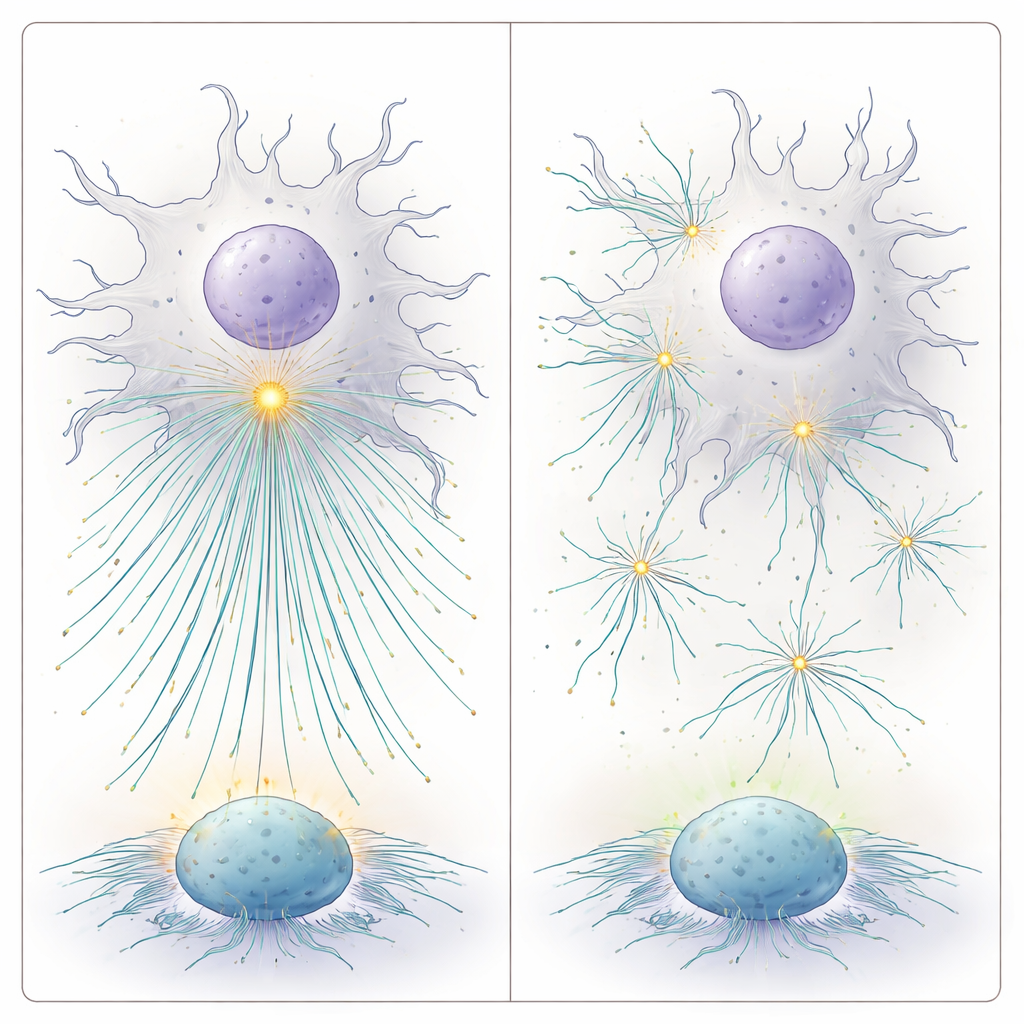
Citation: Stötzel, I., Weier, AK., Sarkar, A. et al. A centrally positioned cluster of multiple centrioles in antigen-presenting cells fosters T cell activation. Nat Commun 17, 536 (2026). https://doi.org/10.1038/s41467-026-68286-7
Keywords: dendritic cells, T cell activation, centrosomes, microtubules, immune synapse