Clear Sky Science · en
Heteroaryl derivatives for hole-transport layers improve thermal stability of perovskite solar cells
Making Solar Panels Last Longer in the Heat
Solar panels work best when they can endure years of sun and heat without losing power. A promising type of solar cell called a perovskite solar cell can already convert sunlight to electricity as efficiently as today’s silicon panels, but it tends to degrade at high temperatures. This study explores a clever, chemistry-based way to make these cutting‑edge cells far more heat‑resistant, bringing them closer to use on real rooftops and solar farms.
The Weak Link in a Promising Solar Technology
Perovskite solar cells have surged in efficiency over the past decade and are now among the top performers in the lab. They are thin, lightweight, and can be made using relatively simple solution processes, which makes them attractive for low‑cost mass production. However, their long‑term stability, especially at high temperatures, still falls short of what is needed for power‑grid applications. A major source of trouble lies in a thin organic coating called the hole‑transport layer, which helps move positive charges out of the light‑absorbing perovskite. The standard recipe for this layer relies on a small molecule called 4‑tert‑butylpyridine (tBP). At elevated temperatures, tBP tends to evaporate and react with the perovskite, creating tiny voids and chemical by‑products that slowly kill device performance.
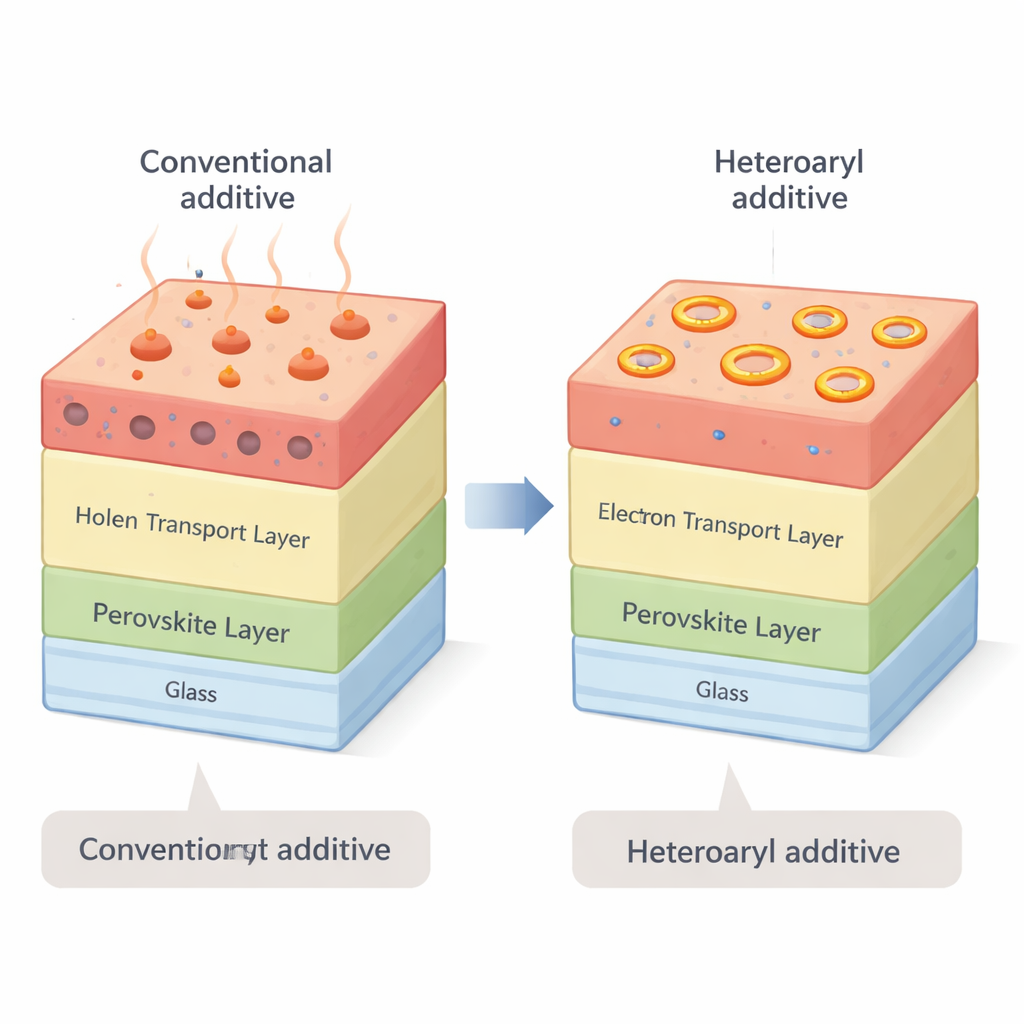
Swapping in Better Helper Molecules
The researchers set out to redesign this vulnerable layer without changing the basic device architecture. They focused on a family of ring‑shaped organic molecules known as heteroaryl derivatives, which can be tuned by changing where and how extra chemical groups are attached. By systematically comparing 36 different derivatives and 60 combinations with other additives, they looked for versions that would stay put in the hole‑transport layer, avoid attacking the perovskite, and still support fast charge extraction. Three compounds with a phenyl–pyridine structure—4‑phenylpyridine, 3‑phenylpyridine, and 2‑phenylpyridine—emerged as standout candidates. These molecules have higher boiling points than tBP and bulkier shapes that reduce unwanted reactions at the interface.
Watching Solar Cells Age in the Oven
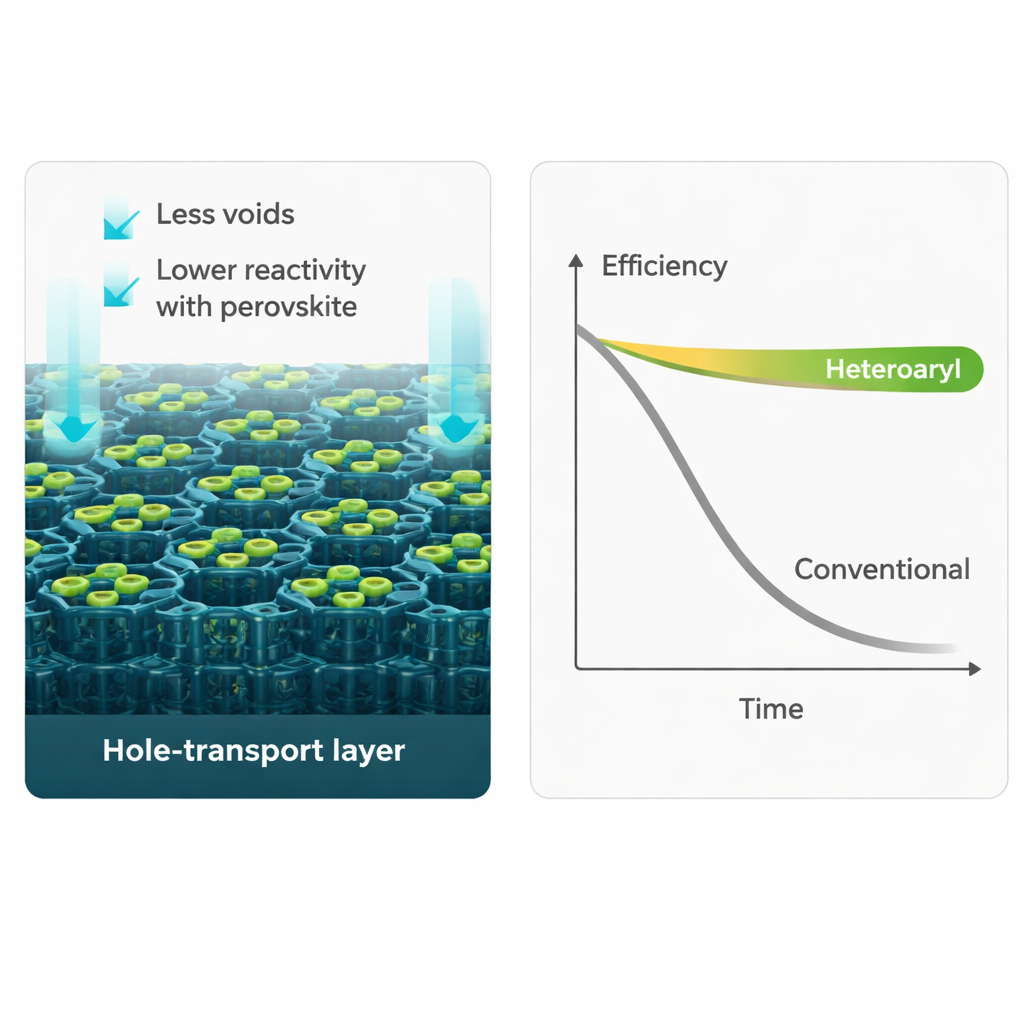
To test real‑world toughness, the team operated solar cells at 85 °C for thousands of hours, a standard accelerated‑aging test. Devices made with the conventional tBP additive suffered a dramatic drop in power conversion efficiency within days. In contrast, cells using 3‑phenylpyridine or 2‑phenylpyridine not only retained their performance but even showed slight gains, keeping 101% and 104% of their initial efficiency after roughly 2,400 hours in the heat. Microscopy images revealed why: in tBP‑based devices, the hole‑transport layer developed large voids and cracks that disrupted electrical contact. With the new additives, this layer remained smooth and continuous, even when made thick enough for scalable coating methods.
How the New Additives Protect the Cell
Multiple measurements helped piece together the underlying mechanism. X‑ray diffraction showed that 2‑ and 3‑phenylpyridine reacted far less with the perovskite, forming fewer unwanted compounds. Depth‑profiling experiments indicated that, unlike tBP, these additives stayed largely confined to the hole‑transport layer instead of migrating into the perovskite. Computer simulations and spectroscopic studies suggested that their particular shapes and bonding patterns reduce volatility and reactivity while still coordinating properly with lithium dopants. Photoluminescence tests further revealed that the new additives enable faster and more efficient transfer of charge from the perovskite into the hole‑transport layer, which supports higher operating voltages and better overall efficiency.
High Efficiency in the Lab and Under Real Sun
Importantly, the thermal benefits did not come at the cost of performance. Optimized cells using 2‑phenylpyridine reached a power conversion efficiency of 25%, putting them on par with the best perovskite devices reported to date. Small solar modules made with these additives also showed strong performance, demonstrating that the strategy can be scaled beyond tiny test cells. In outdoor tests under real sunlight, devices with 2‑phenylpyridine retained about 90% of their operating voltage and 94% of their power output after more than 1,500 hours of continuous tracking at the maximum power point, a demanding cycling condition.
Bringing Durable Perovskite Solar Cells Closer to Reality
For non‑specialists, the takeaway is straightforward: by carefully redesigning a supporting layer in perovskite solar cells with better‑behaved molecules, the authors were able to greatly extend how long these cells survive high heat while also boosting their power output. The work shows that stability problems are not an unavoidable flaw of perovskites, but can be tackled through smart chemistry at the interfaces. If such thermally robust designs can be integrated into large‑area manufacturing, perovskite solar panels could become practical contenders for long‑lived rooftop and utility‑scale installations.
Citation: Kanda, H., Mondal, S., Eguchi, N. et al. Heteroaryl derivatives for hole-transport layers improve thermal stability of perovskite solar cells. Nat Commun 17, 1664 (2026). https://doi.org/10.1038/s41467-025-68236-9
Keywords: perovskite solar cells, thermal stability, hole transport layer, organic additives, photovoltaic durability