Clear Sky Science · en
Viscoelastic hydrogel primed CAR-macrophage for pulmonary fibrosis treatment
Turning the Body’s Janitors into Precision Repair Crews
Pulmonary fibrosis is a devastating lung disease in which normal, springy lung tissue is slowly replaced by stiff scar. People struggle to breathe, yet today’s drugs mostly just slow the decline. This study explores a new idea: converting the body’s own cleanup cells, macrophages, into engineered “smart janitors” and then boosting their power with a specially designed soft gel. Together, these tricks help the cells hunt down scar‑forming culprits and even loosen the rigid tissue in damaged lungs.
When Healing Becomes Harmful
In healthy lungs, fibroblasts help maintain the delicate web of collagen that supports the air sacs. In pulmonary fibrosis, these cells become overactive, laying down thick strands of collagen that choke off air spaces. Existing medicines can damp these processes but rarely reverse them. The authors focus on a cell‑surface flag called fibroblast activation protein (FAP), which is abundant on overactive fibroblasts but scarce in normal tissue. If immune cells could be trained to recognize FAP, they might selectively remove the “runaway” fibroblasts that drive scarring while sparing healthy neighbors.
Reprogramming Macrophages to Target Scar Builders
Macrophages are roaming immune cells that normally engulf dead cells, microbes, and cellular debris. The team equipped them with a chimeric antigen receptor (CAR) that recognizes FAP, creating CAR‑macrophages (CAR‑Ms). In lab dishes, these CAR‑Ms efficiently swallowed FAP‑rich fibroblasts and killed them, while largely ignoring cells with reduced FAP. They also chewed through collagen‑rich gels more vigorously than unmodified macrophages, suggesting a double benefit: eliminating the cells that create scar and directly breaking down the scar matrix itself. These effects held true both in standard cell lines and in primary mouse macrophages, which are closer to the real situation in tissues. 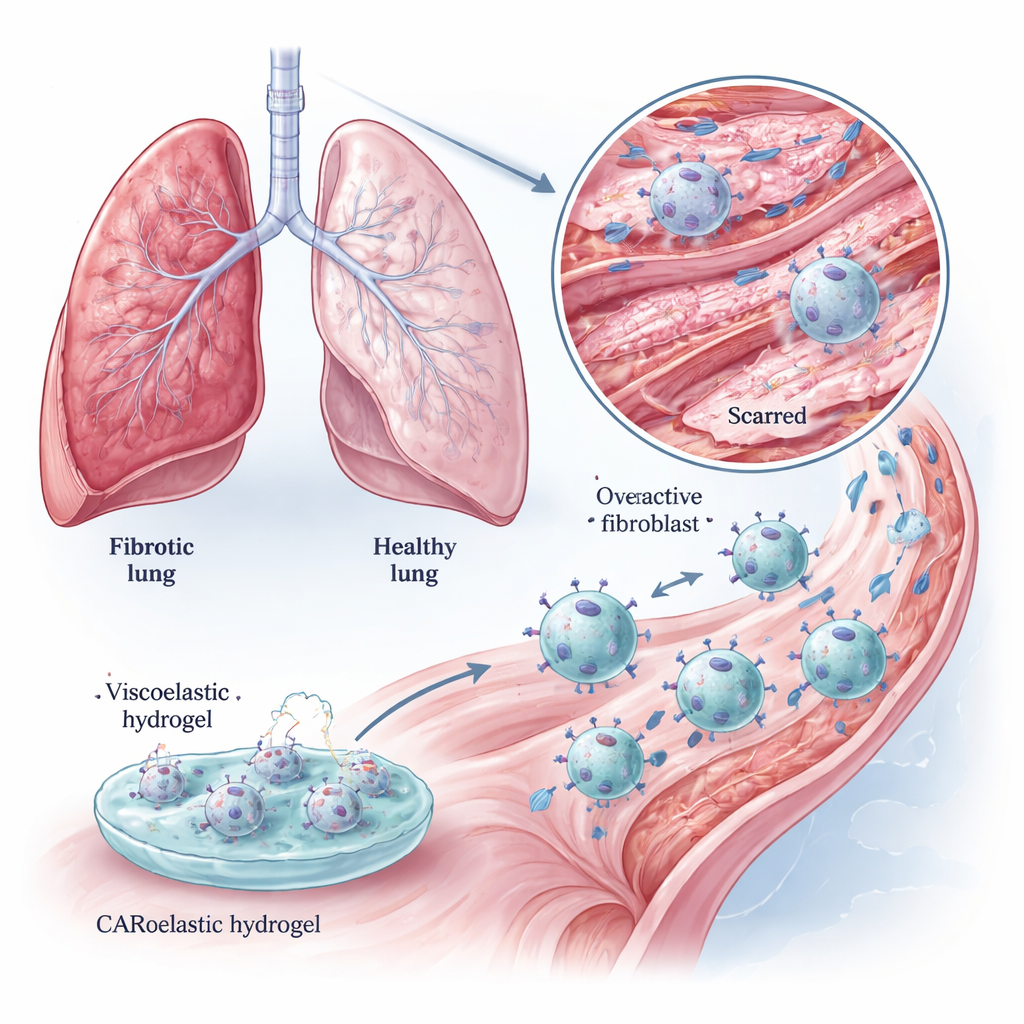
Priming Immune Cells with a Soft, Viscoelastic Gel
Genetically engineering cells is powerful but complex. The researchers therefore asked whether a purely physical cue—a soft, viscoelastic hydrogel—could further tune CAR‑M behavior. They built a gelatin–alginate hydrogel whose “loss modulus,” a measure of how it slowly relaxes under stress, could be finely tuned without changing overall stiffness. When CAR‑Ms were briefly cultured on gels with the right viscoelasticity, their ability to kill target fibroblasts increased more than with common chemical stimulants, and the boost lasted for at least two days. These “Gel‑CAR‑Ms” also turned on genes linked to immune activation and tissue remodeling, while dialing down genes associated with fibrosis, hinting that the gel experience imprinted a more anti‑scar identity onto the cells.
How a Softer Surface Rewires Cell Behavior
To understand why the gel mattered, the team examined the physical state of the CAR receptors on the cell surface. A fluorescent probe revealed that hydrogel‑primed CAR‑Ms had lower membrane tension—the outer skin of the cell was more relaxed. Under these conditions, CAR molecules spread out from tight clusters into more isolated single units and pairs. Biochemical tests showed that these dispersed receptors favored forms that more readily sparked internal signaling, especially along pathways like ERK that control activation and killing capacity. Mimicking the tension drop with a small molecule reproduced the same receptor spreading and improved tumor‑cell killing, supporting the idea that mechanical relaxation of the membrane is enough to “pre‑arm” the cells before they ever meet their targets. 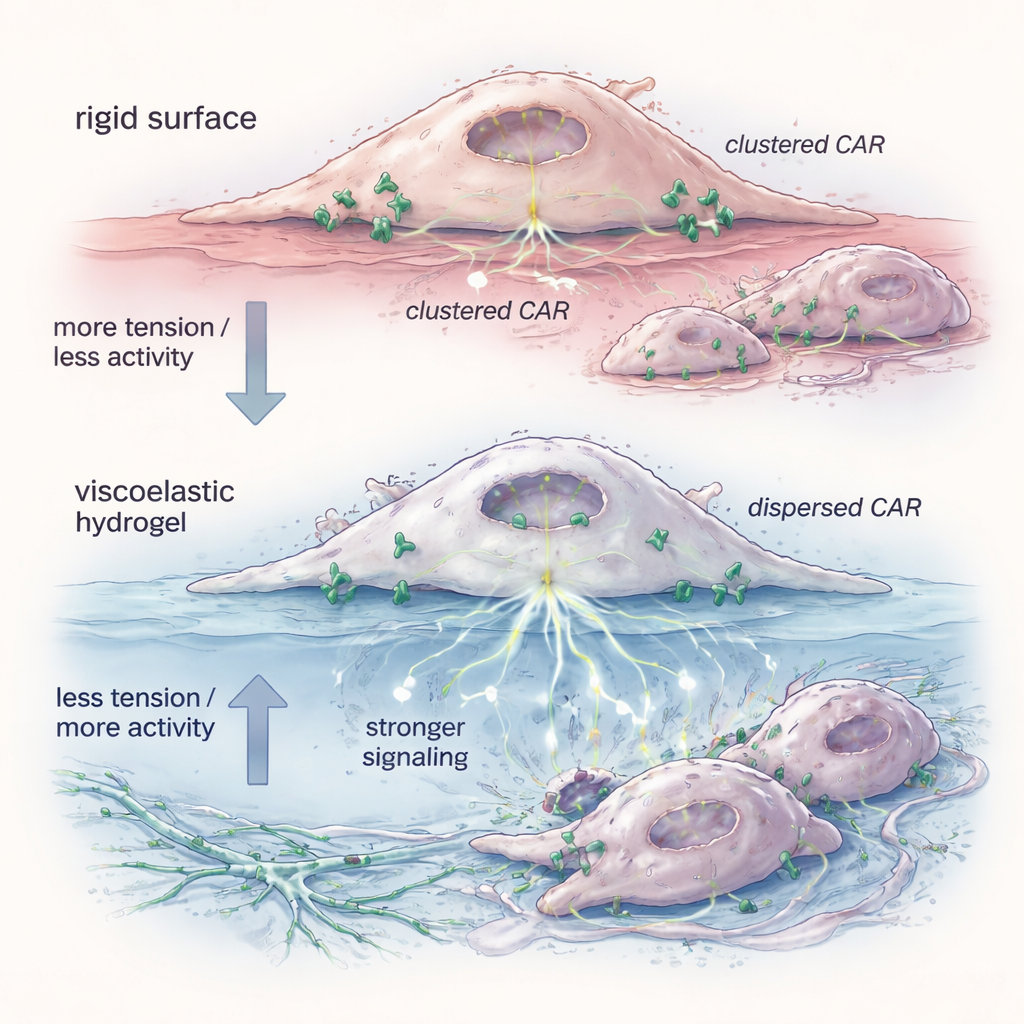
Repairing Scarred Lungs in Mice
The ultimate test was a mouse model of drug‑induced pulmonary fibrosis. Animals received either plain macrophages, standard CAR‑Ms, or hydrogel‑primed Gel‑CAR‑Ms. All treated mice improved compared with untreated controls, but Gel‑CAR‑Ms performed best: their lungs contained less collagen, more normal air spaces, and fewer FAP‑positive fibroblasts. Gene readouts from lung tissue showed that Gel‑CAR‑Ms calmed inflammatory signals and reduced expression of scar‑related genes more effectively than standard CAR‑Ms. Tracked cells persisted in the lung for at least a week, and extended safety checks over 12 weeks found no major organ damage, blood abnormalities, or signs of autoimmunity.
A New Direction for Cell‑Based Anti‑Fibrosis Therapies
For non‑specialists, the key message is that the combination of targeted cell engineering and smart materials can turn innate immune cells into highly effective, yet non‑genetically overcomplicated, scar‑fighting tools. By briefly resting engineered macrophages on a carefully tuned soft gel, the researchers mechanically “pre‑set” their surface receptors into a more responsive mode. In mice with lung fibrosis, this translated into better removal of harmful fibroblasts, breakdown of excess collagen, and partial restoration of normal lung structure—without obvious safety problems. While much work remains before human trials, this strategy suggests that fine‑tuning the physical environment of therapeutic cells could one day make treatments for fibrotic lung disease more potent and more precise.
Citation: Zhang, Y., Liu, Z., Kong, W. et al. Viscoelastic hydrogel primed CAR-macrophage for pulmonary fibrosis treatment. Nat Commun 17, 1663 (2026). https://doi.org/10.1038/s41467-025-68033-4
Keywords: pulmonary fibrosis, CAR macrophage therapy, viscoelastic hydrogel, fibroblast activation protein, cell mechanobiology