Clear Sky Science · en
Reversible color switching of bright phosphorescence in purely organic materials for advanced data encryption
Glowing Crystals That Can Change Their Minds
Imagine an ink that keeps glowing after the lights are turned off—and can change its color on command to hide or reveal secret information. This study introduces a new purely organic material that does exactly that. It switches its long‑lasting glow back and forth between blue and green using only gentle heating or exposure to common solvents, opening the door to more secure data encryption and anti‑counterfeiting technologies without relying on metals or complicated mixtures.
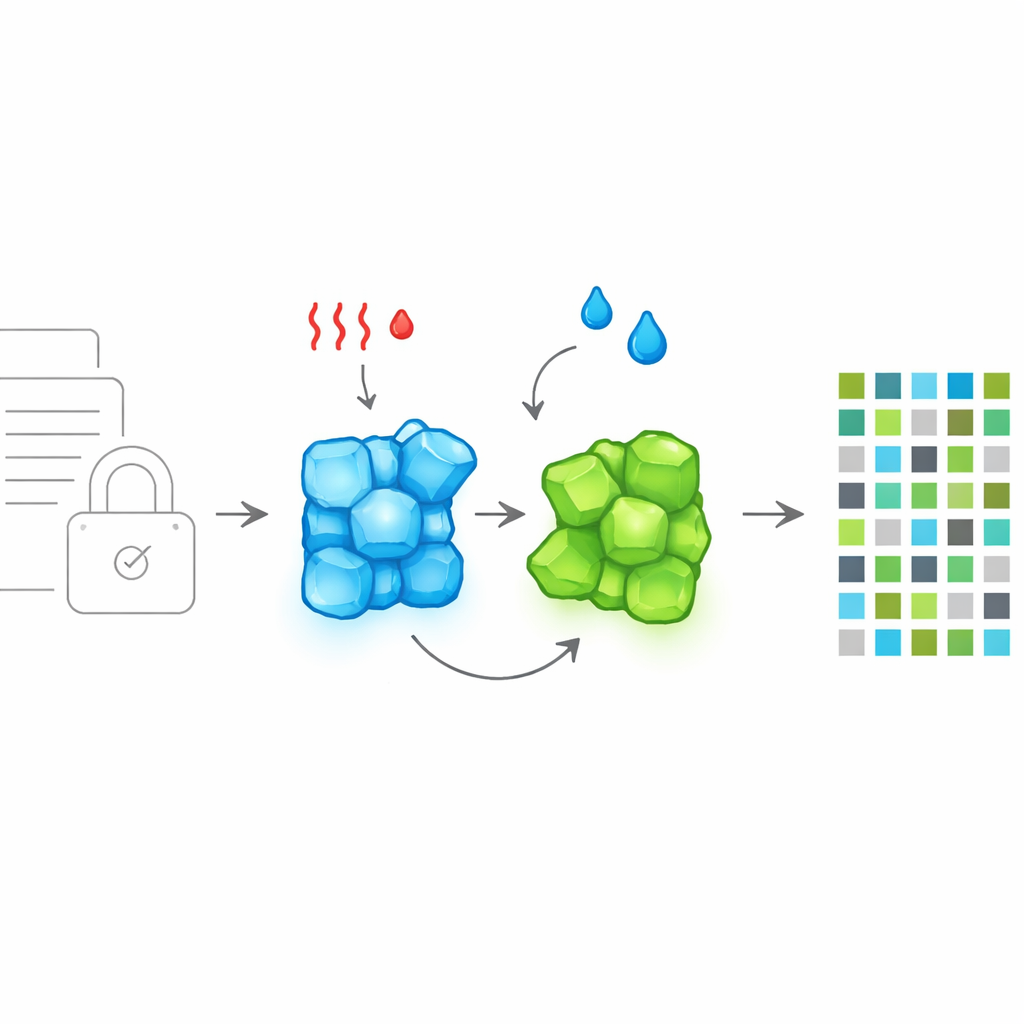
Why Long-Lasting Light Matters for Security
Many familiar glowing materials stop shining the moment you turn off the light that excites them. In contrast, some special compounds store energy and release it slowly as a faint afterglow, a behavior known as phosphorescence. Such long‑lived emission can be used as a time‑gated signal that appears only when viewed under the right conditions or with sensitive detectors. However, most existing organic phosphorescent systems either require multiple components, harsh conditions, or lose brightness when they are switched or stressed. That makes it hard to build practical, tamper‑resistant security features that must survive real‑world handling.
A Single Crystal with Two Personalities
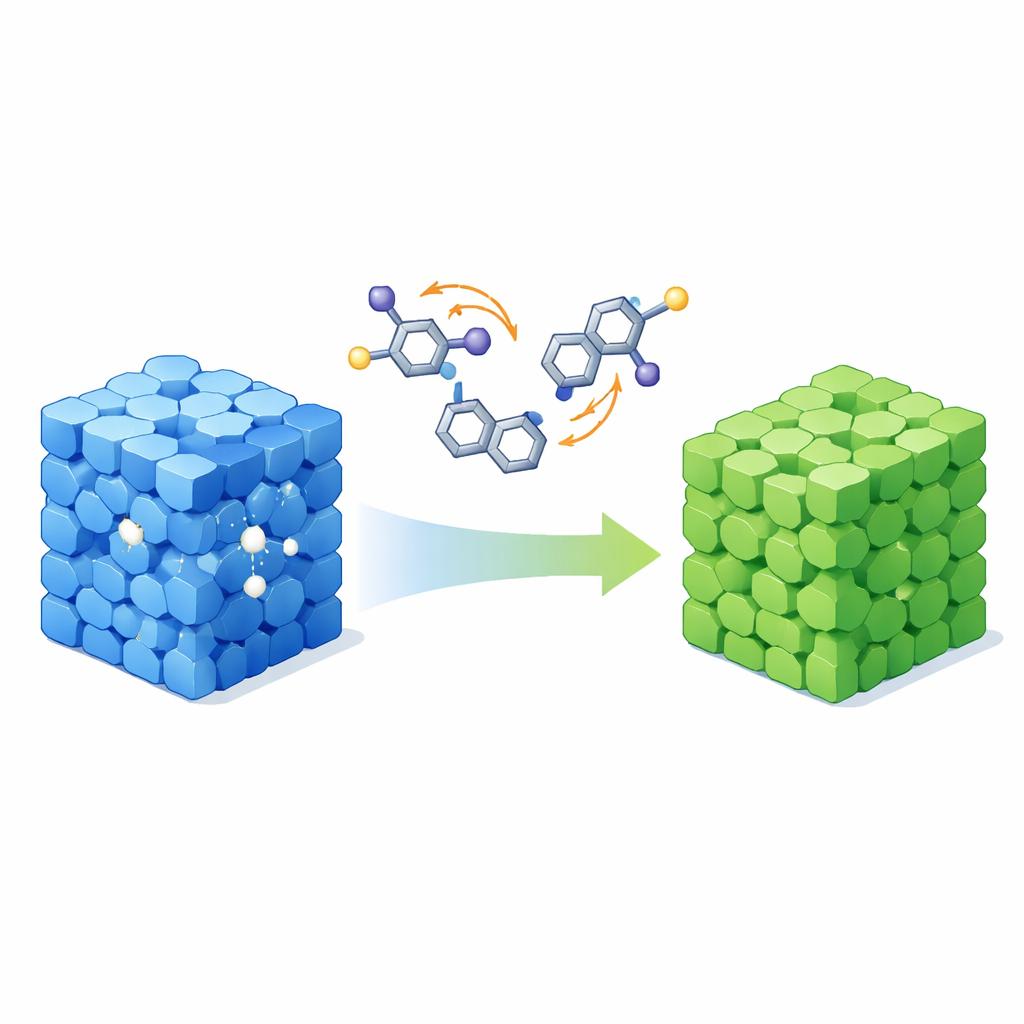
The researchers designed a single molecule, called BrGlu, that forms crystals with two distinct personalities. Under normal crystallization, it grows as a green‑emitting solid, called the G‑crystal. When crystallized in the presence of chloroform, it instead forms a blue‑emitting version, the B‑crystal, that includes solvent molecules in its structure. Both forms shine purely from organic phosphorescence at room temperature and remain bright: the green crystals emit with very high efficiency, while the blue crystals are still strongly glowing. Crucially, the material can be toggled between these two states under mild conditions. Dissolving and recrystallizing in specific halogenated solvents turns green crystals into blue ones, while gentle heating removes the solvent and restores the green form, allowing fully reversible color switching.
Tiny Shape Changes Control the Color
At the heart of this behavior is a subtle twist in the molecule’s shape inside the crystal. BrGlu carries bromine atoms and carbonyl groups whose relative orientation can adopt two arrangements, referred to as syn and anti. In the blue solvent‑rich crystals, solvent molecules sit in the lattice and form hydrogen‑bond‑like contacts that stabilize the syn arrangement, slightly raising the energy of the glowing state and shifting the phosphorescence toward blue. In the green solvent‑free crystals, the molecule relaxes into the anti arrangement, lowering that energy and producing greener light. X‑ray diffraction, Raman spectroscopy, and detailed quantum‑chemical calculations all point to this conformational flip as the key switch. The computed energy barrier between syn and anti is modest, explaining why mild heating or solvent treatment is enough to drive the reversible transformation without damaging the crystal.
Gentle Triggers with Surprising Selectivity
Not every solvent can coax the crystals into their blue state. Experiments with a range of halogenated liquids showed that only those with “activated” hydrogen atoms—such as chloroform, its deuterated cousin, bromoform, and tetrachloroethane—can trigger the dissolution–recrystallization cycle that yields the blue form. Solvents lacking suitable hydrogen donors, or even strongly polar alcohols and acetonitrile, failed to induce a phase change. Thermal measurements confirmed that the blue crystals indeed contain trapped solvent that is released around 65–70 °C, converting them back into the robust green form, which stays structurally intact up to much higher temperatures. Repeated cycling between blue and green produced almost no loss of brightness or shift in color, demonstrating that the system is durable enough for repeated use.
Hiding Messages in Time, Space, and Color
Using these properties, the team built proof‑of‑concept encryption devices. In one demonstration, patterns made from BrGlu crystals and a conventional fluorescent dye show a misleading message under ultraviolet light. After a brief heating step and then switching the UV lamp off, only BrGlu’s long‑lasting green afterglow remains, revealing the true pattern. In a second “3D” scheme, a grid of BrGlu pixels is selectively exposed to different solvents so that some regions switch from green to blue faster than others. Reading the pattern at carefully chosen times decodes hidden words, while reading it too early or too late yields meaningless output. A short heating step erases the solvent and resets the grid for reuse. Together, these demonstrations show how a single, metal‑free organic crystal that can reversibly change its glow color under gentle stimuli can underpin multi‑layered, difficult‑to‑forge data encryption and anti‑counterfeiting technologies.
Citation: Heo, JM., Woo, H., Flórez-Angarita, M.F. et al. Reversible color switching of bright phosphorescence in purely organic materials for advanced data encryption. Nat Commun 17, 3039 (2026). https://doi.org/10.1038/s41467-025-65225-w
Keywords: room temperature phosphorescence, stimuli responsive crystals, organic data encryption, anti counterfeiting materials, solvent induced color switching