Clear Sky Science · en
Sample delivery methods for protein X-ray crystallography with a special focus on sample consumption
Watching Molecules in Action
X-ray crystallography lets scientists see the arrangement of atoms in proteins, the tiny machines that power life. A newer approach, called serial crystallography, goes a step further: it can capture “molecular movies” of proteins in action, such as enzymes processing a drug or photosynthetic proteins splitting water. But there is a catch. Many important proteins are hard to make and crystallize, and current experiments can burn through milligrams to grams of precious material. This review article asks a deceptively simple question: how can we deliver crystals into powerful X-ray beams while wasting as little sample as possible?
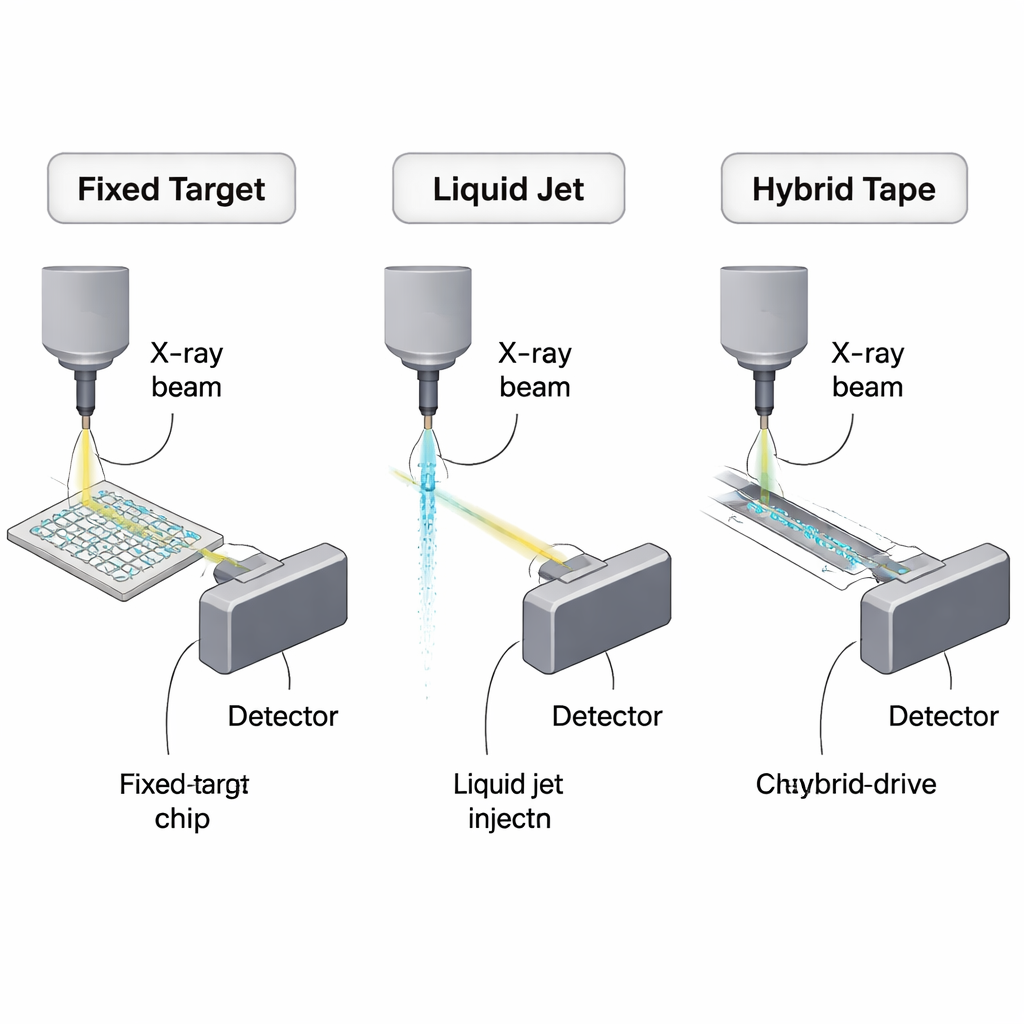
Why Serial Crystallography Needs Better Delivery
Traditional crystallography relied on a single large crystal rotated in an X-ray beam. Serial crystallography flips the script: instead of one big crystal, thousands of microcrystals, each used only once, are shot or scanned through ultrabright X-ray pulses from synchrotrons or X-ray free-electron lasers (XFELs). This enables room‑temperature data collection and fast “snapshots” of chemical reactions, but it also means constantly replenishing crystals at rates matching X-ray pulse trains that can reach up to millions of pulses per second. Much of the crystal suspension never actually meets the beam and is discarded, so reducing sample consumption has become a central technical and economic challenge for the field.
Fixed Targets: Tiny Chips That Stretch Every Drop
One major strategy is to immobilize microcrystals on small solid supports called fixed targets. Instead of spraying crystals past the beam, researchers arrange them in arrays on silicon or polymer chips and move the chip so that each crystal is brought into the X-ray focus once. In the best‑case thought experiment, about 10,000 microcrystals of a model protein could, in principle, yield a complete dataset using only about 450 nanograms of protein. Real devices are not yet that frugal, but they already cut needs to tens of micrograms to a few tenths of a milligram—orders of magnitude better than early serial experiments. The review compares silicon meshes, ultrathin polymer films, and multilayer plastic chips, weighing their strengths (low background scattering, on‑chip crystal growth, compatibility with room‑temperature studies) against practical issues like dehydration, stray scattering from the chip material, and extra “dead volume” introduced by manual pipetting.
Liquid Jets and Viscous Streams: Fast but Hungry
Another family of methods keeps crystals suspended in liquid and continuously delivers them across the beam. Gas‑dynamic virtual nozzles create hair‑thin jets that can keep up with fast XFEL pulse trains, making them a workhorse for time‑resolved studies and mix‑and‑inject experiments where reactions are triggered by rapid mixing just before exposure. However, because jets run continuously, most of the stream never meets an X-ray pulse. Even with careful tuning, practical experiments consume far more protein than the theoretical minimum—typically tens to hundreds of microliters of concentrated crystal slurry. To soften the blow, researchers have developed more efficient designs, such as double‑flow nozzles that sheath the crystal stream with a sacrificial liquid, electrospinning “MESH” injectors that work at lower flow rates, and high‑viscosity extruders that push crystals embedded in gels or lipidic cubic phases at microliter‑per‑minute or slower. These viscous methods are especially valuable for fragile membrane proteins and for room‑temperature studies at synchrotrons, but their thicker streams increase background scattering and are less suited to the very fastest X-ray sources.
Droplets, Tapes, and Hybrids: Matching Pulses One by One
A third, increasingly creative, class of “hybrid” approaches combines solid supports with controlled liquid or droplet delivery. Tape‑drive systems, for example, deposit droplets or thin liquid stripes onto a moving polymer film that passes through the beam; timing the motion allows reaction steps or gas exposure to be probed at defined delays. Drop‑on‑demand systems go further, using acoustic or piezoelectric devices to eject nanoliter or even picoliter droplets only when an X-ray pulse is expected, dramatically cutting waste. Some designs mix a ligand droplet into a pre‑placed crystal‑containing droplet on tape just before it reaches the beam, enabling time‑resolved enzymology with carefully rationed reagents. Other hybrids, like the LAMA on‑chip method, add tiny droplets of substrate directly onto crystals pre‑mounted on fixed‑target chips. Across these designs, reported protein usage spans a wide range—from near‑milligram levels down to a few milligrams for full time‑resolved series—showing both the promise and the current engineering challenges of synchronizing droplets, crystals, and X-ray pulses.
How Close Are We to the Theoretical Minimum?
By comparing dozens of published experiments across fixed targets, liquid injectors, and hybrid systems, the authors show that no existing method comes close to the ideal 450‑nanogram benchmark; even the best devices still overshoot by roughly two orders of magnitude. Yet clear trends emerge. Fixed targets typically use the least protein and are attractive whenever time‑resolved mixing is not essential or can be engineered on‑chip. Liquid jets still dominate the most demanding time‑resolved XFEL studies but remain sample‑intensive, especially when many time points are needed. Hybrid droplet‑and‑tape schemes offer some of the largest relative savings, especially when droplet timing is tightly synchronized to the X-ray source. Looking ahead, the article argues that further progress will come from better microfluidic control, automation to eliminate wasteful handling steps, and the use of compact X-ray sources and data‑driven optimization to co‑design experiments and delivery systems that push protein usage ever closer to the theoretical limit.
Citation: Manna, A., Doppler, D., Sripati, M.P. et al. Sample delivery methods for protein X-ray crystallography with a special focus on sample consumption. Nat Commun 16, 9856 (2025). https://doi.org/10.1038/s41467-025-65173-5
Keywords: serial crystallography, protein X-ray crystallography, sample delivery, X-ray free-electron laser, microfluidics